Abstract
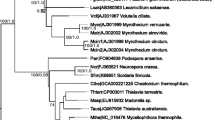
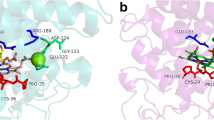
Recently, a novel group of fungal peroxidases, known as the aromatic peroxygenases (APO), has been discovered. Members of these extracellular biocatalysts produced by agaric basidiomycetes such as Agrocybe aegerita or Coprinellus radians catalyze reactions—for example, the peroxygenation of naphthalene, toluene, dibenzothiophene, or pyridine—which are actually attributed to cytochrome P450 monooxygenases. Here, for the first time, genetic information is presented on this new group of peroxide-consuming enzymes. The gene of A. aegerita peroxygenase (apo1) was identified on the level of messenger RNA and genomic DNA. The gene sequence was affirmed by peptide sequences obtained through an Edman degradation and de novo peptide sequencing of the purified enzyme. Quantitative real-time reverse transcriptase polymerase chain reaction demonstrated that the course of enzyme activity correlated well with that of mRNA signals for apo1 in A. aegerita. The full-length sequences of A. aegerita peroxygenase as well as a partial sequence of C. radians peroxygenase confirmed the enzymes’ affiliation to the heme-thiolate proteins. The sequences revealed no homology to classic peroxidases, cytochrome P450 enzymes, and only little homology (<30%) to fungal chloroperoxidase produced by the ascomycete Caldariomyces fumago (and this only in the N-terminal part of the protein comprising the heme-binding region and part of the distal heme pocket). This fact reinforces the novelty of APO proteins. On the other hand, homology retrievals in genetic databases resulted in the identification of various APO homologous genes and transcripts, particularly among the agaric fungi, indicating APO’s widespread occurrence in the fungal kingdom.





Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl Acids Res 25:3389–3402
Anh DH, Ullrich R, Benndorf D, Svatoś A, Muck A, Hofrichter M (2007) The coprophilous mushroom Coprinus radians secretes a haloperoxidase that catalyzes aromatic peroxygenation. Appl Environ Microbiol 73:5477–5485
Aranda E, Kinne M, Kluge M, Ullrich R, Hofrichter M (2009) Conversion of dibenzothiophene by the mushrooms Agrocybe aegerita and Coprinellus radians and their extracellular peroxygenases. Appl Microbiol Biotechnol 82:1057–1066
Bannai H, Tamada Y, Maruyama O, Nakai K, Miyano S (2002) Extensive feature detection of N-terminal protein sorting signals. Bioinformatics 18:298–305
Bennett-Lovsey RM, Herbert AD, Sternberg MJE, Kelley LA (2008) Exploring the extremes of sequence/structure space with ensemble fold recognition in the program Phyre. Proteins 70:611–625
Bernhardt R (2006) Cytochromes P450 as versatile biocatalysts. J Biotechnol 124:128–145
Brown DW, Yu JH, Kelkar HS, Fernandes M, Nesbitt TC, Keller NP, Adams TH, Leonard TJ (1996) Twenty-five coregulated transcripts define a sterigmatocystin gene cluster in Aspergillus nidulans. Proc Natl Acad Sci U S A 93:1418–1422
Conesa A, van de Velde F, van Rantwijk F, Sheldon RA, van den Hondel CA, Punt PJ (2001) Expression of the Caldariomyces fumago chloroperoxidase in Aspergillus niger and characterization of the recombinant enzyme. J Biol Chem 276:17635–17640
Cooper B, Neelam A, Campbell KB, Lee J, Liu G, Garrett WM, Scheffler B, Tucker ML (2007) Protein accumulation in the germinating Uromyces appendiculatus uredospore. Mol Plant Microbe Interact 20:857–866
Espagne E, Lespinet O, Malagnac F, da Silva C, Jaillon O, Porcel BM, Couloux A, Aury JM, Seguren B, Poulain J, Anthouard V, Grossetete S, Khalili H, Coppin E, Dequard-Chablat M, Picard M, Contamine V, Arnaise S, Bourdais A, Berteaux-Lecellier V, Gautheret D, de Vries RP, Battaglia E, Coutinho PM, Danchin EG, Henrissat B, Khoury RE, Sainsard-Chanet A, Boivin A, Pinan-Lucarré B, Sellem CH, Debuchy R, Wincker P, Weissenbach J, Silar P (2008) The genome sequence of the model ascomycete fungus Podospora anserina. Genome Biol 9:R77 Available via: http://genomebiology.com/2008/9/5/R77
Felsenstein J (1989) PHYLIP—Phylogeny Inference Package (Version 3.2). Cladistics 5:164–166
Felsenstein J (2005) PHYLIP (Phylogeny Inference Package) version 3.6. Distributed by the author. Department of Genome Sciences, University of Washington, Seattle
Frohman MA, Dush MK, Martin GR (1988) Rapid production of full-length cDNAs from rare transcripts: amplification using a single gene-specific oligonucleotide primer. Proc Natl Acad Sci U S A 85:8998–9002
Gasteiger E, Hoogland C, Gattiker A, Duvaud S, Wilkins MR, Appel RD, Bairoch A (2005) Protein identification and analysis tools on the ExPASy server. In: Walker JM (ed) The proteomics protocols handbook. Humana, Clifton, NJ, pp 571–607
González S, Pabón ML, Carulla J (2002) Effects of tannins on in vitro ammonia release and dry matter degradation of soybean meal. Arch Latinoam Prod Anim 10:97–101
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hane JK, Lowe RG, Solomon PS, Tan KC, Schoch CL, Spatafora JW, Crous PW, Kodira C, Birren BW, Galagan JE, Torriani SF, McDonald BA, Oliver RP (2007) Dothideomycete plant interactions illuminated by genome sequencing and EST analysis of the wheat pathogen Stagonospora nodorum. Plant Cell 19:3347–3368
Hofrichter M, Ullrich R (2006) Heme-thiolate haloperoxidases: versatile biocatalysts with biotechnological and environmental significance. Appl Microbiol Biotechnol 71:276–288
Hulo N, Bairoch A, Bulliard V, Cerutti L, Cuche BA, de Castro E, Lachaize C, Langendijk-Genevaux PS, Sigrist CJA (2008) The 20 years of PROSITE. Nucleic Acids Res 36(Database issue):D245–249. doi:https://doi.org/10.1093/nar/gkm977
Julenius K, Mølgaard A, Gupta R, Brunak S (2005) Prediction, conservation analysis and structural characterization of mammalian mucin-type O-glycosylation sites. Glycobiology 15:153–164
Kincaid J, Stein P, Spiro TG (1979) Absence of heme-localized strain in T state hemoglobin: Insensitivity of heme-imidazole resonance Raman frequencies to quaternary structure. Proc Natl Acad Sci U S A 76:549–552
Kinne M, Ullrich R, Hammel KE, Hofrichter M (2008) Regioselective preparation of (R)-2-(4-hydroxyphenoxy) propionic acid with a fungal peroxygenase. Tetrahedron Lett 49:5950–5953
Kluge M, Ullrich R, Scheibner K, Hofrichter M (2007) Spectrophotometric assay for detection of aromatic hydroxylation catalyzed by fungal haloperoxidase–peroxygenase. Appl Microbiol Biotechnol 75:1473–1478
Kluge M, Ullrich R, Dolge C, Scheibner K, Hofrichter M (2009) Hydroxylation of naphthalene by aromatic peroxygenase from Agrocybe aegerita proceeds via oxygen transfer from H2O2 and intermediary epoxidation. Appl Microbiol Biotechnol 81:1071–1076
Lambilliotte R, Cooke R, Samson D, Fizames C, Gaymard F, Plassard C, Tatry MV, Berger C, Laudie M, Legeai F, Karsenty E, Delseny M, Zimmermann S, Sentenac H (2004) Large scale identification of genes in the fungus Hebeloma cylindrosporum paves the way to molecular analyses of ectomycorrhizal symbiosis. New Phytol 164:505–513
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948. doi:https://doi.org/10.1093/bioinformatics/btm404
Lee SH, Kim BG, Kim KJ, Lee JS, Yun DW, Hahn JH, Kim GH, Lee KH, Suh DS, Kwon ST, Lee CS, Yoo YB (2002) Comparative analysis of sequences expressed during the liquid-cultured mycelia and fruit body stages of Pleurotus ostreatus. Fungal Genet Biol 35:115–134
Matz M, Shagin D, Bogdanova E, Britanova O, Lukyanov S, Diatchenko L, Chenchik A (1999) Amplification of cDNA ends based on template-switching effect and step-out PCR. Nucleic Acids Res 27:1558–1560
Nikolcheva LG, Bärlocher F (2002) Phylogeny of Tetracladium based on 18S rDNA. Czech Mycol 53:285–295
Omura T (2005) Heme-thiolate proteins. Biochem Biophys Res Commun 338:404–409
Ortiz de Montellano PRO (1992) Catalytic sites of hemoprotein peroxidases. Annu Rev Pharmacol Toxicol 32:89–107
Posada-Buitrago ML, Frederick RD (2005) Expressed sequence tag analysis of the soybean rust pathogen Phakopsora pachyrhizi. Fungal Genet Biol 42:949–962
Rychlik W, Rhoads RE (1989) A computer program for choosing optimal oligonucleotides for filter hybridization, sequencing and in vitro amplification of DNA. Nucleic Acids Res 17:8543–8551
Semova N, Storms R, John T, Gaudet P, Ulycznyj P, Min XJ, Sun J, Bulter G, Tsang A (2006) Generation, annotation, and analysis of an extensive Aspergillus niger EST collection. BMC Microbiol 6:7. doi:https://doi.org/10.1186/1471-2180-6-7
Saloniemi H, Wähälä K, Nykänen-Kurki P, Kallela K, Saastamoinen I (1995) Phytoestrogen content and estrogenic effect of legume fodder. Proc Soc Exp Biol Med 208:13–17
Shapiro MB, Senapathy P (1987) RNA splice junctions of different classes of eukaryotes: sequence statistics and functional implications in gene expression. Nucleic Acids Res 15:7155–7174
Sundaramoorthy M, Terner J, Poulos TL (1995) The crystal structure of chloroperoxidase: a heme peroxidase-cytochrome P450 functional hybrid. Structure 3:1367–1377
Ullrich R, Nüske J, Scheibner K, Spantzel J, Hofrichter M (2004) Novel haloperoxidase from the agaric basidiomycete Agrocybe aegerita oxidizes aryl alcohols and aldehydes. Appl Environ Microbiol 70:4575–4581
Ullrich R, Hofrichter M (2005) The haloperoxidase of the agaric fungus Agrocybe aegerita hydroxylates toluene and naphthalene. FEBS Lett 579:6247–6250
Ullrich R, Hofrichter M (2007) Enzymatic hydroxylation of aromatic compounds. Cell Mol Life Sci 64:271–293
Ullrich R, Dolge C, Kluge M, Hofrichter M (2008) Pyridine as novel substrate for regioselective oxygenation with aromatic peroxygenase from Agrocybe aegerita. FEBS Lett 582:4100–4106
Wilkins MR, Lindskog I, Gasteiger E, Bairoch A, Sanchez J-C, Hochstrasser DF, Appel RD (1997) Detailed peptide characterisation using PEPTIDEMASS—a World-Wide Web accessible tool. Electrophoresis 18:403–408
Woodman ME (2008) Direct PCR of intact bacteria (colony PCR). Curr Protoc Microbiol Appendix 3:Appendix 3D
Wu Q, Wang M, Simon JE (2004) Analytical methods to determine phytoestrogenic compounds. J Chromatogr B Analyt Technol Biomed Life Sci 812:325–355
Zhao J, Hyman L, Moore C (1999) Formation of mRNA 3′ ends in eukaryotes: mechanism, regulation, and interrelationships with other steps in mRNA synthesis. Microbiol Mol Biol R 63:405–445
Zhou NY, Jenkins A, Chan Kwo Chion CK, Leak DJ (1999) The alkene monooxygenase from Xanthobacter strain Py2 is closely related to aromatic monooxygenases and catalyzes aromatic monohydroxylation of benzene, toluene, and phenol. Appl Environ Microbiol 65:1589–1595
Acknowledgments
We gratefully acknowledge the financial support from the German Environmental Foundation (Deutsche Bundesstiftung Umwelt; M.P., M.H.; grant 13225-32), the German Research Foundation (Deutsche Forschungsgemeinschaft; M.P., M.H.; grant HO1961/4-1), and the Federal Ministry of Education and Research (Bundesministerium für Bildung und Forschung; R.U., A.C., R.S., K.S., M.H.; grant 0313433D). We would like to thank Mss. U. Schneider, H. Hohlfeld, M. Brandt, and S. Schimpke for their excellent technical assistance and our colleagues E. Aranda, C. Liers, M. Kinne, M. Kluge (Inge), and R. Junek for fruitful discussions. Not least, we thank G. Gröbe for useful pieces of information on fungal cultivation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pecyna, M.J., Ullrich, R., Bittner, B. et al. Molecular characterization of aromatic peroxygenase from Agrocybe aegerita . Appl Microbiol Biotechnol 84, 885–897 (2009). https://doi.org/10.1007/s00253-009-2000-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2000-1