Abstract
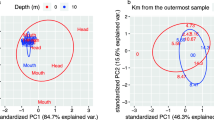
In intertidal sediments, circadian oscillations (i.e., tidal and diel rhythms) and/or depth may affect prokaryotic activity. However, it is difficult to distinguish the effect of each single force on active community changes in these natural and complex intertidal ecosystems. Therefore, we developed a tidal mesocosm to control the tidal rhythm and test whether diel fluctuation or sediment depth influence active prokaryotes in the top 10 cm of sediment. Day- and nighttime emersions were compared as they are expected to display contrasting conditions through microphytobenthic activity in five different sediment layers. A multiple factor analysis revealed that bacterial and archaeal 16S ribosomal RNA (rRNA) transcript diversity assessed by pyrosequencing was similar between day and night emersions. Potentially active benthic Bacteria were highly diverse and influenced by chlorophyll a and phosphate concentrations. While in oxic and suboxic sediments, Thaumarchaeota Marine Group I (MGI) was the most active archaeal phylum, suggesting the importance of the nitrogen cycle in muddy sediments, in anoxic sediments, the mysterious archaeal C3 group dominated the community. This work highlighted that active prokaryotes organize themselves vertically within sediments independently of diel fluctuations suggesting adaptation to physicochemical-specific conditions associated with sediment depth.




Similar content being viewed by others
References
Kirchman DL, Dittel AI, Malmstrom RR, Cottrell MT (2005) Biogeography of major bacterial groups in the Delaware Estuary. Limnol Oceanogr 50:1697–1706
Böer S, Hedtkamp SI, van Beusekom JE, et al. (2009) Time- and sediment depth-related variations in bacterial diversity and community structure in subtidal sands. ISME J 3:780–791. doi:10.1038/ismej.2009.29
Herlemann DPR, Labrenz M, Jurgens K, et al. (2011) Transitions in bacterial communities along the 2000 km salinity gradient of the Baltic Sea. ISME J 5:1571–1579 http://www.nature.com/ismej/journal/v5/n10/suppinfo/ismej201141s1.html
Liu J, Yang H, Zhao M, Zhang XH (2014) Spatial distribution patterns of benthic microbial communities along the Pearl Estuary, China. Syst Appl Microbiol 37:578–589. doi:10.1016/j.syapm.2014.10.005
Gobet A, Boer S, Huse SM, et al. (2012) Diversity and dynamics of rare and of resident bacterial populations in coastal sands. ISME J 6:542–553. doi:10.1038/ismej.2011.132
Consalvey M, Paterson DM, Underwood GJC (2004) The ups and downs of life in a benthic biofilm: migration of benthic diatoms. Diatom Res 19:181–202. doi:10.1080/0269249X.2004.9705870
Ritzrau W, Graf G (1992) Increase of microbial biomass in the benthic turbidity zone of Kiel Bight after resuspension by a storm event. Limnol Oceanogr 37:1081–1086. doi:10.4319/lo.1992.37.5.1081
Friend PL, Collins MB, Holligan PM (2003) Day–night variation of intertidal flat sediment properties in relation to sediment stability. Estuar Coast Shelf Sci 58:663–675. doi:10.1016/S0272-7714(03)00178-1
Friend PL, Lucas CH, Rossington SK (2005) Day–night variation of cohesive sediment stability. Estuar Coast Shelf Sci 64:407–418
Guizien K, Dupuy C, Ory P, et al. (2014) Microorganism dynamics during a rising tide: disentangling effects of resuspension and mixing with offshore waters above an intertidal mudflat. J Mar Syst 129:178–188. doi:10.1016/j.jmarsys.2013.05.010
Harrison SJ, Phizacklea AP (1987) Vertical temperature gradients in muddy intertidal sediments in the forth estuary, Scotland1. Limnol Oceanogr 32:954–963. doi:10.4319/lo.1987.32.4.0954
Montagna PA, Bauer JE, Hardin D, Spies RB (1989) Vertical distribution of microbial and meiofaunal populations in sediments of a natural coastal hydrocarbon seep. J Mar Res 47:657–680. doi:10.1357/002224089785076226
Edlund A, Hårdeman F, Jansson JK, Sjöling S (2008) Active bacterial community structure along vertical redox gradients in Baltic Sea sediment. Environ Microbiol 10:2051–2063. doi:10.1111/j.1462-2920.2008.01624.x
Fan X, Xing P (2016) The vertical distribution of sediment archaeal community in the “Black Bloom” Disturbing Zhushan Bay of Lake Taihu Archaea. doi:10.1155/2016/8232135
Hewson I, Vargo GA, Fuhrman JA (2003) Bacterial diversity in shallow oligotrophic marine benthos and overlying waters: effects of virus infection, containment, and nutrient enrichment. Microb Ecol 46:322–336. doi:10.1007/s00248-002-1067-3
Kristensen E (2000) Organic matter diagenesis at the oxic/anoxic interface in coastal marine sediments, with emphasis on the role of burrowing animals. Hydrobiologia 426:1–24. doi:10.1023/A:1003980226194
Laverock B, Gilbert JA, Tait K, et al. (2011) Bioturbation: impact on the marine nitrogen cycle. Biochem Soc Trans 39:315–320
Passarelli C, Olivier F, Paterson DM, et al. (2014) Organisms as cooperative ecosystem engineers in intertidal flats. J Sea Res 92:92–101. doi:10.1016/j.seares.2013.07.010
Risgaard-Petersen N (2003) Coupled nitrification-denitrification in autotrophic and heterotrophic estuarine sediments: on the influence of benthic microalgae. Limnol Oceanogr 48:93–105. doi:10.4319/lo.2003.48.1.0093
Hochard S, Pinazo C, Grenz C, et al. (2010) Impact of microphytobenthos on the sediment biogeochemical cycles: a modeling approach. Ecol Model 221:1687–1701. doi:10.1016/j.ecolmodel.2010.04.002
Admiraal W (1984) The ecology of estuarine sediment inhabiting diatoms. Prog Phycol Res 3:269–314
Blanchard GF, Guarini J-M, Orvain F, Sauriau P-G (2001) Dynamic behaviour of benthic microalgal biomass in intertidal mudflats. J Exp Mar Bio Ecol 264:85–100. doi:10.1016/s0022-0981(01)00312-4
Underwood GJC, Paterson DM (2003) The importance of extracellular carbohydrate production by marine epipelic diatoms. Adv Bot Res 40:183–240. doi:10.1016/S0065-2296(05)40005-1
Herlory O, Guarini J-M, Richard P, Blanchard G (2004) Microstructure of microphytobenthic biofilm and its spatio-temporal dynamics in an intertidal mudflat (Aiguillon Bay, France). Mar Ecol Prog Ser 282:33–44
Haynes K, Hofmann TA, Smith CJ, et al. (2007) Diatom-derived carbohydrates as factors affecting bacterial community composition in estuarine sediments. Appl Environ Microbiol 73:6112–6124. doi:10.1128/aem.00551-07
Saburova MA, Polikarpov IG (2003) Diatom activity within soft sediments: behavioural and physiological processes. Mar Ecol Prog Ser 251:15–126
Thornton DCO, Underwood GJ, Nedwell DB (1999) Effect of illumination and emersion period on the exchange of ammonium across the estuarine sediment-water interface. Mar Ecol Prog Ser 184:11–20. doi:10.3354/meps184011
Lindström ES, Langenheder S (2012) Local and regional factors influencing bacterial community assembly. Environ Microbiol Rep 4:1–9. doi:10.1111/j.1758-2229.2011.00257.x
Michaud E, Aller RC, Stora G (2010) Sedimentary organic matter distributions, burrowing activity, and biogeochemical cycling: natural patterns and experimental artifacts. Estuar Coast Shelf Sci 90:21–34
Agogué H, Mallet C, Orvain F, et al. (2014) Bacterial dynamics in a microphytobenthic biofilm: a tidal mesocosm approach. J Sea Res 92:36–45. doi:10.1016/j.seares.2014.03.003
Lorenzen CJ (1966) A method for the continuous measurement of in vivo chlorophyll concentration. Deep Res Oceanogr Abstr 13:223–227
Muyzer G, de Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Einen J, Thorseth IH, Øvreås L (2008) Enumeration of Archaea and Bacteria in seafloor basalt using real-time quantitative PCR and fluorescence microscopy. FEMS Microbiol Lett 282:182–187. doi:10.1111/j.1574-6968.2008.01119.x
Muyzer G, Teske A, Wirsen C, Jannasch H (1995) Phylogenetic relationships of Thiomicrospira species and their identification in deep-sea hydrothermal vent samples by denaturing gradient gel electrophoresis of 16S rDNA fragments. Arch Microbiol 164:165–172. doi:10.1007/bf02529967
Herfort L, Schouten S, Abbas B, et al. (2007) Variations in spatial and temporal distribution of Archaea in the North Sea in relation to environmental variables. FEMS Microbiol Ecol 62:242–257. doi:10.1111/j.1574-6941.2007.00397.x
Casamayor EO, Massana R, Benlloch S, et al. (2002) Changes in archaeal, bacterial and eukaryal assemblages along a salinity gradient by comparison of genetic fingerprinting methods in a multipond solar saltern. Environ Microbiol 4:338–348. doi:10.1046/j.1462-2920.2002.00297.x
Giongo A, Crabb DB, Davis-Richardson AG, et al. (2010) PANGEA: pipeline for analysis of next generation amplicons. Isme J 4:852–861. doi:10.1038/ismej.2010.16
Edgar RC, Haas BJ, Clemente JC, et al. (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. doi:10.1093/bioinformatics/btr381
Kim M, Morrison M, Yu Z (2011) Evaluation of different partial 16S rRNA gene sequence regions for phylogenetic analysis of microbiomes. J Microbiol Methods 84:81–87. doi:10.1016/j.mimet.2010.10.020
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461. doi:10.1093/bioinformatics/btq461
Price MN, Dehal PS, Arkin AP (2009) FastTree: computing large minimum evolution trees with profiles instead of a distance matrix. Mol Biol Evol 26:1641–1650. doi:10.1093/molbev/msp077
Pruesse E, Quast C, Knittel K, et al. (2007) SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res 35:7188–7196. doi:10.1093/nar/gkm864
Taib N, Mangot J-F, Domaizon I, et al. (2013) Phylogenetic affiliation of SSU rRNA genes generated by massively parallel sequencing: new insights into the freshwater protist diversity. PLoS One 8:e58950. doi:10.1371/journal.pone.0058950
Legendre P, Legendre L (1998) Numerical ecology. Developments in environmental modelling. Elsevier Science & Technology, Amsterdam
Dray S, Legendre P, Blanchet G (2013) packfor: Forward Selection with permutation (Canoco p.46). R package version 0.0-8/r109. http://R-Forge.R-project.org/projects/sedar/
Escofier B, Pagès J (1994) Multiple factor analysis (AFMULT package). Comput Stat Data Anal 18:121–140. doi:10.1016/0167-9473(94)90135-X
Husson F, Josse J, Lê S, Mazet J (2007) FactoMineR: Factor Analysis and Data Mining with R. R package version 1.04. URL http://CRAN.R-project.org/package=FactoMineR
Dray S, Dufour A-B (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22:1–20
Oksanen J, Blanchet FG, Kindt R, et al (2013) vegan: community ecology package. R Package version 2.0–7. https://cran.r-project.org/web/packages/vegan/index.html
Spang A, Saw JH, Jørgensen SL, et al. (2015) Complex archaea that bridge the gap between prokaryotes and eukaryotes. Nature. doi:10.1038/nature14447
Kubo K, Lloyd KG, Biddle JF, et al. (2012) Archaea of the Miscellaneous Crenarchaeotal Group are abundant, diverse and widespread in marine sediments. ISME J 6:1949–1965. doi:10.1038/ismej.2012.37
Lozupone CA, Knight R (2007) Global patterns in bacterial diversity. Proc Natl Acad Sci 104:11436–11440. doi:10.1073/pnas.0611525104
Zinger L, Amaral-Zettler LA, Fuhrman JA, et al. (2011) Global patterns of bacterial beta-diversity in seafloor and seawater ecosystems. PLoS One 6:e24570. doi:10.1371/journal.pone.0024570
Kopf A, Bicak M, Kottmann R, et al. (2015) The ocean sampling day consortium. Gigascience 4:1–5. doi:10.1186/s13742-015-0066-5
Pesant S, Not F, Picheral M, et al. (2015) Open science resources for the discovery and analysis of Tara Oceans data. Sci Data. doi:10.1038/sdata.2015.23
Poulin M, Massé G, Belt ST, et al. (2004) Morphological, biochemical and molecular evidence for the transfer of Gyrosigma nipkowii Meister to the genus Haslea (Bacillariophyta). Eur J Phycol 39:181–195. doi:10.1080/0967026042000202136
Blazewicz SJ, Barnard RL, Daly RA, Firestone MK (2013) Evaluating rRNA as an indicator of microbial activity in environmental communities: limitations and uses. ISME J 7:2061–2068. doi:10.1038/ismej.2013.102
Hugoni M, Agogué H, Taib N, et al. (2015) Temporal dynamics of active prokaryotic Nitrifiers and archaeal communities from river to sea. Microb Ecol 70:473–483. doi:10.1007/s00248-015-0601-z
Campbell LI, Rota-Stabelli O, Edgecombe GD, et al. (2011) MicroRNAs and phylogenomics resolve the relationships of Tardigrada and suggest that velvet worms are the sister group of Arthropoda. Proc Natl Acad Sci U S A 108:15920–15924. doi:10.1073/pnas.1105499108
Lavergne C, Agogué H, Leynaert A, et al. (2017) Factors influencing prokaryotes in an intertidal mudflat and the resulting depth gradients. Estuar Coast Shelf Sci 189C:74–83. doi:10.1016/j.ecss.2017.03.008
Brock J, Schulz-Vogt HN (2011) Sulfide induces phosphate release from polyphosphate in cultures of a marine Beggiatoa strain. ISME J 5:497–506. doi:10.1038/ismej.2010.135
Sun MY, Dafforn KA, Johnston EL, Brown MV (2013) Core sediment bacteria drive community response to anthropogenic contamination over multiple environmental gradients. Environ Microbiol 15:2517–2531. doi:10.1111/1462-2920.12133
Bowman JP, McCuaig RD (2003) Biodiversity, community structural shifts, and biogeography of prokaryotes within Antarctic continental shelf sediment. Appl Environ Microbiol 69:2463–2483. doi:10.1128/aem.69.5.2463-2483.2003
Ravenschlag K, Sahm K, Knoblauch C, et al. (2000) Community structure, cellular rRNA content, and activity of sulfate-reducing bacteria in marine Arctic sediments. Appl Environ Microbiol 66:3592–3602. doi:10.1128/aem.66.8.3592-3602.2000
Pfeffer C, Larsen S, Song J, et al. (2012) Filamentous bacteria transport electrons over centimetre distances. Nature 491:218–221 http://www.nature.com/nature/journal/v491/n7423/abs/nature11586.html#supplementary-information
Lee YK, Hong SG, Cho HH, et al. (2007) Dasania marina gen. nov., sp. nov., of the order Pseudomonadales, isolated from Arctic marine sediment. J Microbiol 45:505
Fuerst JA, Sagulenko E (2011) Beyond the bacterium: planctomycetes challenge our concepts of microbial structure and function. Nat Rev Micro 9:403–413
Schlesner H (1994) The development of media suitable for the microorganisms morphologically resembling Planctomyces spp., Pirellula spp., and other Planctomycetales from various aquatic habitats using dilute media. Syst Appl Microbiol 17:135–145. doi:10.1016/S0723-2020(11)80042-1
Papineau D, Walker JJ, Mojzsis SJ, Pace NR (2005) Composition and structure of microbial communities from stromatolites of Hamelin Pool in Shark Bay, Western Australia. Appl Environ Microbiol 71:4822–4832. doi:10.1128/AEM.71.8.4822-4832.2005
Kulichevskaya IS, Ivanova AO, Baulina OI, et al. (2008) Singulisphaera acidiphila gen. nov., sp. nov., a non-filamentous, Isosphaera-like planctomycete from acidic northern wetlands. Int J Syst Evol Microbiol 58:1186–1193. doi:10.1099/ijs.0.65593-0
Giovannoni SJ, Schabtach E, Castenholz RW (1987) Isosphaera pallida, gen. and comb. nov., a gliding, budding eubacterium from hot springs. Arch Microbiol 147:276–284. doi:10.1007/BF00463488
Strous M, Fuerst JA, Kramer EHM, et al. (1999) Missing lithotroph identified as new planctomycete. Nature 400:446–449
Devol AH (2003) Nitrogen cycle: solution to a marine mystery. Nature 422:575–576
Kuenen JG (2008) Anammox bacteria: from discovery to application. Nat Rev Micro 6:320–326
Kartal B, Kuypers MMM, Lavik G, et al. (2007) Anammox bacteria disguised as denitrifiers: nitrate reduction to dinitrogen gas via nitrite and ammonium. Environ Microbiol 9:635–642. doi:10.1111/j.1462-2920.2006.01183.x
Jetten MSM, van Niftrik L, Strous M, et al. (2009) Biochemistry and molecular biology of anammox bacteria. Crit Rev Biochem Mol Biol 44:65–84. doi:10.1080/10409230902722783
Agogué H, Brink M, Dinasquet J, Herndl GJ (2008) Major gradients in putatively nitrifying and non-nitrifying Archaea in the deep North Atlantic. Nature 456:788–791. doi:10.1038/nature07535
Santoro AE, Casciotti KL, Francis CA (2010) Activity, abundance and diversity of nitrifying archaea and bacteria in the central California current. Environ Microbiol 12:1989–2006. doi:10.1111/j.1462-2920.2010.02205.x
Villanueva L, Schouten S, Sinninghe Damsté JS (2015) Depth-related distribution of a key gene of the tetraether lipid biosynthetic pathway in marine Thaumarchaeota. Environ Microbiol 17:3527–3539. doi:10.1111/1462-2920.12508
Smith JM, Casciotti KL, Chavez FP, Francis CA (2014) Differential contributions of archaeal ammonia oxidizer ecotypes to nitrification in coastal surface waters. ISME J 8:1704–1714. doi:10.1038/ismej.2014.11
Damashek J, Smith JM, Mosier AC, Francis CA (2015) Benthic ammonia oxidizers differ in community structure and biogeochemical potential across a riverine delta. Front Microbiol 5:743. doi:10.3389/fmicb.2014.00743
Francis CA, Robert KJ, Beman JM, et al. (2005) Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. Proc Natl Acad Sci 102:14683–14688
Smith JM, Mosier A, Francis CA (2014) Spatiotemporal relationships between the abundance, distribution, and potential activities of ammonia-oxidizing and denitrifying microorganisms in intertidal sediments. Microb Ecol 1–12. doi: 10.1007/s00248-014-0450-1
Auguet J-C, Casamayor EO (2013) Partitioning of Thaumarchaeota populations along environmental gradients in high mountain lakes. FEMS Microbiol Ecol 84:154–164. doi:10.1111/1574-6941.12047
Hugoni M, Domaizon I, Taib N, et al. (2015) Temporal dynamics of active Archaea in oxygen-depleted zones of two deep lakes. Environ Microbiol Rep 7:321–329. doi:10.1111/1758-2229.12251
Jung M-Y, Park S-J, Kim S-J, et al. (2014) A mesophilic, autotrophic, ammonia-oxidizing archaeon of Thaumarchaeal group I.1a cultivated from a deep oligotrophic soil horizon. Appl Environ Microbiol 80:3645–3655. doi:10.1128/aem.03730-13
Prosser JI, Nicol GW, Venter JC, et al. (2012) Archaeal and bacterial ammonia-oxidisers in soil: the quest for niche specialisation and differentiation. Trends Microbiol 20:523–531. doi:10.1016/j.tim.2012.08.001
Dang H, Zhou H, Yang J, et al. (2013) Thaumarchaeotal signature gene distribution in sediments of the northern South China sea: an indicator of the metabolic intersection of the marine carbon, nitrogen, and phosphorus cycles? Appl Environ Microbiol 79:2137–2147. doi:10.1128/AEM.03204-12
Bale NJ, Villanueva L, Hopmans EC, et al. (2013) Different seasonality of pelagic and benthic Thaumarchaeota in the North Sea. Biogeosciences 10:7195–7206. doi:10.5194/bg-10-7195-2013
Stauffert M, Duran R, Gassie C, Cravo-Laureau C (2014) Response of archaeal communities to oil spill in bioturbated mudflat sediments. Microb Ecol 67:108–119. doi:10.1007/s00248-013-0288-y
Meng J, Xu J, Qin D, et al. (2014) Genetic and functional properties of uncultivated MCG archaea assessed by metagenome and gene expression analyses. ISME J 8:650–659. doi:10.1038/ismej.2013.174
Hawkins AN, Johnson KW, Bräuer SL (2014) Southern Appalachian peatlands support high archaeal diversity. Microb Ecol 67:587–602. doi:10.1007/s00248-013-0352-7
Hirayama H, Sunamura M, Takai K, et al. (2007) Culture-dependent and -independent characterization of microbial communities associated with a shallow submarine hydrothermal system occurring within a coral reef off Taketomi Island, Japan. Appl Environ Microbiol 73:7642–7656. doi:10.1128/aem.01258-07
Wemheuer B, Taube R, Akyol P, et al. (2013) Microbial diversity and biochemical potential encoded by thermal spring metagenomes derived from the Kamchatka peninsula. Archaea 2013:13. doi:10.1155/2013/136714
Fang J, Shizuka A, Kato C, Schouten S (2006) Microbial diversity of cold-seep sediments in Sagami Bay, Japan, as determined by 16S rRNA gene and lipid analyses. FEMS Microbiol Ecol 57:429–441. doi:10.1111/j.1574-6941.2006.00126.x
Dang H, Luan X-W, Chen R, et al. (2010) Diversity, abundance and distribution of amoA-encoding archaea in deep-sea methane seep sediments of the Okhotsk Sea. FEMS Microbiol Ecol 72:370–385. doi:10.1111/j.1574-6941.2010.00870.x
Acknowledgements
This research was supported by a PhD grant from the Charente Maritime Department and by the national program CPER 2006-2013 (Contrat Projet Etat Région) of Charente Maritime, the French national program EC2CO (CAPABIOC, 2012-2014), and the CNRS organism. The tidal mesocosms were built by M. Prineau and N. Lachaussée. We acknowledge the Molecular and the Cytometry Core Facilities at LIENSs laboratory. The authors are grateful to A. Leynaert (LEMAR, Brest, France) for the rhizon technique, to P. Pineau (LIENSs, La Rochelle, France) for the nutrient measurement, and to N. Lachaussée (LIENSs, La Rochelle, France) for field sampling. We also thank V. Becquet, C. Dussud, M. Bréret, J.C. Gama de Matos, and J. Lavaud (LIENSs, La Rochelle, France) who help us during the experiment. We thank N. Taib and JC. Charvy (LMGE, Clermont-Ferrand, France) for their help in phylogenetic analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Electronic Supplementary Material
ESM 1
(PDF 940 kb)
Rights and permissions
About this article
Cite this article
Lavergne, C., Hugoni, M., Hubas, C. et al. Diel Rhythm Does Not Shape the Vertical Distribution of Bacterial and Archaeal 16S rRNA Transcript Diversity in Intertidal Sediments: a Mesocosm Study. Microb Ecol 75, 364–374 (2018). https://doi.org/10.1007/s00248-017-1048-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-017-1048-1