Abstract
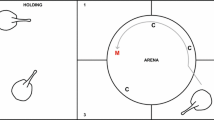
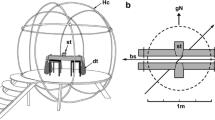
Elasmobranch fishes (sharks, skates, and rays) have been hypothesized to use the geomagnetic field (GMF) to maintain a sense of direction as they navigate throughout their environment. However, it is difficult to test the sensory ecology and spatial orientation ability of large highly migratory fishes in the field. Therefore, we performed behavioral conditioning experiments on a small magnetically sensitive species, the yellow stingray (Urobatis jamaicensis), in the laboratory. We trained individuals to use the polarity, or the north–south direction, of the GMF as a cue to orient in space and navigate a T-maze for a food reward. Subjects were split into two groups that learned to associate the direction of magnetic north or south as the indicator of the reward location. Stingrays reached the learning criterion within a mean (± SE) of 158.6 ± 28.4 trials. Subjects were then reverse trained to use the previously unrewarded magnetic stimulus of the opposite polarity as the new cue for the reward location. Overall, the stingrays reached the reversal criterion in significantly fewer trials (120 ± 13.8) compared to the initial procedure. These data show that the yellow stingray can learn to associate changes in GMF polarity with a reward, relearn a behavioral task when the reward contingency is modified, and learn a reversal procedure faster than the initial association. These data support the idea that the yellow stingray, and perhaps other elasmobranchs, might use GMF polarity as a cue to orient and maintain a heading during navigation.




Similar content being viewed by others
Data availability
The datasets created during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Able KP (1991) Common themes and variations in animal orientation systems. Amer Zool 31:157–167
Adrianov GN, Brown HR, Ilyinsky OB (1974) Responses of central neurons to electrical and magnetic stimuli of the ampullae of Lorenzini in the Black Sea skate. J Comp Physiol 93:287–299
Akoev GN, Ilyinsky OB, Zadan PM (1976) Responses of electroreceptors (ampullae of Lorenzini) of skates to electric and magnetic fields. J Comp Physiol 106:127–136
Anderson JM, Clegg TM, Véras LVMV, Holland KH (2017) Insight into shark magnetic field perception from empirical observations. Sci Rep 7:1042. https://doi.org/10.1038/s41598-017-11459-8
Berthold P (2001) Bird Migration: a general survey. Oxford University Press, Oxford, UK, p 272
Bitterman ME (1965) Phyletic differences in learning. Am Psychol 20:395–410
Bond AB, Kamil A, Balda RP (2007) Serial reversal learning and the evolution of behavioral flexibility in three species of North American corvids Gymnorhinus cyanocephalus, Nucifraga Columbiana, Aphelocoma californica. J Comp Psychol 121(4):372–379
Brown HR, Ilyinsky OB (1978) The ampullae of Lorenzini in the magnetic field. J Comp Physiol 126:333–341
Burgess N (2006) Spatial memory how egocentric and allocentric combine. Trends Cogn Sci 10:551–557
Chapman DD, Feldheim KA, Papastamatiou YP, Hueter RE (2015) There and back again: a review of residency and return migrations in sharks, with implications for population structure and management. Ann Rev Mar Sci 7:547–570
Cheng K (2012) How to navigate without maps: the power of taxon-like navigation in ants. Comp Cogn Behav Rev 7:1–22. https://doi.org/10.3819/ccbr.2012.70001
Chow PKY, Leaver LA, Wang M, Lea SEG (2015) Serial reversal learning in gray squirrels: learning efficiency as a function of learning and change of tactics. J Exp Psych 41:343–353
Daniel MMM, Schluessel V (2019) Serial reversal learning in freshwater stingrays (Potamotrygon motoro). Anim Cogn. https://doi.org/10.1007/s10071-019-01321
Day LB, Crews D, Wilczynski W (1999) Spatial and reversal learning in congeneric lizards with different foraging strategies. Anim Behav 57:393–407
Fahy DP (2004) Diel activity patterns, space utilization, seasonal distribution and population structure of the yellow stingray (Urobatis jamaicensis) in South Florida with comments on reproduction. (MS Thesis) Nova Southeastern University Oceanographic Center, Dania Beach, Florida, USA
Flowers KI, Ajemian MJ, Bassos-Hull K, Feldheim KA, Hueter RE, Papastamatiou YP, Chapman DD (2016) A review of batoid philopatry, with implications for future research and population management. Mar Ecol Prog Ser 562:251–261
Fuss T, Schluessel V (2018) Immediate early gene expression related to learning and retention of a visual discrimination task in bamboo sharks (Chiloscyllium griseum). Brain Struct Func 223:3975–4003. https://doi.org/10.1007/s00429-018-1728-8
Fuss T, Bleckmann H, Schluessel V (2014a) The shark (Chiloscyllium griseum) can orient using turn responses before and after partial telencephalon ablation. J Comp Physiol A 200:19–35
Fuss T, Bleckmann H, Schluessel V (2014b) Place learning prior to and after telencephalon ablation in bamboo and coral cat sharks (Chiloscyllium griseum and Atelomycterus marmoratus). J Comp Physiol A 200:37–52
Fuss T, Schluessel V, Bleckmann H (2014c) Visual discrimination abilities in the gray bamboo shark (Chiloscyllium griseum). Zool 117:104–111
Gardiner JM, Atema J, Hueter RE, Motta PJ (2014) Multisensory integration and behavioral plasticity in sharks from different ecological niches. PLoS ONE 9(4):e93036
Gould JL (1998) Sensory bases of navigation. Curr Biol 8:R731–R738
Gould JL (2004) Animal navigation. Curr Biol 14:R221–R224
Hueter RE, Heupel MR, Heist EJ, Keeney DB (2005) Evidence of philopatry in sharks and implications for the management of shark fisheries. J Northw Atl Fish Sci 35:239–247
Kalmijn AJ (1974) The detection of electric fields from inanimate and animate sources other than electric organs. In: Fessard A (ed) Handbook of sensory physiology III/3: electroreceptors and other specialized receptors in lower vertebrates. Heidelberg, Berlin, pp 147–200
Kalmijn AJ (1978) Experimental evidence of geomagnetic orientation in elasmobranch fishes. In: Schmidt-Koening K, Keaton WT (eds) Animal Migration, Navigation, and Homing. Springer-Verlag, Berlin, pp 347–353
Kirschvink JL (1989) Magnetite biomineralization and geomagnetic sensitivity in animals: an update and recommendations for future study. Bioelectromagnetics 10:239–259
Klimley AP (1993) Highly directional swimming by scalloped hammerhead sharks, Sphyrna lewini, and subsurface irradiance, temperature, bathymetry, and geomagnetic field. Mar Biol 117:1–22
Krylov VV, Osipova EA, Pavlova VV, Batrakova AA (2016) Influence of magnetic field on spatial orientation in zebrafish (Danio rerio) and roach (Rutilus rutilus) (Cyprinidae). J Ichthyol 56(3):456–461
Light P, Salmon M, Lohmann KJ (1993) Geomagnetic orientation of loggerhead sea turtles: evidence for an inclination compass. J Exp Biol 182:1–10
Lohmann KJ (1991) Magnetic orientation by hatchling loggerhead sea turtles (Caretta caretta). J Exp Biol 155:37–49
Marhold S, Burda H, Wiltschko W (1997) A magnetic polarity compass for direction finding in a subterranean mammal. Naturwissenschaften 84:421–423
Meyer CG, Holland KN, Papastamatiou YP (2005) Sharks can detect changes in the geomagnetic field. J R Soc Interface 2:129–130
Molet M, Miller RM (2014) Timing an attribute of associative learning. Behav Processes 0:4–14
Molteno TCA, Kennedy WL (2009) Navigation by induction-based magnetoreception in elasmobranch fishes. J Biophys 4575:380976
Myklatun A, Lauri A, Eder SHK, Cappetta M, Shcherbakov D, Wurst W, Winklhofer M, Westmeyer GG (2018) Zebrafish and medaka offer insights into the neurobehavioral correlates of vertebrate magnetoreception. Nature Comm 9:802. https://doi.org/10.1038/s41467-018-03090-6
Naisbett-Jones LC, Putman NF, Stephenson JF, Ladak S, Young KA (2017) A magnetic map leads juvenile European eels to the Gulf Stream. Curr Biol 27:1236–1240
Newton KC, Kajiura SM (2017) Magnetic field discrimination, learning and memory in the yellow stingray (Urobatis jamaicensis). Anim Cogn 20:603–614
Nosal AP, Chao Y, Farrara JD, Chai F, Hastings PA (2016) Olfaction contributes to pelagic navigation in a coastal shark. PLoS ONE 11(1):e0143758. https://doi.org/10.1371/journal.pone.0143758
Osipova EA, Pavlova VV, Nepomnyaschchikh VA, Kylov VV (2016) Influence of magnetic field on zebrafish activity and orientation in a plus maze. Behav Proc 122:80–86
Parker MO, Gavira J, Haigh A, Millington ME, Brown VJ, Combe FJ, Brennan CH (2012) Discrimination reversal and attentional sets in zebrafish (Danio rerio). Behav Brain Res 232:264–268
Paulin MG (1995) Electroreception and the compass sense of sharks. J Theor Biol 174:325–339
Pavlov IP (1927) Lectures on conditioned reflexes. International Publishers, New York, NY
Piercy AN, Snelson FF, Grubbs RD (2006) Urobatis jamaicensis. IUCN Red List of Threatened Species. Version 2016–1. https://www.iucnredlist.org/details/60109/0 Accessed 30 March 2019
Putman NF, Endres CS, Lohmann CMF, Lohmann KJ (2011) Longitude perception and bicoordinate magnetic maps in sea turtles. Curr Biol 21:463–466
Putman NF, Scanlon MM, Billman EJ, O’Neil JP, Couture RB, Quinn TP, Lohmann KJ, Noakes DLG (2014) An inherited magnetic map guides ocean navigation in juvenile pacific salmon. Curr Biol 24:446–450
Quinn TP (1980) Evidence of celestial and magnetic compass orientation in lake migrating sockeye salmon fry. J Comp Physiol 137:243–248. https://doi.org/10.1007/BF00657119
Quinn TP, Brannon EL (1982) The use of celestial and magnetic cues by orienting sockeye salmon smolts. J Comp Physiol 147:547–552. https://doi.org/10.1007/BF00612020
Quinn TP, Groot C (1983) Orientation of chum salmon (Oncorhynchus keta) after internal and external magnetic field alteration. Can J Fish Aquat Sci 40:1598–1606. https://doi.org/10.1139/f83-185
Quinn TP, Merrill RT, Brannon EL (1981) Magnetic field detection in Sockeye Salmon. J Exp Zool 217:137–142. https://doi.org/10.1002/jez.1402170114
Schluessel V (2015) Who would have thought that “Jaws” also has brains? Cognitive functions in elasmobranchs. Anim Cogn 18(1):19–37
Schluessel V, Bleckmann H (2005) Spatial memory and orientation strategies in the elasmobranch (Potamotrygon motoro). J Comp Physiol A 191:695–706
Schluessel V, Ober C (2018) How to get out of a maze? Stingrays (Potamotrygon motoro) use directional over landmark information when provided with both in a spatial task. Evol Ecol Res 19:619–637
Schluessel V, Herzog H, Scherpenstein M (2015) Seeing the forest before the trees-spatial orientation in freshwater stingrays (Potamotrygon motoro) in a hole-board task. Behav Proc 119:105–115
Shettleworth SJ (2010) Cognition, evolution and behavior, 2nd edn. Oxford University Press, New York
Shettleworth SJ, Sutton JE (2005) Multiple systems of spatial learning: dead reckoning and beacon-homing in rats. J Exp Psychol Anim Behav Process 31:125–141
Siciliano AM, Kajiura SM, Long JH, Porter MP (2013) Are you positive? Electric dipole polarity discrimination in the yellow stingray (Urobatis jamaicensis). Biol Bull 225:85–91
Souza JJ, Poluhowich JJ, Guerra RJ (1988) Orientation responses of American eels, (Anguilla rostrata) to varying magnetic fields. Comp Biochem Physiol A 90:57–61. https://doi.org/10.1016/0300-9629(88)91005-5
Speed CW, Field IC, Meekan MG, Bradshaw CJA (2010) Complexities of coastal shark movements and their implications for management. Mar Ecol Prog Ser 408:275–293
Strang CG, Sherry DF (2014) Serial reversal learning in bumblebees (Bombus impatiens). Anim Cogn 17:723–734
Takebe A, Furutani T, Wada T, Koinuma M, Kubo Y, Okano K, Okano T (2012) Zebrafish respond to the geomagnetic field by bimodal and group-dependent orientation. Sci Rep 2:727. https://doi.org/10.1038/srep00727
Tesch FW, Wendt T, Karlsson L (1992) Influence of geomagnetism and salinity on orientation of the eel (Anguilla anguilla), as evident from laboratory experiments. Ecol Freshwater Fish 1:52–60. https://doi.org/10.1111/j.1600-0633.1992
Vaudo JJ, Lowe CG (2006) Movement patterns of the round stingray (Urobatis halleri) (Cooper) near a thermal outfall. J Fish Biol 68:1756–1766
Walker MM, Diebel CE, Kirschvink JL (2003) Detection and use of the Earth’s magnetic field by aquatic vertebrates. In: Collin SP, Marshall NJ (eds) Sensory processing in aquatic environments. Springer-Verlag, New York, pp 53–74
Warburton K, Hughes R (2011) Learning of foraging skills by fish. In: Brown C, Laland K, Krause J (eds) Fish cognition and behavior, 2nd edn. Wiley-Blackwell, Chichester, UK, pp 10–35
Wiltschko W, Wiltschko R (1972) Magnetic compass of European robins. Science 176:62–64
Acknowledgements
This research was supported by grants to KCN from the Florida Atlantic University Graduate Research and Inquiry Program Award, the Save Our Seas Foundation Small Grant, the Student Research Award from the American Elasmobranch Society, The Grant in Aid of Research from the Society for Integrative and Comparative Biology, the Gordon Gilbert Graduate Scholarship from the Friends of Gumbo Limbo Nature Center, and the PADI Foundation Grant. We thank S. Creager, A. Murakami, E. Cave, L. Celano, J. Noble, G. Gil, B. Bowers, K. Kramer, and S. Ramirez for help with stingray collection and husbandry, and R. Stackman and M. Salmon for assistance with experimental design and animal training protocols. Animals were collected pursuant to Florida Fish and Wildlife Conservation Commission Special Activities License SAL 15-1413A-SR
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of animal welfare and ethics
All applicable international, national, and institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the Florida Atlantic University Institutional Animal Care and Use Committee.
Additional information
Responsible Editor: J. Carlson.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by undisclosed experts.
Rights and permissions
About this article
Cite this article
Newton, K.C., Kajiura, S.M. The yellow stingray (Urobatis jamaicensis) can use magnetic field polarity to orient in space and solve a maze. Mar Biol 167, 36 (2020). https://doi.org/10.1007/s00227-019-3643-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-019-3643-9