Abstract
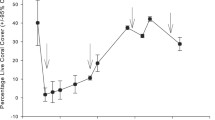
The 1997/1998 El Niño Southern Oscillation (ENSO) was the most severe coral bleaching event in recent history, resulting in the loss of 16 % of the world’s coral reefs. Mortality was particularly severe in French Polynesia, where unprecedented mortality of massive Porites was observed in lagoonal sites of Rangiroa Atoll. To assess the recovery of massive Porites 15 years later, we resurveyed the size structure and extent of partial mortality of massive Porites at Tivaru (Rangiroa). Surveys revealed an abundance of massive Porites colonies rising from the shallow lagoonal floor. Colony width averaged 2.65 m, reaching a maximum of 7.1 m (estimated age of ~391 ± 107 years old). The relative cover of recently dead skeleton within quadrats declined from 42.8 % in 1998 to zero in 2013, yet the relative cover of old dead skeleton increased only marginally from 22.1 % in 1998 to 26.1 % in 2013. At a colony level, the proportion of Porites dominated by living tissue increased from 34.9 % in 1998 to 73.9 % in 2013, indicating rapid recovery of recent dead skeleton to living tissue rather than transitioning to old dead skeleton. Such rapid post-bleaching recovery is unprecedented in massive Porites and resulted from remarkable self-regeneration termed the ‘Phoenix effect’, whereby remnant cryptic patches of tissue that survived the 1997/1998 ENSO event regenerated and rapidly overgrew adjacent dead skeleton. Contrary to our earlier predictions, not only are large massive Porites relatively resistant to stress, they appear to have a remarkable capacity for recovery even after severe partial mortality.





Similar content being viewed by others
References
Adjeroud M, Augustin D, Galzin R, Salvat B (2002) Natural disturbances and interannual variability of coral reef communities on the outer slope of Tiahura (Moorea, French Polynesia): 1991 to 1997. Mar Ecol-Prog Ser 237:121–131
Adjeroud M, Pratchett MS, Kospartov MC, Lejeusne C, Penin L (2007) Small-scale variability in the size structure of scleractinian corals around Moorea, French Polynesia: patterns across depths and locations. Hydrobiologia 589:117–126. doi:10.1007/s10750-007-0726-2
Anthony KRN, Ridd PV, Orpin AR, Larcombe P, Lough J (2004) Temporal variation of light availability in coastal benthic habitats: effects of clouds, turbidity, and tides. Limnol Oceanogr 49:2201–2211
Baird AH, Marshall PA (2002) Mortality, growth and reproduction in scleractinian corals following bleaching on the Great Barrier Reef. Mar Ecol-Prog Ser 237:133–141
Bak RPM, Stewardvanes Y (1980) Regeneration of superficial damage in the scleractinian corals agaricia-agaricites-F-purpurea and porites-astreoides. Bull Mar Sci 30:883–887
Bessat F, Buigues D (2001) Two centuries of variation in coral growth in a massive Porites colony from Moorea (French Polynesia): a response of ocean-atmosphere variability from south central Pacific. Palaeogeogr Palaeoclimatol Palaeoecol 175:381–392
Brown BE, Letissier MDA, Scoffin TP, Tudhope AW (1990) Evaluation of the environmental impact of dredging on intertidal coral reefs at Ko Phuket, Thailand, using ecological and physiological parameters. Mar Ecol-Prog Ser 65:273–281
Cheal AJ, MacNeil MA, Cripps E, Emslie MJ, Jonker M, Schaffelke B, Sweatman H (2010) Coral–macroalgal phase shifts or reef resilience: links with diversity and functional roles of herbivorous fishes on the Great Barrier Reef. Coral Reefs 29:1005–1015. doi:10.1007/s00338-010-0661-y
Darke WM, Barnes DJ (1993) Growth trajectories of corallites and ages of polyps in massive colonies of reef-building corals of the genus Porites. Mar Biol 117:321–326
Davies PS (1991) Effect of daylight variations on the energy budgets of shallow-water corals. Mar Biol 108:137–144. doi:10.1007/Bf01313481
Denis V, Debreuil J, De Palmas S, Richard J, Guillaume MMM, Bruggemann JH (2011) Lesion regeneration capacities in populations of the massive coral Porites lutea at Reunion Island: environmental correlates. Mar Ecol Prog Ser 428:105–117. doi:10.3354/Meps09060
Diaz-Pulido G, McCook L, Dove S, Berkelmans R, Roff G, Kline D, Weeks SJ, Evans RD, Williamson DH, Hoegh-Guldberg O (2009) Doom and boom on a resilient reef: climate change, algal overgrowth and coral recovery. PLoS ONE 4:e5239
Done TJ (1988) Simulation of recovery of pre-disturbance size structure in populations of Porites spp damaged by the crown of thorns starfish acanthaster-planci. Mar Biol 100:51–61. doi:10.1007/Bf00392954
Done TJ, Potts DC (1992) Influences of habitat and natural disturbances on contributions of massive Porites corals to reef communities. Mar Biol 114:479–493
Donner SD, Skirving WJ, Little CM, Oppenheimer M, Hoegh-Guldberg O (2005) Global assessment of coral bleaching and required rates of adaptation under climate change. Glob Change Biol 11:2251–2265
Edwards AJ, Clark S, Zahir H, Rajasuriya A, Naseer A, Rubens J (2001) Coral bleaching and mortality on artificial and natural reefs in Maldives in 1998, sea surface temperature anomalies and initial recovery. Mar Pollut Bull 42:7–15
Gilmour JP, Smith LD, Heyward AJ, Baird AH, Pratchett MS (2013) Recovery of an isolated coral reef system following severe disturbance. Science 340:69–71. doi:10.1126/Science.1232310
Glynn PW (2011) In tandem reef coral and cryptic metazoan declines and extinctions. Bull Mar Sci 87:767–794. doi:10.5343/Bms.2010.1025
Harmelin-Vivien ML, Laboute P (1986) Catastrophic impact of hurricanes on atoll outer reef slopes in the Tuamotu (French-Polynesia). Coral Reefs 5:55–62. doi:10.1007/Bf00270353
Hoegh-Guldberg O (1999) Climate change, coral bleaching and the future of the world’s coral reefs. Mar Freshw Res 50:839–866
Hughes TP, Jackson JBC (1985) Population dynamics and life histories of foliaceous corals. Ecol Monogr 55:141–166
IPCC (2007) Climate change 2007: the physical science basis. Cambridge University Press, Cambridge
Jompa J, McCook LJ (2003) Contrasting effects of turf algae on corals: massive Porites spp. are unaffected by mixed-species turfs, but killed by the red alga Anotrichium tenue. Mar Ecol Prog Ser 258:79–86
Jordan-Dahlgren E (1992) Recolonization patterns of acropora-palmata in a marginal environment. Bull Mar Sci 51:104–117
Kayal M, Vercelloni J, de Loma TL, Bosserelle P, Chancerelle Y, Geoffroy S, Stievenart C, Michonneau F, Penin L, Planes S, Adjeroud M (2012) Predator crown-of-thorns starfish (Acanthaster planci) outbreak, mass mortality of corals, and cascading effects on reef fish and benthic communities. PLoS ONE. doi:10.1371/journal.pone.0047363
Kojis BL, Quinn NJ (1982) Reproductive strategies in four species of Porites (Scleractinia). In: Gomez ED, Birkeland CE, Buddemeier RW, Johannes RE, Marsh JA Jr, Tsuda RT (eds) Proceedings of the 4th international coral reef symposium. Marine Science Center, University of the Philippines, Manila, Philippines, pp 145–151
Krupp DA, Jokiel PL, Chartrand TS (1992) Asexual reproduction by the solitary Scleractinian coral Fungia scutaria on dead parent corall in Kaneohe Bay, Oahu, Hawaiian Islands. In: Richmond BM (ed) Proceedings of the seventh international coral reef symposium, Hawaii, pp 527–534
Lang JC, Chornesky EA (1990) Competition between scleractinian reef corals—a review of mechanisms and effects. In: Dubinsky Z (ed) Ecosystems of the world: coral reefs. Elsevier, Amsterdam, pp 209–252
Lima FP, Wethey DS (2012) Three decades of high-resolution coastal sea surface temperatures reveal more than warming. Nat Commun. doi:10.1038/Ncomms1713
Lirman D, Manzello D, Macia S (2002) Back from the dead: the resilience of Siderastrea radians to severe stress. Coral Reefs 21:291–292
Loch K, Loch W, Schuhmacher H, See WR (2002) Coral recruitment and regeneration on a Maldivian reef 21 months after the coral bleaching event of 1998. Mar Ecol-Pubbl Della Stn Zool Di Nap I 23:219–236
Lough JM (2012) Small change, big difference: sea surface temperature distributions for tropical coral reef ecosystems, 1950–2011. J Geophys Res-Ocean. doi:10.1029/2012jc008199
Loya Y, Sakai K, Yamazato K, Nakano Y, Sambali H, van Woesik R (2001) Coral bleaching: the winners and the losers. Ecol Lett 4:122–131
Marshall PA, Baird AH (2000) Bleaching of corals on the Great Barrier Reef: differential susceptibilities among taxa. Coral Reefs 19:155–163
McWilliams JP, Cote IM, Gill JA, Sutherland WJ, Watkinson AR (2005) Accelerating impacts of temperature-induced coral bleaching in the Caribbean. Ecology 86:2055–2060
Meesters EH, Wesseling I, Bak RPM (1996) Partial mortality in three species of reef-building corals and the relation with colony morphology. Bull Mar Sci 58:838–852
Mumby PJ, Chisholm JRM, Edwards AJ, Clark CD, Roark EB, Andrefouet S, Jaubert J (2001) Unprecedented bleaching-induced mortality in Porites spp. at Rangiroa Atoll, French Polynesia. Mar Biol 139:183–189
Mumby PJ, Hedley JD, Chisholm JRM, Clark CD, Ripley H, Jaubert J (2004) The cover of living and dead corals from airborne remote sensing. Coral Reefs 23:171–183. doi:10.1007/s00338-004-0382-1
Peyrot-Clausade M, LeCampionAlsumard T, Hutchings P, LeCampion J, Payri C, Fontaine MC (1995) Initial bioerosion and bioaccretion on experimental substrates in high island and atoll lagoons (French Polynesia). Oceanol Acta 18:531–541
Potts DC, Done TJ, Isdale PJ, Fisk DA (1985) Dominance of a coral community by the genus Porites (Scleractinia). Mar Ecol Prog Ser 23:79–84
R (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Riegl B, Piller WE (2001) “Cryptic” tissues inside Acropora frame works (Indonesia): a mechanism to enhance tissue survival in hard times while also increasing framework density. Coral Reefs 20:67–68
Salvat B, Aubanel A, Adjeroud M, Bouisset P, Calmet D, Chancerelle Y, Cochennec N, Davies N, Fougerousse A, Galzin R, Lagouy E, Lo C, Monier C, Ponsonnet C, Remoissenet G, Schneider D, Stein A, Tatarata M, Villiers L (2008) Monitoring of French Polynesia coral reefs and their recent development. Rev D Ecol-La Terre Et La Vie 63:145–177
Selig ER, Casey KS, Bruno JF (2010) New insights into global patterns of ocean temperature anomalies: implications for coral reef health and management. Glob Ecol Biogeogr 19:397–411. doi:10.1111/J.1466-8238.2009.00522.X
Tanzil JTI, Brown BE, Tudhope AW, Dunne RP (2009) Decline in skeletal growth of the coral Porites lutea from the Andaman Sea, South Thailand between 1984 and 2005. Coral Reefs 28:519–528. doi:10.1007/S00338-008-0457-5
Thompson A, Dolman AM (2010) Coral bleaching: one disturbance too many for near-shore reefs of the Great Barrier Reef. Coral Reefs 29:637–648. doi:10.1007/s00338-009-0562-0
Titlyanov EA, Titlyanova TV (2009) The dynamics of the restoration of mechanical damage to colonies of the scleractinian coral Porites lutea under conditions of competition with algal settlers for substratum. Russ J Mar Biol 35:230–235. doi:10.1134/S1063074009030067
van Oppen MJH, Souter P, Howells EJ, Heyward A, Berkelmans R (2011) Novel genetic diversity through somatic mutations: fuel for adaptation of reef corals? Diversity 3:405–423. doi:10.3390/d3030405
van Woesik R (1998) Lesion healing on massive Porites spp. corals. Mar Ecol Prog Ser 164:213–220. doi:10.3354/Meps164213
van Woesik R, Sakai K, Ganese A, Loya Y (2011) Revisiting the winners and the losers a decade after coral bleaching. Mar Ecol Prog Ser 434:67–76
Wooldridge S (2009) Water quality and coral bleaching thresholds: formalising the linkage for the inshore reefs of the Great Barrier Reef, Australia. Mar Pollut Bull 58:745–751
Acknowledgments
We thank the Khaled bin Sultan Living Oceans Foundation and crew of the Golden Shadow for the opportunity to revisit Tivaru on the French Polynesia leg of the Global Reef Expedition.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by T. L. Goulet.
Rights and permissions
About this article
Cite this article
Roff, G., Bejarano, S., Bozec, YM. et al. Porites and the Phoenix effect: unprecedented recovery after a mass coral bleaching event at Rangiroa Atoll, French Polynesia. Mar Biol 161, 1385–1393 (2014). https://doi.org/10.1007/s00227-014-2426-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-014-2426-6