Abstract
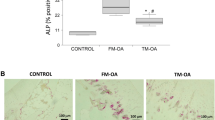
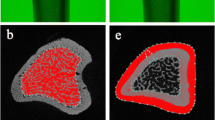
Evidence suggests that chiropractic manipulation might exert positive effects in osteoporotic patients. The aim of this study was to evaluate the effects of chiropractic manipulation on bone structure and skeletal muscle in rats with bone loss caused by ovariectomy (OVX). The 6-month old Sprague-Dawley rats at 10 weeks following OVX or sham operation (Sh) did not suffer chiropractic manipulation (NM group) or were submitted to true chiropractic manipulation using the chiropractic adjusting instrument Activator V® three times/week for 6 weeks as follows: Force 1 setting was applied onto the tibial tubercle of the rat right hind limb (TM group), whereas the corresponding left hind limb received a false manipulation (FM group) consisting of ActivatorV® firing in the air and slightly touching the tibial tubercle. Bone mineral density (BMD) and bone mineral content (BMC) were determined in long bones and L3–L4 vertebrae in all rats. Femora and tibia were analyzed by μCT. Mechano growth factor (MGF) was detected in long bones and soleus, quadriceps and tibial muscles by immunohistochemistry and Western blot. The decrease of BMD and BMC as well as trabecular bone impairment in the long bones of OVX rats vs Sh controls was partially reversed in the TM group versus FM or NM rats. This bone improvement by chiropractic manipulation was associated with an increased MGF expression in the quadriceps and the anterior tibial muscle in OVX rats. These findings support the notion that chiropractic manipulation can ameliorate osteoporotic bone at least partly by targeting skeletal muscle.



Similar content being viewed by others
References
González JM (2000) Osteoporosis. In: Rozman C, Farreras PV (eds) Principios de medicina interna, vol 1, 14th edn. Editorial Harcourt, Madrid, pp 1233–1242
Krane SM, Michael FH (1998) Metabolic bone diseases. In: Harrison J (ed) Principles of internal medicine, vol 2, 14th edn. Mc Graw-Hill-Interamericana, New York, pp 2557–2563
Kemmler W, von Stengel S, Bebenek M, Engelke K, Hentschke C, Kalender WA (2012) Exercise and fractures in postmenopausal women: 12-year results of the Erlangen fitness and osteoporosis prevention study (EFOPS). Osteoporos Int. 23:1267–1276
Turner CH (1998) Three rules for bone adaptation to mechanical stimuli. Bone 23:399–407
Burger EH, Klein-Nulend J (1999) Mechano transduction in bone-role of the lacuno-canalicular network. FASEB J 13:S101–S112
Pedersen BK (2011) Muscles and their myokines. J ExpBiol 214:337–346
Jähn K, Lara-Castillo N, Brotto L, Mo CL, Johnson ML, Brotto M, Bonewald LF (2012) Skeletal muscle secreted factors prevent glucocorticoid-induced osteocyte apoptosis through activation of B-catenin. Eur Cell Mater 24:197–210
Matheny RW Jr, Nindl BC, Adamo ML (2010) Minireview: mechano-growth factor: a putative product of IGF-I gene expression involved in tissue repair and regeneration. Endocrinology 151:865–875
Goldspink G (2005) Mechanical signals, IGF-I gene splicing, and muscle adaptation. Physiology (Bethesda) 20:232–238
Kravchenko IV, Furalyov VA, Lisitsina ES, Popov VO (2011) Stimulation of mechano-growth factor expression by second messengers. Arch Biochem Biophys 507:323–331
Tang LL, Xian CY, Wang YL (2006) The MGF expression of osteoblasts in response to mechanical overload. Arch Oral Biol 51:1080–1085
Juffer P, Jaspers RT, Lips P, Bakker AD, Klein-Nulend J (2012) Expression of muscle anabolic and metabolic factors in mechanically loaded MLO-Y4 osteocytes. Am J Physiol Endocrinol Metab 302:E389–E395
Deng M, Zhang B, Wang K, Liu F, Xiao H, Zhao J, Liu P, Li Y, Lin F, Wang Y (2010) Mechano growth factor E peptide promotes osteoblast proliferation and bone-defect healing in rabbits. Int Orthop 35:1099–1106
Xin C, Bingbing Z, Yuanliang W, Chengyu X, Li Y, Moyuan D, Qin P, Yuxiao L (2012) Mechano-growth factor E peptide inhibits the differentation and mineralization of osteoblasts. ArchOralBiol 57:720–727
Kaji H (2014) Interaction between muscle and bone. J Bone Metab 21:29–40
Chapman-Smith D (2004) Quiropráctica. Talleres Gráficos Edelvives, Zaragoza
Fuhr AW (2009) The activator methods, 2nd edn. Mosby, Maryland Heights
Pickar J (2002) Neurophysiological effects of spinal manipulation. Spine J 2:357–371
Johnson IP (2008) Hypothesis: upregulation of a muscle-specific isoform of insulin-like growth factor-1 (IGF-1) by spinal manipulation. Med Hypotheses 71:715–721
Bonewald LF, Kiel DP, Clemens TL, Esser K, Orwoll ES, O’Keefe RJ, Fielding RA (2013) Forum on bone and skeletal muscle interactions: summary of the proceedings of an ASBMR workshop. J Bone Miner Res 28:1857–1865
Russell WMS, Burch RL (1959) The principles of humane experimental technique. Methuen and Co. Ltd, London
Trierweiler J, Gottert DN, Gehlen G (2012) Evaluation of mechanical allodynia in an animal immobilization model using the von Frey method. J Manip Physiol Ther 35:18–25
Portal-Núñez S, Lozano D, de Castro LF, de Gortázar AR, Nogués X, Esbrit P (2010) Alterations of the Wnt/beta-catenin pathway and its target genes for the N- and C-terminal domains of parathyroid hormone-related protein in bone from diabetic mice. FEBS Lett 584:3095–3100
Lelovas PP, Xanthos TT, Thoma SE, Lyritis GP, Dontas IA (2008) The laboratory rat as an animal model for osteoporosis research. Comp Med 58:424–430
Kennedy Oran D, Brennan Orlaith, Rackard Susan M, O’Brien Fergal J, Taylor David, Clive Lee T (2009) Variation of trabecular microarchitectural parameters in cranial, caudal and mid-vertebral regions of the ovine L3 vertebra. J Anat 214:729–735
Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Müller R (2010) Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res 25:1468–1486
Woodhouse D (2003) Post-traumatic compression fracture. Clin Chiropr 6:67–72
Stock JL, Amantea JH, Overdorf AD, Samar AD, Pickens FL (1997) The role of chiropractic in the diagnosis, prevention, and treatment of osteoporosis. Complement Ther Med 5:36–39
Hawk C, Schneider M, Dougherty P, Gleberzon BJ, Killinger LZ (2010) Best practices recommendations for chiropractic care for older adults: results of a consensus process. J Manip Physiol Ther 33:464–473
Liebschner MA, Chun K, Kim N, Ehni B (2014) In vitro biochemical evaluation of single impulse and repetitive mechanical shock wave devices utilized for spinal manipulative therapy. Ann Biomed Eng 42:2524–2536
Colloca CJ, Keller TS, Black P, Normand MC, Harrison DE, Harrison DD (2005) Comparison of mechanical force of manually assisted chiropractic adjusting instruments. J Manip Physiol Ther 28:414–422
Smith DB, Fuhr AW, Davis BP (1989) Skin accelerometer displacement and relative bone movement of adjacent vertebrae in response to chiropractic percussion thrusts. J Manip Physiol Ther 12:26–37
Fuhr AW, Smith DB (1986) Accuracy of piezoelectric accelerometers measuring displacement of a spinal adjusting instrument. J Manip Physiol Ther 9:15–21
Brown JW, Le NMP (2011) The effect of thermopreference on circadian thermoregulation in sprague-dawley and fisher 344 rats. J Therm Biol 37:309–315
Iwaniec UT, Philbrick KA, Wong CP, Gordon JL, Kahler-Quesada AM, Olson DA, Branscum AJ, Sargent JL, DeMambro VE, Rosen CJ, Turner RT (2016) Room temperature housing results in premature cancellous bone loss in growing female mice: implications for the mouse as a preclinical model for age-related bone loss. Osteoporos Int 27:3091–3101
Wronski TJ, Dann LM, Horner SL (1990) Time course of vertebral osteopenia inovariectomized rats. Bone 10:295–301
Turner RT, Vandersteenhoven JJ, Bell NH (1989) The effects of ovariectomy and 17 beta-estradiol on cortical bone histomorphometry in growing rats. J Bone Miner Res 2:115–122
Kalu DN (1991) The ovarectomized rat model of postmenopausal bone loss. Bone Miner 15:175–191
Ahtiainen M, Pöllänen E, Ronkainen PH, Alen M, Puolakka J, Kaprio J, Sipilä S, Kovanen V (2012) Age and estrogen-based hormone therapy affect systemic and local IL-6 and IGF-1 pathways in women. Age (Dordr) 34:1249–1260
Adams GR (1998) Role of insulin-like growth factor-I in the regulation of skeletal muscle adaptation to increased loading. Exerc Sport Sci Rev 26:31–60
Wang LC, Kernell D (2001) Quantification of fibre type regionalisation: an analysis of lower hindlimb muscles in the rat. J Anat 198:295–308
Punkt K, Naupert A, Asmussen G (2004) Differentiation of rat skeletal muscle fibres during development and ageing. Acta Histochem 106:145–154
Jaspers RT, Bravenboer N (2014) Biochemical interaction between muscle and bone: a physiological reality? Clinic Rev Bone Miner Metab 12:27–43
Wang L, Ma R, Guo Y, Sun J, Liu H, Zhu R, Liu C, Li J, Li L, Chen B, Sun L, Tang J, Zhao D, Mo F, Niu J, Jiang G, Fu M, Brömme D, Zhang D, Gao S (2017) Antioxidant effect of fructus ligustri lucidi aqueous extract in ovariectomized rats is mediated through Nox4-ROS-NF-κB pathway. Front Pharmacol 8:266–279
Hamrick MW, McNeil PL, Patterson SL (2010) Role of muscle-derived growth factors in bone formation. J Musculoskelet Neuronal Interact 10:64–70
Acknowledgements
This work was supported in part by grants from the Spanish Chiropractic Association (AEQ) and Fondation de Recherche Chiropratique du Québec (Québec, Canada).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. López-Herradón, R. Fujikawa, M. Gómez-Marín, J. P. Stedile-Lovatel, F. Mulero, J. A. Ardura, P. Ruiz, I. Muñoz, P. Esbrit, I. Mahíllo-Fernández and A.Ortega-de Mues declare that they do not have conflict of interest.
Ethical Approval
All animal procedures were carried out according to the guidelines of European Union directives (2010/63/EU). All experimental protocols were approved by the Institutional Animal Care and Use Committee at Fundación Jiménez Díaz.
Rights and permissions
About this article
Cite this article
López-Herradón, A., Fujikawa, R., Gómez-Marín, M. et al. Impact of Chiropractic Manipulation on Bone and Skeletal Muscle of Ovariectomized Rats. Calcif Tissue Int 101, 519–529 (2017). https://doi.org/10.1007/s00223-017-0304-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-017-0304-1