Abstract
Dairy products provide a package of essential nutrients that is difficult to obtain in low-dairy or dairy-free diets, and for many people it is not possible to achieve recommended daily calcium intakes with a dairy-free diet. Despite the established benefits for bone health, some people avoid dairy in their diet due to beliefs that dairy may be detrimental to health, especially in those with weight management issues, lactose intolerance, osteoarthritis, rheumatoid arthritis, or trying to avoid cardiovascular disease. This review provides information for health professionals to enable them to help their patients make informed decisions about consuming dairy products as part of a balanced diet. There may be a weak association between dairy consumption and a possible small weight reduction, with decreases in fat mass and waist circumference and increases in lean body mass. Lactose intolerant individuals may not need to completely eliminate dairy products from their diet, as both yogurt and hard cheese are well tolerated. Among people with arthritis, there is no evidence for a benefit to avoid dairy consumption. Dairy products do not increase the risk of cardiovascular disease, particularly if low fat. Intake of up to three servings of dairy products per day appears to be safe and may confer a favourable benefit with regard to bone health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dairy products have been an important part of the human diet for some 8000 years and are part of the official nutritional recommendations in many countries worldwide. They provide a package of key nutrients that are difficult to obtain in diets with limited or no dairy products, such as vegan or dairy restrictive diets. Indeed, dairy products are rich in calcium, protein, potassium and phosphorus. They contribute around 52–65 % of the dietary reference intake (DRI) of calcium and 20–28 % of the protein requirement, depending on the age of the consumer [1–5]. The contribution of dairy products to providing recommended calcium intakes has largely driven the dietary recommendations for dairy intake in most guidelines. Up to two-thirds of the population’s calcium intake in Western countries is supplied by dairy products [6, 7], while at the same time dairy foods represent only 9–12 % of the total energy consumption [8].
Much research is still ongoing into the health effects of dairy product consumption. This article examines some clinical evidence and provides information for health professionals to enable them to help their patients make informed decisions about consuming dairy products as part of a balanced diet. In particular, the effect of dairy products on bone health, obesity, arthritis and cardiovascular disease are briefly discussed.
Methods
This commentary is based on a narrative literature review. It focusses on the most robust available evidence where possible, for example meta-analyses and prospective studies, with the most recent publications consulted. Relevant articles were identified through a systematic search, from 1966 to 2013 in MEDLINE with the keywords «dairy products», «bone» and «muscle». Only articles published in English were considered. Following this extensive search of the literature, a critical appraisal was obtained through three face-to-face consensus experts meetings, held during the second part of the year 2013 and the first part of the year 2014. For the purposes of this article, dairy products refer to animal milks and derived products, excluding butter and vegetable-derived products like soy or almond ‘milks’.
Dairy Products as a Source of Key Nutrients
Worldwide, many people fail to achieve an adequate dietary calcium intake. The adequacy of dietary calcium consumption varies geographically and reflects milk consumption. Intakes of calcium are generally low across Europe [9]; judged against the World Health Organization/Food and Agriculture Organization of the United Nations (WHO/FAO) adult recommend nutrient intake (RNI) of 1000 mg/d, mean calcium intakes of 16 European countries were 687–1171 mg/day in men and 508–1047 mg/d in women [9]. Other nations fall far short of RNIs; for example, in Brazil 99 % of adults (19–60 years) consume inadequate levels of calcium [10].
To meet the daily dietary calcium requirement, dairy products, green vegetables and mineral waters are important, easily available, sources of calcium. Indeed, dairy products represent good dietary sources of calcium due to their high calcium and nutrient contents, high absorptive rate, availability and relatively low cost, which makes the regular consumption of dairy products feasible. They provide more calcium, protein, magnesium, potassium, zinc and phosphorus per calorie than any other typical food found in the adult diet [11, 12]. Many dietary recommendations include the consumption of 3 servings of dairy products per day (for example, 1 glass of milk, 1 portion of cheese, 1 yogurt)—an amount that provides most of the DRI of calcium for the general population [13]. For example, 250 mg of calcium may be obtained from a 200 ml glass of milk, a 125 g serving of yogurt or 35 g of hard cheese (Table 1) [14].
There are non-dairy sources of calcium, such as mineral water, kale and dark greens, dried beans and legumes, but it is difficult to meet daily requirements with these. A variety of calcium-fortified foods, such as orange juice and soy juice, are now available and provide the same amount of calcium as a single serving of dairy; the amounts have been evaluated at 1.1 servings of fortified soy beverage, 0.6 servings of fortified orange juice, 1.2 servings of bony fish or 2.2 servings of leafy greens [15]. However, these foods do not provide the equivalent profile of other nutrients and the amounts needed are unrealistic in some cases [16]. Using data from the National Health and Nutrition Examination Survey (NHANES), it has been determined that it is impossible to meet calcium recommendations while meeting other nutrient recommendations with a dairy-free diet within the current US dietary pattern [17]. The nutrients most at risk if dairy products are excluded are calcium, potassium and magnesium. For women of 19–50 years of age who do not consume dairy products, only 44 % of calcium and 57 % of magnesium and potassium recommendations are met [18].
Under normal dietary conditions, about 30–40 % of the calcium contained in milk and cheese is absorbed in the gut either through vitamin D-dependent transport across the duodenum, facilitated diffusion or under the influence of lactose in the distal small intestine via the paracellular route [12]. In contrast, only 28–36 % of the calcium is absorbed from fortified cereals, soy (if not dephytized) and rice juice [19]. Calcium is found in green leafy vegetables in reasonable quantity (Table 2 ) [20]; however, a high proportion of the calcium is made insoluble by the presence of fibres, phytic acid and oxalic acid, which reduce the bioavailability of calcium. For example, cooked spinach contains 115 mg of calcium per serving but only 5 % of spinach calcium is absorbed, as spinach contains a high proportion of oxalates and phytates, which bind calcium and form insoluble salt compounds [20]. Thus, while consumption of 1 cup of milk per day may be considered feasible to provide 100 mg of absorbed calcium, the consumption of 16 servings of spinach per day to provide the same amount of bioavailable calcium may be considered unpalatable (Table 2).
Dairy foods also contain protein, with a 200 ml glass of milk also providing around 6–7 g of mostly casein and whey proteins [21]. The casein phosphopeptides (CPP) and lactose in dairy foods can facilitate intestinal calcium absorption [6, 22]. For example, the enzymatic hydrolysis of casein protein leads to the formation of CPP. These molecular species have been shown to bind calcium and therefore protect it against precipitation with anions such as phosphates in the small intestine. The net result is an increase of passive calcium absorption in the ileum [23].
Phosphorus (P) ions are also present in milk, generally combined with proteins and peptides. At the kidney level, increased inorganic phosphate intake leads to decreased urinary calcium and increased calcium retention. During growth and adulthood, the administration of calcium and inorganic phosphate in a ratio close to that found in dairy products leads to positive effects on bone health [24]. Calcium and inorganic phosphate interact at both the intestinal and renal levels and impact on bone maintenance and osteoporosis management. Interactions between calcium, inorganic phosphate, protein and vitamin D reduce bone resorption, attenuating age-related bone loss [24].
Dairy Foods or Calcium Supplements?
Increased awareness about the impact of nutrition on health and the availability of dietary supplements in shops has made them popular choices, with 43 % of Americans using calcium supplements [25]. The absorption of calcium from commonly used supplements, such as calcium carbonate and calcium citrate, is around 30–40 % and is optimized by concurrent food intake and by taking divided doses with protein-containing meals throughout the day [6]. However, calcium supplements can interact with several medications: the absorption of calcium from calcium carbonate is reduced by proton pump inhibitors, while calcium supplements may interact with antibiotics, thiazide diuretics, digoxin and phenytoin. Furthermore, calcium supplements do not contain the additional nutrients—including protein, potassium and magnesium—provided by dairy foods.
Although studies have generally failed to demonstrate that dairy calcium is better absorbed than calcium from mineral salts, the availability for bone mineralisation appears to be greater for dairy foods and the effects may be longer lasting [11]. Consequently, head-to-head intervention trials show bone-health benefits for dairy foods over calcium supplements. In postmenopausal women, those randomised to dairy (low-fat milk and yogurt products, fortified with concentrated milk protein) had greater improvements in arm, pelvis, total spine and total-body bone mineral density (BMD) than those receiving calcium supplements in one trial [26, 27], though not at other sites in another trial [28]. In another randomised trial, cheese was more beneficial than calcium supplements for tibia cortical bone mass accrual in children [22]. The authors suggested that this may be a result of better absorption of dairy calcium, differences in the distribution of intake over the day (small regular amounts of dairy versus two large doses from the supplement) and differences in protein or micronutrient intakes [22]. Greater increases in insulin-like growth factor-I (IGF-I), which favours bone formation, have also been reported for dairy compared with calcium supplements [26].
Dairy sources of calcium are considered to be at least as effective for bone health as calcium supplements, and probably more so [11]. A consensus statement from the Belgian Bone Club suggests that for the non-pharmacological management of osteoporosis, single-nutrient supplements will frequently be inadequate and preference should go to the use of complete supplements or complete foods such as dairy products [29]. Calcium supplementation should only be targeted to those who do not get sufficient calcium from their diet and who are at high risk for osteoporosis and/or fracture [30].
Bone Health: Effect of Dairy Products Consumption
Bone mineral mass is determined by the amount of bone accumulated at the end of skeletal growth (peak bone mass) and by the amount of bone lost subsequently. Building a strong skeleton from foetal life to adulthood and maintaining healthy bones through the menopause and ageing are vital to minimize frailty in the elderly. After the age of 20 years, there is little change in bone mineral mass until the menopause in women, when a rapid drop in oestrogen leads to an increase in bone remodelling. Menopause is associated with an average annual bone loss of 3–5 % during the first few years and around 1 % thereafter [31]. In men, bone loss is slower and more linear, but men also develop osteoporosis with ageing. The risk of osteoporosis significantly increases after 50 years of age.
There is growing evidence that the consequences of age-related or postmenopausal bone loss on fracture risk depend on the level of peak bone mass achieved during childhood and adolescence, as well as on the rate of bone loss [32]. Maximizing peak bone mass may be an important contributor to fracture risk reduction in children as well as in the elderly. While 60–80 % of the variance in peak bone mass is explained by genetic factors, the remainder may be amenable to interventions aimed at maximizing it [32, 33]. Such interventions include increasing physical activity and decreasing exposure to risk factors such as cigarette smoking and excessive alcohol intake, as well as optimizing nutrition [21, 32]. Adequate dietary calcium and protein intakes are essential to achieve optimal peak bone mass during skeletal growth and to prevent bone loss in the elderly [34].
Dairy Consumption for Bone Development in Children
The importance of dietary calcium for bone growth is evident even prior to birth. Diets rich in calcium and other micronutrients (as supplied by dairy foods, green leafy vegetables, fruits) given to pregnant women are associated with increased skeletal growth and/or bone mass/density in the offspring, with beneficial effects on bone size and BMD apparent up to the ages of 6–9 years [35, 36]. This is compatible with the hypothesis that dairy consumption by pregnant women might promote bone health in the child.
During childhood, the available data indicate that dairy products are important for growth and bone health [37]. In growing children, long-term milk avoidance is associated with smaller stature, lower bone mineral mass, and increased fracture risk before puberty of around 2.7-fold higher than a matched birth cohort [38, 39]. The largest randomised controlled trial (RCT) with dairy products found significantly higher gains in height, body weight, bone mineral content (BMC) and BMD in school girls aged 10 years receiving milk on school days for 2 years compared with the control group [40]. The beneficial effects of calcium and dairy products on bone mineral mass during growth in children are supported by meta-analyses of numerous clinical studies on milk-derived calcium phosphate supplementation and increased dietary dairy products, with a statistically and clinically higher gain of bone mineral content in those with low basal calcium intake [41, 42]. This significant increase in bone mass following calcium enrichment of the diet observed in pre-pubertal girls and boys [43, 44] was maintained for 1–3 years after the end of the trial [44, 45], suggesting a possible optimization of peak bone mass when calcium supply is sufficient. Increasing dairy product consumption in girls aged 10–12 years was found to be more beneficial for cortical bone mass accrual than calcium supplementation in tablet form for the same calcium intake (1000 mg/d) [22]. Besides mineral intake, a higher protein intake interacts with physical activity to enhance bone mass acquisition before the onset of puberty [3].
In summary, numerous observational studies and RCTs have shown a favourable effect of dairy products on bone health during childhood and adolescence. Conversely, avoiding milk is linked to a lower BMC [22] and increased fracture risk [22] in children.
Dairy Consumption for Osteoporosis Prevention and Reduction of Fracture Risk in Adults
European guidance on osteoporosis incorporates nutritional recommendations for bone health, including at least 1000 mg/d calcium and 800 IU/d vitamin D [30]. For comparison, Recommended Dietary Allowance (RDA, mg/d) for calcium is 1200 for postmenopausal women according to the US Institute of Medicine [46] and 600 in Japan [47]. Recommended nutritional intakes are 700 in UK [48] and recommended dietary intakes 1300 in Australian postmenopausal women [49]. Although scientific evidence to support the daily dose of calcium is rather weak, recent recommendations for postmenopausal women (aged 50–70 years) include optimal dietary protein intake of 1.0–1.2 g/kg body weight/d with at least 20–25 g of high-quality protein at each main meal, with adequate vitamin D intake at 800 IU/d to maintain serum 25-hydroxyvitamin D levels >50 nmol/L as well as calcium intake of 1000 mg/d, alongside regular physical activity/exercise 3–5 times/week combined with protein intake in close proximity to exercise [50]. Several meta-analyses support the role of calcium and vitamin D (mostly from supplements) for the prevention of osteoporosis [51–53]. As a rich source of calcium, vitamin D and protein, dairy products could be of help for reducing the risk of osteoporosis.
The healthcare costs of osteoporosis are substantial. In a review on the effects of dairy products in several medical conditions, it was suggested that intake of the recommended quantities of dairy products in the USA would yield 5-year savings (limited to healthcare costs) of $209 billion [54]. Of this, $14 billion relate to savings on the healthcare costs for osteoporosis (limited to treating fractures) [54]. A recent study shows that, especially for France, the Netherlands and Sweden, the societal burden of hip fractures associated with low calcium intake is quite substantial. Costs of fractures that might potentially be reduced by improving dairy consumption have been calculated at around €129 million in France and €34 million in Sweden [55].
Protein malnutrition is frequent in the elderly and contributes to the development of osteoporosis. Approximately 2 % (1–8 %) of the variance in BMD/BMC may be explained by dietary protein intake, possibly via an increase in serum IGF-I levels, with a reduction of proximal femur BMD loss and reduction of bone fracture risk [56–59]. However, the role of dietary protein in osteoporosis remains controversial, as diets high in animal protein (and sodium) have been linked to increased urinary calcium excretion [60, 61], which could, in theory, lead to bone loss and osteoporosis [62]. This ‘acid-ash hypothesis’—first proposed 40 years ago—theorizes that high animal protein intake results in metabolic acidosis and increased urinary excretion of calcium. However, recent human studies show that there is no relationship between nutritional variations in urinary acid excretion and calcium balance, bone metabolism and osteoporotic fracture risk [63]. Furthermore, a systematic review and meta-analysis concluded that a causal association between dietary acid load and osteoporotic bone disease is not supported by evidence [64]. The systematic review found weaknesses in the acid-ash hypothesis, in terms of a lack of direct evidence of osteoporosis progression (fragility fractures or bone strength) from the intervention studies, a lack of control for osteoporosis risk factors (such as weight loss, family history of osteoporosis, baseline BMD and oestrogen status) in the supporting prospective cohort studies, no biological mechanism functioning at physiological pH and no evidence for an adverse role on osteoporosis of phosphate, milk and grain foods in the randomised studies [64]. Changes in urinary calcium are not reflected in calcium balance [64], with high protein intakes increasing calcium excretion—but without impairing calcium balance—and possibly increasing intestinal calcium absorption [65]. Intakes of aromatic amino acids, which are prevalent in dairy foods, increase IGF-I and stimulate the intestinal absorption of calcium. A high dairy-protein diet has been demonstrated to increase urinary calcium excretion but at the same time improve calcium intake and attenuate bone loss in overweight individuals on a weight loss and weight maintenance diet [66]. A 2-year controlled trial has shown beneficial effects on BMD of daily alkali supplements [67]. In summary, theories proposing harmful effects of dairies through the production of acid are not supported by evidence.
Fracture Risk
Although, ideally, clinical outcome measures (fracture incidence) would be used to assess the impact of dairy on osteoporosis, in practice, research studies mostly utilise surrogate outcomes, including BMD [68] and bone turnover markers. Controlled trials show a statistically significant positive association between dairy food intake and BMC or BMD, and an inverse association with bone turnover markers [26, 27].
In terms of actual fracture rates, meta-analyses support some role of calcium and vitamin D (mostly from supplements) for reducing the risk [51–53]. However, globally, the relationship between dietary calcium intakes and fracture rates is not straightforward [69, 70]. Hip fracture rates are in fact highest in countries with high calcium intake and lowest where calcium intake is lower (the opposite to what might be expected) [69]. Caucasian women living in temperate climates have the highest rate of hip fracture, while rates are somewhat lower in Mediterranean and Asian women and lowest in African women [69]. This incongruence may be due to the influence of vitamin D status (linked to latitude) and high levels of bone-protective physical activity in poorer countries with low dairy intakes [71] as well as differences in life expectancy. Furthermore, there is a considerable physiological capacity to adapt to low dietary calcium intakes without compromising bone health [72].
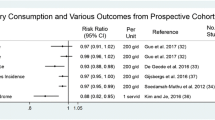
Data on the relationship between dairy food intake and fracture risk are currently limited. The available prospective studies are summarised in Table 3, comprising observational data (albeit many with sizable populations) but no RCTs. Overall, the studies to date have found variable associations between dairy consumption and fracture risk (Table 3).
A meta-analysis of prospective cohort studies in middle-aged or older men and women demonstrated no overall association between total milk intake and hip fracture risk in women, with a reduction in relative risk of fracture of 9 % (relative risk 0.91, 95 % CI 0.81–1.01) per daily glass of milk (containing approx. 300 mg calcium) among men, which requires further validation [73]. However, the data on women were strongly influenced by one study from Sweden; the authors excluded this study from the analysis on the basis of its disproportionate influence, whereupon there was a marginally significant 5 % lower hip fracture risk per glass of milk daily (pooled relative risk 0.95, 95 % CI 0.90–1.00, P = 0.049) [73].
One of the largest populations with the longest follow-up was the Nurses’ Health Study (Table 3). With a 12-year follow-up, over 77,000 women aged 34–59 years assessed dietary intake of calcium with a food-frequency questionnaire at intervals of 2–4 years, and there was no evidence that higher intakes of milk or calcium from food sources reduce fracture incidence, with relative risks of 1.45 (95 % CI 0.87–2.43) for hip fracture and 1.05 (95 % CI 0.88–1.25) for forearm fracture for women drinking two or more glasses of milk per day compared with women consuming one glass or less per week [74]. Furthermore, women consuming greater amounts of calcium from dairy foods had a modest but significantly increased risk of hip fracture. However, as the authors point out, there was no reason to suggest that dairy calcium itself was responsible for this risk [74]. At 18 years of follow-up, there was similarly no relationship between milk and fracture risk in women [2].
The association between dairy product consumption and hip fracture risk was also examined in a 12-year follow-up of the Framingham Offspring cohort [75]. There was a trend to a correlation between higher milk intake and reduced hip fracture. Participants in the higher tertiles (T2–3) of milk intake had a lower but non-significant risk of hip fracture than those in the lowest tertile (T1) (T2 HR 0.78, 95 % CI 0.37–1.63; T3 HR 0.50, 95 % CI 0.22–1.13, P trend = 0.09). Similarly, participants in the higher categories (C2–3) of yogurt intake showed a protective but non-significant association with the risk of hip fracture as compared to participants in the lowest category (C1) (C2 HR 0.39, 95 % CI 0.15–1.02, C3 HR 0.57, 95 % CI 0.19–1.68, P trend = 0.10). Participants in the highest tertile (T3) of fluid dairy intake had a lower risk of hip fracture than those in the lowest tertile (T1) (T3 vs T1, P = 0.05) (T2 HR 0.92, 95 % CI 0.46–1.87, T3 HR 0.40, 95 % CI 0.17–0.99, P trend = 0.06) [75]. A protective effect of milk was also identified in the Framingham Original cohort [76]. When milk intake was analysed as low versus medium/high intake, there was a trend to correlation, with participants with medium (>1 and <7 servings/wk) or higher (≥7 servings/wk) milk intake tending to have lower hip fracture risk than those with low (≤1 servings/wk) intake (high vs low intake: HR 0.58, 95 % CI 0.31–1.06, P = 0.078; medium vs low intake: HR 0.61, 95 % CI 0.36–1.08, P = 0.071; P trend: 0.178) [76]. Participants with medium/high milk intake (>1 serving/wk) had a 40 % lower risk of hip fracture, compared with those with low milk intake (≤1 serving/wk) (medium/high milk intake: HR 0.60, 95 % CI 0.36–1.02, P = 0.061) [76]. However, both of these populations [75, 76] were relatively small compared with the largest studies in Table 3.
A prospective study of 60,000 Swedish women aged 40–74 years who completed a self-administered food-frequency questionnaire at baseline found no correlation between dietary calcium or vitamin D intake for the primary prevention of osteoporotic fractures over 11 years of follow-up [77]. However, with 20.1 years of prospective follow-up (61,000 Swedish women aged 39–74), fermented milk products (yogurt and other soured milk products) and cheese consumption were associated with a significant decrease in fracture incidence and with lower mortality [78]. For each serving, the rate of mortality and hip fractures was reduced by 10–15 % (P < 0.001). However, in the same study, high intakes of milk (3 or more glasses of milk; more than 600 ml/day) were associated with increased fracture rate, and there was an association between higher cardiovascular mortality and high milk intake at levels exceeding the recommended doses (≥3 glasses/d; ≥600 g/d), along with a lesser effect of milk intake on cancer mortality [78]. This level of consumption is much higher than the mean milk intake of adults of most countries, and it is worth noting that dietary questionnaires were performed in 1987–1990 and 1997, when milk in Sweden was fortified with high dose of vitamin A; high levels of vitamin A intake have been linked to an increased risk of fracture [79].
It comes as no surprise that the findings of studies in this area are variable (Table 3). The studies are observational in nature and vary widely in terms of the age of the participants, background diets and length of follow-up. There is a lack of RCTs investigating the relationship between dairy food intake and fracture risk, which requires further study. Incidence of fracture is the key clinical outcome that needs to be measured in a longitudinal study; however, it is difficult to construct a dairy-free diet for the comparator group that meets nutritional requirements for calcium and vitamin D levels. Dietary studies are always difficult to control, since increasing intake of one nutrient (or foodstuff) may have an impact on other nutrients (or foodstuffs), affecting the overall dietary composition and nutrient density. With multiple nutrients known to affect bone health, it is particularly important that intake of each is considered. Quantitating intake may also be particularly difficult and variable between studies, due to differences in serving size (for example ‘a glass of milk’, ‘a pot of yogurt’). Furthermore, the study of such a long-term effect requires either prohibitively expensive long-term prospective studies or the reliance on the memory of elderly subjects concerning dairy product consumption for periods extending as far back as childhood. Since a wide range of genetic and lifestyle factors affect bone health, there is great population variability and plenty of potentially confounding factors that require controls. Although epidemiological studies potentially allow large sample numbers and stratification for confounding factors, this design does not allow any causal relationship to be established between dairy consumption and fracture risk.
Other Potential Effects of Dairy Consumption
In this section, some of the perceived beliefs associated with dairy consumption are briefly discussed, including obesity, lactose intolerance, arthritis and cardiovascular disease.
Dairy Products and Body Weight
Unfavourable body weight is a risk factor for many diseases, including osteoporosis (under weight) and osteoarthritis (over weight) [80], and obesity is a concern of many patients and health professionals alike. Dairy consumption has been studied extensively for its possible roles in body weight regulation. However, the data are conflicting. Four reviews or meta-analyses concluded that neither calcium nor dairy intakes were reliably demonstrated to aid in weight loss with or without caloric restriction [81–84]. There is evidence to suggest that consumption of dairy products reduces body fat but not necessarily body weight, due to a preservation of lean body mass [85–87]; and recent evidence from prospective cohort studies suggests a protective effect of dairy consumption on risk of overweight and obesity [88, 89]. Consumption of dairy products has been associated with decreased prevalence of metabolic-related disorders [90], and even high-fat dairy consumption within typical dietary patterns has been inversely associated with risk of obesity.
The effect of dairy consumption on weight and body composition has been investigated in two recent meta-analyses [86, 87]. The first meta-analysis of 14 RCTs involving 883 adults found that increasing dairy consumption to recommended daily intake levels in adults who do not follow any calorie-restricted diet had a small effect on weight loss, but also a decrease in fat mass and waist circumference and an increase in lean body mass [86]. When combined with energy restriction, dairy intake showed a modest additional benefit on weight reduction (−0.61 kg, 95 % CI −1.29 to 0.07, P = 0.08), greater reduction in fat mass (−0.72 kg, 95 % CI −1.29 to −0.14, P = 0.01), gain in lean mass (+0.58 kg, 95 % CI 0.18–0.99, P < 0.01) and reduction in waist circumference (−2.19 cm, 95 % CI −3.42 to −0.96, P < 0.001) compared with controls [86]. The second meta-analysis of 29 RCTs involving 2101 participants found that overall consumption of dairy products did not result in a significant reduction in weight. There was no significant difference in body weight changes between the dairy intervention and control groups (−0.14 kg, 95 % CI −0.66 to 0.38 kg); however, a significant reduction that favoured dairy products in body fat was shown (−0.45 kg, 95 % CI −0.79 to –0.11 kg) [87]. Dairy products significantly reduced body weight in the short-term interventions (−0.47 kg, 95 % CI −0.90 to −0.03 kg) but moderately increased weight gain in long-term interventions (+0.66 kg, 95 % CI −0.14 to 1.46 kg). However, in the context of energy restriction, the sub-group analysis demonstrated that consumption of dairy products reduced body weight [87]. These two meta-analyses suggest that dairy intake can aid weight and body fat loss.
Other studies show the beneficial impact of specific dairy products on weight management, particularly yogurt. Changes in diet and lifestyle factors were evaluated prospectively to determine their impact on long-term weight gain among 22,557 men and 98,320 women included in health studies in the USA [85]. Over a 4-year period, it was found that yogurt consumption was associated with a significant reduction of weight gain [85]. In another study, a cohort of 8516 Mediterranean men and women was prospectively evaluated [91]. A high (≥7 servings/wk) consumption of total and whole-fat yogurt was associated with lower incidence of overweight/obesity in comparison with low consumption (0–2 servings/wk) [91]. Together, these studies suggest a benefit of including yogurt in the diet in terms of weight management.
Mechanistic explanations for the possible association between high dairy intake and lower body weight/body fat mass found in observational studies include an effect of increased dairy calcium intake on energy balance [92, 93]. Epidemiological data have shown that low calcium intake is a risk factor for overweight and obesity. The clinical implications of this relationship have been confirmed in weight loss studies performed in low-calcium consumers in whom calcium or dairy supplementation accentuated body weight and fat loss [94–96]. Studies have demonstrated that this effect may be explained by an increase in fat oxidation and fat faecal loss as well as a facilitation of appetite control. Indeed, dietary calcium may interfere with fat absorption in the intestine by forming insoluble calcium soaps with fatty acids and/or binding of bile acids, resulting in a decrease in the digestible energy of the diet.
Dairy foods may also modulate body weight regulation by calcium independent mechanisms. Dairy proteins suppress short-term food intake, increase satiety and stimulate food intake regulatory mechanisms known to signal satiation and satiety. Milk proteins may be another important factor explaining the association between dairy consumption and healthier body weights [97].
Lactose Intolerance
In some people, milk ingestion causes symptoms of bloating, abdominal pain, flatulence and diarrhoea that can be severe enough to prompt avoidance of all dairy foods. The symptoms are caused by a deficiency of the enzyme lactase (the lactose-digesting enzyme that breaks down lactose into galactose and glucose for absorption) causing undigested lactose to increase the osmolarity in the small intestine (causing diarrhoea) and enter the colon where it is fermented by the resident microflora, resulting in gastrointestinal symptoms. While self-diagnosis of lactase deficiency is common, it is often incorrect and leads to unnecessary elimination of dairy products and their nutritional components. It may also result in osteoporosis, since an increased incidence of lactose intolerance (along with a significantly lower daily intake of calcium derived from milk) has been found among women with ‘idiopathic’ osteoporosis [98, 99].
Some years ago, the only possible treatment for lactose intolerance was the avoidance of lactose-containing foods, including most dairy products. Indeed, many believe that lactose intolerant individuals should not consume dairy products. However, the avoidance of dairy products in people with lactose intolerance is an area of controversy. Most individuals with lactose intolerance can tolerate up to 12 g of lactose (250 ml of milk, representing 300 mg of calcium and 30 % of recommended calcium intakes) without suffering gastrointestinal symptoms, although symptoms become more prominent at doses above 12 g and are appreciable after 24 g of lactose [100, 101]. The 2010 Dietary Guidelines for Americans encourage daily consumption of dairy foods and concur that it is important for people with lactose intolerance to obtain the health and nutritional benefits associated with dairy products, recommending yogurt or cheese, or smaller portions, and lactose-free varieties where necessary [13]. A National Institutes of Health (NIH) Consensus and State-of-the-Science Statement confirms that even in persons with lactose maldigestion, small amounts of milk, yogurt and hard cheese, particularly if ingested with other foods and distributed throughout the day, and reduced-lactose food may be effective management approaches, though the amount of lactose subjects with lactose intolerance can take, is based on low-quality evidence [102].
Yogurts (those with the two active bacterial cultures: Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus) and hard cheeses contain more pre-digested lactose and may be more readily tolerated than fluid milk [103, 104]. The bacterial lactase survives the acidic conditions of the stomach, apparently being physically protected within the bacterial cells and facilitated by the buffering capacity of yogurt. The increasing pH, as the yogurt enters the small intestine, and a slower gastrointestinal transit time allow the bacterial lactase to be active, digesting lactose from yogurt sufficiently to prevent symptoms in lactose intolerant people [104]. Consequently, the avoidance of all dairy products in lactose intolerant patients is no longer recommended. Dietetic advice should aim to ensure nutritional adequacy of the diet and to avoid nutritional deficiencies—in particular a low calcium intake. Since yogurt and hard cheese in particular are easily digestible and well tolerated by people who find lactose difficult to digest, they can be recommended as part of a balanced diet to help lactose intolerant people take advantage of the nutritional benefits of dairy products [105].
In summary, to varying degrees depending on the cultural history of dairy consumption, genetic lactase persistence allows most of the population to continue to consume some milk beyond the weaning period without gastrointestinal adverse effects. For people who are lactose intolerant, it is no longer necessary to avoid all dairy foods, and in particular yogurt or hard cheese are well tolerated and provide the nutritional benefits of dairy products.
Dairy Products and Joint Diseases
In people with chronic diseases, dietary manipulation is common as they try to alleviate the symptoms. This is particularly the case with painful conditions such as osteoarthritis (OA) and rheumatoid arthritis (RA). However, scientific studies are required to control for all the possible confounding factors and bias that might contribute to any perceived dietary effect, and the available evidence suggests that there is no reason why dairy consumption should be avoided.
Best practice guidelines for OA emphasize self-management including weight control and exercise [106]. There is some evidence to suggest that a Western diet and inactivity are pro-inflammatory and that low-grade inflammation and oxidative stress underlying OA often coexist with lifestyle-related risk factors and conditions [107]. While dairy products have in the past been considered as pro-inflammatory [107], more recent data do not support this hypothesis. In fact, full-fat dairy products and dairy fats have either a neutral or inverse effect on levels of circulating inflammatory biomarkers [108–112]. A meta-analysis of nutritional interventional studies performed in overweight or obese people found that dairy product consumption does not exert adverse effects on inflammatory biomarkers [113], and in an RCT among subjects with low-grade systemic inflammation, consumption of a combination of low-fat and high-fat dairy products as part of a healthy diet had no adverse effects on inflammation [114].
An association between frequent milk consumption and reduced OA progression has been found in a recent study in women, although no significant association was found in men. Analysis of 2148 participants in the Osteoarthritis Initiative identified a significant dose–response relationship between baseline milk intake and OA progression in women over a 4-year follow-up period [115]. Replication of these novel findings in other prospective studies demonstrating that increase in milk consumption leads to delay in knee OA progression is needed.
For decades, patients have used different diets to try to improve the symptoms of RA, and dietary manipulation is still widely used today. A wide range of conflicting dietary advice is available, yet a lack of scientific information regarding its efficacy often leaves sufferers vulnerable. The common dietary regimens used by people with RA include vegetarian or vegan, Mediterranean, ‘elemental’ and ‘elimination’ diets (including dairy-free diets).
Diet, nutrition and weight loss have shown some promise in alleviating some of the RA disease burden [116]. Prospective studies suggest that dietary antioxidants may be protective for RA, while high coffee consumption, alcohol intake, smoking and obesity increase the risk of developing RA [117]. Some studies suggest that certain dietary elements including polyunsaturated fatty acids (PUFAs), the Mediterranean diet and antioxidants have anti-inflammatory effects and decrease RA disease activity [118, 119].
However, the effects of dietary manipulation on RA are still uncertain due to the studies being small, single trials with moderate to high risk of bias [120, 121]. A Cochrane review of 14 RCTs and 1 controlled clinical trial, with a total of 837 patients, assessed the potential benefits and harms associated with certain dietary regimens in RA [121]. Due to heterogeneity of interventions and outcomes, baseline imbalance and inadequate data reporting, no overall effects were detected and the effects of vegan and elimination diets were uncertain [121].
Dairy products contain a high content of saturated fatty acids, and many of the shorter chain fatty acids found in milk fat have beneficial health effects, with important immune response functions. There is also evidence that the proteins, fats and calcium in milk are beneficial in inflammation [122]. Given the lack of understanding of the nutritional requirements in RA, plus the variability in its clinical course, it is difficult to produce specific dietary recommendations for RA. Elimination diets, particularly those with low intakes of dairy products, should be discouraged [123], and suspected food intolerance tested under strict clinical supervision [124]. Nutrient mega-dosing is inadvisable, although dietary supplementation with calcium, vitamin D, folic acid or multivitamins and minerals may be recommended where necessary [124].
In summary, more research is needed, but based on the available data, there appears to be no evidence why patients with OA or RA should avoid dairy consumption, considering the overall beneficial effect of dairy products for health.
Dairy Consumption and Cardiovascular Disease
Globally, cardiovascular disease is a major cause of mortality [125], and the influence of diet on known cardiovascular risk factors such as blood pressure, serum cholesterol and body weight, is well established. Consequently, the DASH (Dietary Approaches to Stop Hypertension)-style diet—which incorporates moderate amounts of low-fat dairy—protects against cardiovascular disease, coronary heart disease, stroke and heart failure [126].
A review of the evidence concluded that most studies report no association between dairy consumption and increased risk of cardiovascular disease [127], with a meta-analysis of RCT data finding no significant impact on various cardio-metabolic risk factors following increased dairy consumption (mean increase of 3.6 serves per day), aside from a slight increase in body weight [128]. In fact, dairy consumption may be protective against cardiovascular disease, with beneficial effects demonstrated on known risk factors such as hypertension [129, 130] as well as clinical outcomes [131–133]. For example, meta-analyses of observational data have reported inverse associations between overall cardiovascular disease and milk intake (4 studies; RR 0.94 per 200 mL/d, 95 % CI 0.89–0.99) [132] and dairy consumption (9 studies; RR 0.88, 95 % CI 0.81–0.96) [131]. There was also an inverse association between dairy consumption and overall risk of stroke (12 studies; RR 0.87, 95 % CI 0.77–0.99) [131], whilst another meta-analysis found that total dairy (RR 0.88, 95 % CI 0.82–0.94), low-fat dairy (RR 0.91, 95 % CI 0.85 to 0.97), fermented milk (RR 0.80, 95 % CI 0.71–0.89) and cheese (RR 0.94, 95 % CI 0.89–0.995)] were significantly associated with reduced risk of stroke [133]. There was no significant association between the risk of stroke and intake of full-fat dairy, non-fermented milk, butter and cream [133].
There is currently a lack of research into the effect of full-fat dairy on cardiovascular disease [127], and the relative impact of high-fat dairy versus low-fat dairy is difficult to establish from the current evidence base. European and American guidelines for preventing cardiovascular disease recommend a reduction in total dietary saturated fat [134, 135]. However, it is becoming clear that all saturated fatty acids are not equal. For example, in a study using a food-frequency questionnaire, a higher intake of saturated fatty acids from meat increased incident cardiovascular disease, whereas higher intake of saturated fatty acids from dairy reduced the risk [136]. Research into understanding the metabolic effects of individual fatty acids is still in the early stages, but it would seem that structural characteristics of the individual fatty acid and the food matrix within which it is consumed are likely to have an effect. A meta-analysis of prospective cohort studies reporting circulating fatty acid composition revealed the variable effects of individual saturated fatty acids, with a higher coronary risk for palmitic (RR 1.15, 95 % CI 0.96–1.37) and stearic (RR 1.23, 95 % CI 0.93–1.61) acids, and a significantly lower risk for margaric acid (RR 0.77, 95 % CI 0.63–0.93) [137]. Whilst the saturated fatty acids in whole milk and butter increase low-density lipoprotein, they also increase high-density lipoprotein, which leaves the ratio of total cholesterol to high-density lipoprotein unchanged, or slightly lowered [127].
A recent meta-analysis has concluded that calcium supplements without co-administered vitamin D were associated with an increased risk of myocardial infarction [138]. However, there was no increased risk when calcium was of dietary origin.
In summary, dairy consumption does not seem to significantly increase the risk of cardiovascular disease, particularly if low-fat, and more research is needed for full-fat dairy products.
Discussion
Milk provides a package of essential nutrients that are difficult to obtain in low-dairy or dairy-free diets. Dairy products may represent a useful source of dietary calcium, and for many people it is not possible to achieve recommended daily calcium intakes with a dairy-free diet. The link between dairy product intakes and fracture risk reduction (Table 3) is still challenged. To demonstrate an effect on fracture risk would require further large and well-designed rigorously controlled studies.
Nevertheless, it can be inferred from the above-mentioned data that regular consumption of dairy throughout life is likely to be beneficial for skeletal health. Besides bone health, more controversial are the beliefs around including dairy as part of a healthy balanced diet. Among some members of the public and health professionals, doubts persist that dairy may be detrimental to health. In some cases, these are simply beliefs or a consequence of misinformation, but in other cases plausible mechanisms exist, casting doubt in even the most scientific mind. This review has discussed some of the most common concerns over the inclusion of dairy in the diet, including those of people with weight management issues or lactose intolerance, with OA or RA or with cardiovascular disease. The overall message is that the benefits outweigh any (in many cases perceived, not real) harm.
Lactose intolerance can prompt an avoidance of all dairies, but this is not necessary in most people. In particular, yogurt and hard cheese are well tolerated and provide the nutritional benefits of dairy products.
The effect of dairy consumption on body weight and composition has been investigated extensively, with conflicting results. There may be a weak association between dairy consumption and a possible small weight reduction, with decreases in fat mass and waist circumference and increases in lean body mass.
The evidence for any role of dairy foods in inflammation is conflicting and limited to small observational or clinical trials. Although dairy foods have in the past been considered pro-inflammatory [107], studies of dairy consumption demonstrate that full-fat dairy products and dairy fats in fact have either a neutral or inverse effect on levels of circulating inflammatory biomarkers [108–114]. Recently, a single observational study suggested a reduction in OA progression associated with frequent milk consumption among women [115]. Dietary guidelines are difficult to define for RA, but patients should be discouraged from eliminating dairy [123] in their attempts to control their symptoms, and a balanced nutritionally complete diet should be advised.
Most studies report no association between dairy consumption and increased risk of cardiovascular disease [127], and in fact low-fat dairy may be protective [131–133]. More research is needed in this respect for full-fat dairy products.
In conclusion, dairy products may represent a valuable dietary source of calcium due to their high calcium and nutrient contents, high absorptive rate, availability and relatively low cost. Numerous national nutritional recommendations are for 3 servings of dairy products per day (for example, 1 glass of milk, 1 portion of cheese, 1 yogurt), an amount that provides the recommended daily intake of calcium. By understanding fully the accumulating scientific data in this area, health professionals can play an important role in dispelling these beliefs surrounding dairy products.
References
Smit E, Nieto FJ, Crespo CJ, Mitchell P (1999) Estimates of animal and plant protein intake in US adults: results from the Third National Health and Nutrition Examination Survey, 1988–1991. J Am Diet Assoc 99(7):813–820. doi:10.1016/S0002-8223(99)00193-5
Feskanich D, Willett WC, Colditz GA (2003) Calcium, vitamin D, milk consumption, and hip fractures: a prospective study among postmenopausal women. Am J Clin Nutr 77(2):504–511
Chevalley T, Bonjour JP, Ferrari S, Rizzoli R (2008) High-protein intake enhances the positive impact of physical activity on BMC in prepubertal boys. J Bone Miner Res 23(1):131–142. doi:10.1359/jbmr.070907
Vissers PA, Streppel MT, Feskens EJ, de Groot LC (2011) The contribution of dairy products to micronutrient intake in the Netherlands. J Am Coll Nutr 30(5 Suppl 1):415S–421S
Skinner ML, Simpson JA, Buchholz AC (2011) Dietary and total calcium intakes are associated with lower percentage total body and truncal fat in young, healthy adults. J Am Coll Nutr 30(6):484–490
Gueguen L, Pointillart A (2000) The bioavailability of dietary calcium. J Am Coll Nutr 19(2 Suppl):119S–136S
Gueguen L (2001) Calcium, phosphorus. Apports Nutritionnels Conseillees pour la Population Francaise (Recommended nutrient intakes for the French population). Tech & Doc, Paris
Bonjour JP (2011) Calcium and phosphate: a duet of ions playing for bone health. J Am Coll Nutr 30(5 Suppl 1):438S–448S
Elmadfa I (2009) European Nutrition and Health Report 2009. Forum Nutr. Basel, Switzerland, vol 69. Karger, New York
Lopes AC, Caiaffa WT, Sichieri R, Mingoti SA, Lima-Costa MF (2005) Nutrient consumption by adults and seniors in a population-based study: the Bambui Project. Cadernos de saude publica 21(4):1201–1209. doi:10.1590/S0102-311x2005000400022
Heaney RP (2009) Dairy and bone health. J Am Coll Nutr 28(Suppl 1):82S–90S
Caroli A, Poli A, Ricotta D, Banfi G, Cocchi D (2011) Invited review: dairy intake and bone health: a viewpoint from the state of the art. J Dairy Sci 94(11):5249–5262. doi:10.3168/jds.2011-4578
Agriculture USDo (2010) Dietary guidelines for Americans, 7th edn. Agriculture USDo, Washington, DC
Agriculture USDo (2013) USDA National Nutrient Database for Standard Reference, Release 26
Fulgoni VL 3rd, Keast DR, Auestad N, Quann EE (2011) Nutrients from dairy foods are difficult to replace in diets of Americans: food pattern modeling and an analyses of the National Health and Nutrition Examination Survey 2003–2006. Nutr Res 31(10):759–765. doi:10.1016/j.nutres.2011.09.017
Marcoe K, Juan W, Yamini S, Carlson A, Britten P (2006) Development of food group composites and nutrient profiles for the MyPyramid Food Guidance System. J Nutr Educ Behav 38(6 Suppl):S93–S107. doi:10.1016/j.jneb.2006.05.014
Gao X, Wilde PE, Lichtenstein AH, Tucker KL (2006) Meeting adequate intake for dietary calcium without dairy foods in adolescents aged 9 to 18 years (National Health and Nutrition Examination Survey 2001–2002). J Am Diet Assoc 106(11):1759–1765. doi:10.1016/j.jada.2006.08.019
Weaver CM (2009) Should dairy be recommended as part of a healthy vegetarian diet? Point. Am J Clin Nutr 89(5):1634S–1637S. doi:10.3945/ajcn.2009.26736O
Keller JL, Lanou A, Barnard ND (2002) The consumer cost of calcium from food and supplements. J Am Diet Assoc 102(11):1669–1671
Weaver CM, Proulx WR, Heaney R (1999) Choices for achieving adequate dietary calcium with a vegetarian diet. Am J Clin Nutr 70:543S–548S
Rizzoli R (2008) Nutrition: its role in bone health. Best Pract Res Clin Endocrinol Metab 22(5):813–829. doi:10.1016/j.beem.2008.08.005
Cheng S, Lyytikainen A, Kroger H, Lamberg-Allardt C, Alen M, Koistinen A, Wang QJ, Suuriniemi M, Suominen H, Mahonen A, Nicholson PH, Ivaska KK, Korpela R, Ohlsson C, Vaananen KH, Tylavsky F (2005) Effects of calcium, dairy product, and vitamin D supplementation on bone mass accrual and body composition in 10-12-y-old girls: a 2-y randomized trial. Am J Clin Nutr 82(5):1115–1126
Li Y, Tome D, Desjeux JF (1989) Indirect effect of casein phosphopeptides on calcium absorption in rat ileum in vitro. Reprod Nutr Dev 29(2):227–233
Bonjour JP, Kraenzlin M, Levasseur R, Warren M, Whiting S (2013) Dairy in adulthood: from foods to nutrient interactions on bone and skeletal muscle health. J Am Coll Nutr 32(4):251–263. doi:10.1080/07315724.2013.816604
Bailey RL, Dodd KW, Goldman JA, Gahche JJ, Dwyer JT, Moshfegh AJ, Sempos CT, Picciano MF (2010) Estimation of total usual calcium and vitamin D intakes in the United States. J Nutr 140(4):817–822
Manios Y, Moschonis G, Trovas G, Lyritis GP (2007) Changes in biochemical indexes of bone metabolism and bone mineral density after a 12-mo dietary intervention program: the Postmenopausal Health Study. Am J Clin Nutr 86(3):781–789
Moschonis G, Katsaroli I, Lyritis GP, Manios Y (2010) The effects of a 30-month dietary intervention on bone mineral density: the Postmenopausal Health Study. Br J Nutr 104(1):100–107. doi:10.1017/S000711451000019X
Prince R, Devine A, Dick I, Criddle A, Kerr D, Kent N, Price R, Randell A (1995) The effects of calcium supplementation (milk powder or tablets) and exercise on bone density in postmenopausal women. J Bone Miner Res 10(7):1068–1075
Body JJ, Bergmann P, Boonen S, Boutsen Y, Bruyere O, Devogelaer JP, Goemaere S, Hollevoet N, Kaufman JM, Milisen K, Rozenberg S, Reginster JY (2011) Non-pharmacological management of osteoporosis: a consensus of the Belgian Bone Club. Osteoporos Int 22(11):2769–2788. doi:10.1007/s00198-011-1545-x
Kanis JA, McCloskey EV, Johansson H, Cooper C, Rizzoli R, Reginster JY, Scientific Advisory Board of the European Society for C, Economic Aspects of O, Osteoarthritis, the Committee of Scientific Advisors of the International Osteoporosis F (2013) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 24(1):23–57. doi:10.1007/s00198-012-2074-y
Sunyecz JA (2008) The use of calcium and vitamin D in the management of osteoporosis. Ther Clin Risk Manag 4(4):827–836
Rizzoli R, Bianchi ML, Garabedian M, McKay HA, Moreno LA (2010) Maximizing bone mineral mass gain during growth for the prevention of fractures in the adolescents and the elderly. Bone 46(2):294–305. doi:10.1016/j.bone.2009.10.005
Ferrari S, Rizzoli R, Bonjour JP (1999) Genetic aspects of osteoporosis. Curr Opin Rheumatol 11(4):294–300
Rizzoli R (2014) Dairy products, yogurts, and bone health. Am J Clin Nutr 99(5 Suppl):1256S–1262S. doi:10.3945/ajcn.113.073056
Ganpule A, Yajnik CS, Fall CH, Rao S, Fisher DJ, Kanade A, Cooper C, Naik S, Joshi N, Lubree H, Deshpande V, Joglekar C (2006) Bone mass in Indian children–relationships to maternal nutritional status and diet during pregnancy: the Pune Maternal Nutrition Study. J Clin Endocrinol Metab 91(8):2994–3001. doi:10.1210/jc.2005-2431
Cole ZA, Gale CR, Javaid MK, Robinson SM, Law C, Boucher BJ, Crozier SR, Godfrey KM, Dennison EM, Cooper C (2009) Maternal dietary patterns during pregnancy and childhood bone mass: a longitudinal study. J Bone Miner Res 24(4):663–668. doi:10.1359/jbmr.081212
Dror DK, Allen LH (2014) Dairy product intake in children and adolescents in developed countries: trends, nutritional contribution, and a review of association with health outcomes. Nutr Rev 72(2):68–81. doi:10.1111/nure.12078
Goulding A, Rockell JE, Black RE, Grant AM, Jones IE, Williams SM (2004) Children who avoid drinking cow’s milk are at increased risk for prepubertal bone fractures. J Am Diet Assoc 104(2):250–253. doi:10.1016/j.jada.2003.11.008
Konstantynowicz J, Nguyen TV, Kaczmarski M, Jamiolkowski J, Piotrowska-Jastrzebska J, Seeman E (2007) Fractures during growth: potential role of a milk-free diet. Osteoporos Int 18(12):1601–1607. doi:10.1007/s00198-007-0397-x
Du X, Zhu K, Trube A, Zhang Q, Ma G, Hu X, Fraser DR, Greenfield H (2004) School-milk intervention trial enhances growth and bone mineral accretion in Chinese girls aged 10–12 years in Beijing. Br J Nutr 92(1):159–168. doi:10.1079/BJN20041118
Winzenberg T, Shaw K, Fryer J, Jones G (2006) Effects of calcium supplementation on bone density in healthy children: meta-analysis of randomised controlled trials. BMJ 333(7572):775. doi:10.1136/bmj.38950.561400.55
Huncharek M, Muscat J, Kupelnick B (2008) Impact of dairy products and dietary calcium on bone-mineral content in children: results of a meta-analysis. Bone 43(2):312–321. doi:10.1016/j.bone.2008.02.022
Bonjour JP, Carrie AL, Ferrari S, Clavien H, Slosman D, Theintz G, Rizzoli R (1997) Calcium-enriched foods and bone mass growth in prepubertal girls: a randomized, double-blind, placebo-controlled trial. J Clin Invest 99(6):1287–1294. doi:10.1172/JCI119287
Chevalley T, Bonjour JP, Ferrari S, Hans D, Rizzoli R (2005) Skeletal site selectivity in the effects of calcium supplementation on areal bone mineral density gain: a randomized, double-blind, placebo-controlled trial in prepubertal boys. J Clin Endocrinol Metab 90(6):3342–3349. doi:10.1210/jc.2004-1455
Bonjour JP, Chevalley T, Ammann P, Slosman D, Rizzoli R (2001) Gain in bone mineral mass in prepubertal girls 3.5 years after discontinuation of calcium supplementation: a follow-up study. Lancet 358(9289):1208–1212. doi:10.1016/S0140-6736(01)06342-5
Ross AC, Manson JE, Abrams SA, Aloia JF, Brannon PM, Clinton SK, Durazo-Arvizu RA, Gallagher JC, Gallo RL, Jones G, Kovacs CS, Mayne ST, Rosen CJ, Shapses SA (2011) The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab 96(1):53–58. doi:10.1210/jc.2010-2704
Okano T (2003) Recommended daily allowance and dietary reference intakes for vitamin D and calcium in Japanese. Clin Calcium 13(7):876–881. doi:CliCa0307876881
Theobald HE (2005) Dietary calcium and health - http://www.nutrition.org.uk/attachments/105_Dietary%20calcium%20and%20health.pdf
Rizzoli R, Stevenson JC, Bauer JM, van Loon LJC, Walrand S, Kanis JA, Cooper C, Brandi M-L, Diez-Perez A, Reginster J-Y, Force ftET (2014) The role of dietary protein and vitamin D in maintaining musculoskeletal health in postmenopausal women: A consensus statement from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Maturitas 79:122–132. doi:10.1016/j.maturitas.2014.07.005
Tang BMP, Eslick GD, Nowson C, Smith C, Bensoussan A (2007) Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet 370(9588):657–666. doi:10.1016/s0140-6736(07)61342-7
Boonen S, Lips P, Bouillon R, Bischoff-Ferrari HA, Vanderschueren D, Haentjens P (2007) Need for additional calcium to reduce the risk of hip fracture with vitamin d supplementation: evidence from a comparative metaanalysis of randomized controlled trials. J Clin Endocrinol Metab 92(4):1415–1423. doi:10.1210/jc.2006-1404
Nowson CA (2010) Prevention of fractures in older people with calcium and vitamin D. Nutrients 2(9):975–984
McCarron DA, Heaney RP (2004) Estimated healthcare savings associated with adequate dairy food intake. Am J Hypertens 17(1):88–97
Lotters FJ, Lenoir-Wijnkoop I, Fardellone P, Rizzoli R, Rocher E, Poley MJ (2013) Dairy foods and osteoporosis: an example of assessing the health-economic impact of food products. Osteoporos Int 24(1):139–150. doi:10.1007/s00198-012-1998-6
Schurch MA, Rizzoli R, Slosman D, Vadas L, Vergnaud P, Bonjour JP (1998) Protein supplements increase serum insulin-like growth factor-I levels and attenuate proximal femur bone loss in patients with recent hip fracture. A randomized, double-blind, placebo-controlled trial. Ann Intern Med 128(10):801–809
Munger RG, Cerhan JR, Chiu BC (1999) Prospective study of dietary protein intake and risk of hip fracture in postmenopausal women. Am J Clin Nutr 69(1):147–152
Wengreen HJ, Munger RG, West NA, Cutler DR, Corcoran CD, Zhang J, Sassano NE (2004) Dietary protein intake and risk of osteoporotic hip fracture in elderly residents of Utah. J Bone Miner Res 19(4):537–545. doi:10.1359/jbmr.040208
Darling AL, Millward DJ, Torgerson DJ, Hewitt CE, Lanham-New SA (2009) Dietary protein and bone health: a systematic review and meta-analysis. Am J Clin Nutr 90(6):1674–1692. doi:10.3945/ajcn.2009.27799
Barzel US, Massey LK (1998) Excess dietary protein can adversely affect bone. J Nutr 128(6):1051–1053
Frassetto L, Morris RC Jr, Sellmeyer DE, Todd K, Sebastian A (2001) Diet, evolution and aging–the pathophysiologic effects of the post-agricultural inversion of the potassium-to-sodium and base-to-chloride ratios in the human diet. Eur J Nutr 40(5):200–213
Sellmeyer DE, Stone KL, Sebastian A, Cummings SR (2001) A high ratio of dietary animal to vegetable protein increases the rate of bone loss and the risk of fracture in postmenopausal women. Study of Osteoporotic Fractures Research Group. Am J Clin Nutr 73(1):118–122
Bonjour JP (2013) Nutritional disturbance in acid-base balance and osteoporosis: a hypothesis that disregards the essential homeostatic role of the kidney. Br J Nutr 110(7):1168–1177
Fenton TR, Tough SC, Lyon AW, Eliasziw M, Hanley DA (2011) Causal assessment of dietary acid load and bone disease: a systematic review & meta-analysis applying Hill’s epidemiologic criteria for causality. Nutr J 10:41. doi:10.1186/1475-2891-10-41
Calvez J, Poupin N, Chesneau C, Lassale C, Tome D (2012) Protein intake, calcium balance and health consequences. Eur J Clin Nutr 66(3):281–295. doi:10.1038/ejcn.2011.196
Thorpe MP, Jacobson EH, Layman DK, He X, Kris-Etherton PM, Evans EM (2008) A diet high in protein, dairy, and calcium attenuates bone loss over twelve months of weight loss and maintenance relative to a conventional high-carbohydrate diet in adults. J Nutr 138(6):1096–1100
Jehle S, Hulter HN, Krapf R (2013) Effect of potassium citrate on bone density, microarchitecture, and fracture risk in healthy older adults without osteoporosis: a randomized controlled trial. J Clin Endocrinol Metab 98(1):207–217. doi:10.1210/jc.2012-3099
Bruyère O, Rizzoli R, Coxam V, Avouac B, Chevalier T, Fabien-Soulé V, Kanis JA, Kaufman JM, Tsouderos Y, Reginster JY (2012) Assessment of health claims in the field of bone: a view of the Group for the Respect of Ethics and Excellence in Science (GREES). Osteoporos Int 23(1):193–199
Prentice A (2004) Diet, nutrition and the prevention of osteoporosis. Public Health Nutr 7(1A):227–243
Kanis JA, Odén A, McCloskey EV, Johansson H, Wahl DA, Cooper C (2012) A systematic review of hip fracture incidence and probability of fracture worldwide. Osteoporos Int 23(9):2239–2256
Prentice AM (2014) Dairy products in global public health. Am J Clin Nutr 99(5 Suppl):1212S–1216S. doi:10.3945/ajcn.113.073437
Olausson H, Goldberg GR, Laskey MA, Schoenmakers I, Jarjou LM, Prentice A (2012) Calcium economy in human pregnancy and lactation. Nutr Res Rev 25(1):40–67. doi:10.1017/s0954422411000187
Bischoff-Ferrari HA, Dawson-Hughes B, Baron JA, Kanis JA, Orav EJ, Staehelin HB, Kiel DP, Burckhardt P, Henschkowski J, Spiegelman D, Li R, Wong JB, Feskanich D, Willett WC (2011) Milk intake and risk of hip fracture in men and women: a meta-analysis of prospective cohort studies. J Bone Miner Res 26(4):833–839. doi:10.1002/jbmr.279
Feskanich D, Willett WC, Stampfer MJ, Colditz GA (1997) Milk, dietary calcium, and bone fractures in women: a 12-year prospective study. Am J Public Health 87(6):992–997
Sahni S, Tucker KL, Kiel DP, Quach L, Casey VA, Hannan MT (2013) Milk and yogurt consumption are linked with higher bone mineral density but not with hip fracture: the Framingham Offspring Study. Arch Osteoporos 8(1–2):119. doi:10.1007/s11657-013-0119-2
Sahni S, Mangano KM, Tucker KL, Kiel DP, Casey VA, Hannan MT (2014) Protective association of milk intake on the risk of hip fracture: results from the Framingham Original Cohort. J Bone Miner Res 29(8):1756–1762
Michaelsson K, Melhus H, Bellocco R, Wolk A (2003) Dietary calcium and vitamin D intake in relation to osteoporotic fracture risk. Bone 32(6):694–703
Michaëlsson K, Wolk A, Langenskiöld S, Basu S, Warensjö Lemming E, Melhus H, Byberg L (2014) Milk intake and risk of mortality and fractures in women and men: cohort studies. BMJ 349:g6015
Livsmedelsverket (2009) The National Food Agency, Sweden. Berikning av mjölk med vitamin A (Vitamin A fortification of milk). LIVSMEDELSVERKET 2009-12-11 Nutritionsavdelningen. Available from: http://www.slv.se/upload/dokument/mat/vitaminermineraler/Vitamin%20A%20berikning%20av%20mj%C3%B6lk.pdf. Accessed 05 Jan 2014
Bultink IE, Lems WF (2013) Osteoarthritis and osteoporosis: what is the overlap? Curr Rheumatol Rep 15(5):328
Lanou AJ, Barnard ND (2008) Dairy and weight loss hypothesis: an evaluation of the clinical trials. Nutr Rev 66(5):272–279. doi:10.1111/j.1753-4887.2008.00032.x
Trowman R, Dumville JC, Hahn S, Torgerson DJ (2006) A systematic review of the effects of calcium supplementation on body weight. Br J Nutr 95(6):1033–1038
Huang TT, McCrory MA (2005) Dairy intake, obesity, and metabolic health in children and adolescents: knowledge and gaps. Nutr Rev 63(3):71–80
Winzenberg T, Shaw K, Fryer J, Jones G (2007) Calcium supplements in healthy children do not affect weight gain, height, or body composition. Obesity (Silver Spring, Md) 15(7):1789–1798. doi:10.1038/oby.2007.213
Mozaffarian D, Hao T, Rimm EB, Willett WC, Hu FB (2011) Changes in diet and lifestyle and long-term weight gain in women and men. N Engl J Med 364(25):2392–2404. doi:10.1056/NEJMoa1014296
Abargouei AS, Janghorbani M, Salehi-Marzijarani M, Esmaillzadeh A (2012) Effect of dairy consumption on weight and body composition in adults: a systematic review and meta-analysis of randomized controlled clinical trials. Int J Obes 36(12):1485–1493. doi:10.1038/ijo.2011.269
Chen M, Pan A, Malik VS, Hu FB (2012) Effects of dairy intake on body weight and fat: a meta-analysis of randomized controlled trials. Am J Clin Nutr 96(4):735–747. doi:10.3945/ajcn.112.037119
Louie JC, Flood VM, Hector DJ, Rangan AM, Gill TP (2011) Dairy consumption and overweight and obesity: a systematic review of prospective cohort studies. Obes Rev 12(7):e582–e592. doi:10.1111/j.1467-789X.2011.00881.x
Dougkas A, Reynolds CK, Givens ID, Elwood PC, Minihane AM (2011) Associations between dairy consumption and body weight: a review of the evidence and underlying mechanisms. Nutr Res Rev 24(1):72–95. doi:10.1017/s095442241000034x
McGregor RA, Poppitt SD (2013) Milk protein for improved metabolic health: a review of the evidence. Nutr Metab (Lond) 10(1):46. doi:10.1186/1743-7075-10-46.)
Martinez-Gonzalez MA, Sayon-Orea C, Ruiz-Canela M, de la Fuente C, Gea A, Bes-Rastrollo M (2014) Yogurt consumption, weight change and risk of overweight/obesity: The SUN cohort study. Nutr Metab Cardiovasc Dis. doi:10.1016/j.numecd.02014.00105.00015
McCarron DA, Morris CD, Henry HJ, Stanton JL (1984) Blood pressure and nutrient intake in the United States. Science 224(4656):1392–1398
Zemel MB, Shi H, Greer B, Dirienzo D, Zemel PC (2000) Regulation of adiposity by dietary calcium. FASEB J 14(9):1132–1138
Zemel MB, Thompson W, Milstead A, Morris K, Campbell P (2004) Calcium and dairy acceleration of weight and fat loss during energy restriction in obese adults. Obes Res 12(4):582–590. doi:10.1038/oby.2004.67
Zemel MB, Richards J, Mathis S, Milstead A, Gebhardt L, Silva E (2005) Dairy augmentation of total and central fat loss in obese subjects. Int J Obes 29(4):391–397. doi:10.1038/sj.ijo.0802880
Zemel MB, Richards J, Milstead A, Campbell P (2005) Effects of calcium and dairy on body composition and weight loss in African-American adults. Obes Res 13(7):1218–1225. doi:10.1038/oby.2005.144
Astrup A, Chaput JP, Gilbert JA, Lorenzen JK (2010) Dairy beverages and energy balance. Physiol Behav 100(1):67–75. doi:10.1016/j.physbeh.2010.01.039
Birge SJ Jr, Keutmann HT, Cuatrecasas P, Whedon GD (1967) Osteoporosis, intestinal lactase deficiency and low dietary calcium intake. N Engl J Med 276(8):445–448. doi:10.1056/nejm196702232760805
Finkenstedt G, Skrabal F, Gasser RW, Braunsteiner H (1986) Lactose absorption, milk consumption, and fasting blood glucose concentrations in women with idiopathic osteoporosis. Br Med J (Clin Res Ed) 292(6514):161–162
Wilt TJ, Shaukat A, Shamliyan T, Taylor BC, MacDonald R, Tacklind J, Rutks I, Schwarzenberg SJ, Kane RL, Levitt M (2010) Lactose intolerance and health. Evid Rep/Technol Assess 192:1–410
Savaiano DA, Boushey CJ, McCabe GP (2006) Lactose intolerance symptoms assessed by meta-analysis: a grain of truth that leads to exaggeration. J Nutr 136(4):1107–1113
Suchy FJ, Brannon PM, Carpenter TO, Fernandez JR, Gilsanz V, Gould JB, Hall K, Hui SL, Lupton J, Mennella J, Miller NJ, Osganian SK, Sellmeyer DE, Wolf MA (2010) NIH consensus development conference statement: lactose intolerance and health. NIH Consens State Sci Statements 27(2):1–27
McBean LD, Miller GD (1998) Allaying fears and fallacies about lactose intolerance. J Am Diet Assoc 98(6):671–676. doi:10.1016/s0002-8223(98)00152-7
Savaiano DA (2014) Lactose digestion from yogurt: mechanism and relevance. Am J Clin Nutr 99(5 Suppl):1251S–1255S. doi:10.3945/ajcn.113.073023
Dainese-Plichon R, Schneider S, Piche T, Hébuterne X (2014) Lactose malabsorption and intolerance in adult subjects [article in French]. Nutr Clin Métabol 28(1):46–51
Litwic A, Edwards MH, Dennison EM, Cooper C (2013) Epidemiology and burden of osteoarthritis. Br Med Bull 105:185–199. doi:10.1093/bmb/lds038
Dean E, Gormsen Hansen R (2012) Prescribing optimal nutrition and physical activity as “first-line” interventions for best practice management of chronic low-grade inflammation associated with osteoarthritis: evidence synthesis. Arthritis 2012:560634. doi:10.1155/2012/560634
Nestel PJ, Pally S, MacIntosh GL, Greeve MA, Middleton S, Jowett J, Meikle PJ (2012) Circulating inflammatory and atherogenic biomarkers are not increased following single meals of dairy foods. Eur J Clin Nutr 66:25–31
Gagliardi AC, Maranhao RC, de Sousa HP, Schaefer EJ, Santos RD (2010) Effects of margarines and butter consumption on lipid profiles, inflammation markers and lipid transfer to HDL particles in free-living subjects with the metabolic syndrome. Eur J Clin Nutr 64:1141–1149
Raff M, Tholstrup T, Basu S, Nonboe P, Sorensen MT, Straarup EM (2008) A diet rich in conjugated linoleic acid and butter increases lipid peroxidation but does not affect atherosclerotic, inflammatory, or diabetic risk markers in healthy young men. J Nutr 138:509–514
Panagiotakos DB, Pitsavos CH, Zampelas AD, Chrysohoou CA, Stefanadis CI (2010) Dairy products consumption is associated with decreased levels of inflammatory markers related to cardiovascular disease in apparently healthy adults: the ATTICA study. J Am Coll Nutr 29:357–364
Wang H, Steffen LM, Vessby B, Basu S, Steinberger J, Moran A, Jacobs DR, Hong CP, Sinaiko A (2011) Obesity modifies the relations between serum markers of dairy fats and inflammation and oxidative stress among adolescents. Obesity (Silver Spring, Md) 19:2404–2410
Labonte ME, Couture P, Richard C, Desroches S, Lamarche B (2013) Impact of dairy products on biomarkers of inflammation: a systematic review of randomized controlled nutritional intervention studies in overweight and obese adults. Am J Clin Nutr 97:706–717
Labonte ME, Cyr A, Abdullah MM, Lepine MC, Vohl MC, Jones P, Couture P, Lamarche B (2014) Dairy product consumption has no impact on biomarkers of inflammation among men and women with low-grade systemic inflammation. J Nutr 144:1760–1767
Lu B, Driban JB, Duryea J, McAlindon T, Lapane KL, Eaton CB (2014) Milk consumption and progression of medial tibiofemoral knee osteoarthritis: data from the osteoarthritis initiative. Arthritis Care Res 66(6):802–809. doi:10.1002/acr.22297
Li S, Micheletti R (2011) Role of diet in rheumatic disease. Rheum Dis Clin North Am 37(1):119–133. doi:10.1016/j.rdc.2010.11.006
Lahiri M, Morgan C, Symmons DP, Bruce IN (2012) Modifiable risk factors for RA: prevention, better than cure? Rheumatology (Oxford) 51(3):499–512. doi:10.1093/rheumatology/ker299
Gonzalez Cernadas L, Rodriguez-Romero B, Carballo-Costa L (2014) Importance of nutritional treatment in the inflammatory process of rheumatoid arthritis patients; a review. Nutr Hosp 29(2):237–245. doi:10.3305/nh.2014.29.2.7067
Stamp LK, James MJ, Cleland LG (2005) Diet and rheumatoid arthritis: a review of the literature. Semin Arthritis Rheum 35(2):77–94. doi:10.1016/j.semarthrit.2005.05.001
Smedslund G, Byfuglien MG, Olsen SU, Hagen KB (2010) Effectiveness and safety of dietary interventions for rheumatoid arthritis: a systematic review of randomized controlled trials. J Am Diet Assoc 110(5):727–735. doi:10.1016/j.jada.2010.02.010
Hagen KB, Byfuglien MG, Falzon L, Olsen SU, Smedslund G (2009) Dietary interventions for rheumatoid arthritis. Cochrane Database Syst Rev. doi:10.1002/14651858.CD006400.pub2
Lawrence GD (2013) Dietary fats and health: dietary recommendations in the context of scientific evidence. Adv Nutr 4(3):294–302. doi:10.3945/an.113.003657
Gossec L, Pavy S, Pham T, Constantin A, Poiraudeau S, Combe B, Flipo RM, Goupille P, Le Loët X, Mariette X, Puéchal X, Wendling D, Schaeverbeke T, Sibilia J, Tebib J, Cantagrel A, Dougados M (2006) Nonpharmacological treatments in early rheumatoid arthritis: clinical practice guidelines based on published evidence and expert opinion. Joint Bone Spine 73(4):396–402
Martin RH (1998) The role of nutrition and diet in rheumatoid arthritis. Proc Nutr Soc 57(2):231–234
WHO (2015) World Health Organization. Cardiovascular diseases (CVDs). Fact sheet N°317. Updated January 2015. http://www.who.int/mediacentre/factsheets/fs317/en/. Accessed 21 July 2015
Salehi-Abargouei A, Maghsoudi Z, Shirani F, Azadbakht L (2013) Effects of Dietary Approaches to Stop Hypertension (DASH)-style diet on fatal or nonfatal cardiovascular diseases–incidence: a systematic review and meta-analysis on observational prospective studies. Nutrition 29(4):611–618
Huth PJ, Park KM (2012) Influence of dairy product and milk fat consumption on cardiovascular disease risk: a review of the evidence. Adv Nutr 3(3):266–285
Benatar JR, Sidhu K, Stewart RA (2013) Effects of high and low fat dairy food on cardio-metabolic risk factors: a meta-analysis of randomized studies. PLoS One 8(10):e76480
Ralston RA, Lee JH, Truby H, Palermo CE, Walker KZ (2012) A systematic review and meta-analysis of elevated blood pressure and consumption of dairy foods. J Hum Hypertens 26(1):3–13
Soedamah-Muthu SS, Verberne LD, Ding EL, Engberink MF, Geleijnse JM (2012) Dairy consumption and incidence of hypertension: a dose-response meta-analysis of prospective cohort studies. Hypertension 60(5):1131–1137
Qin LQ, Xu JY, Han SF, Zhang ZL, Zhao YY, Szeto IM (2015) Dairy consumption and risk of cardiovascular disease: an updated meta-analysis of prospective cohort studies. Asia Pac J Clin Nutr 24(1):90–100
Soedamah-Muthu SS, Ding EL, Al-Delaimy WK, Hu FB, Engberink MF, Willett WC, Geleijnse JM (2011) Milk and dairy consumption and incidence of cardiovascular diseases and all-cause mortality: dose-response meta-analysis of prospective cohort studies. Am J Clin Nutr 93(1):158–171
Hu D, Huang J, Wang Y, Zhang D, Qu Y (2014) Dairy foods and risk of stroke: a meta-analysis of prospective cohort studies. Nutr Metab Cardiovasc Dis 24(5):460–469
Perk J, De Backer G, Gohlke H, Graham I, Reiner Z, Verschuren M, Albus C, Benlian P, Boysen G, Cifkova R, Deaton C, Ebrahim S, Fisher M, Germano G, Hobbs R, Hoes A, Karadeniz S, Mezzani A, Prescott E, Ryden L, Scherer M, Syvänne M, Scholte Op Reimer WJ, Vrints C, Wood D, Zamorano JL, Zannad F (2012) European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur Heart J 33(13):1635–1701
Eckel RH, Jakicic JM, Ard JD, de Jesus JM, Houston Miller N, Hubbard VS, Lee IM, Lichtenstein AH, Loria CM, Millen BE, Nonas CA, Sacks FM, Smith SC, Svetkey LP, Wadden TA, Yanovski SZ, Kendall KA, Morgan LC, Trisolini MG, Velasco G, Wnek J, Anderson JL, Halperin JL, Albert NM, Bozkurt B, Brindis RG, Curtis LH, DeMets D, Hochman JS, Kovacs RJ, Ohman EM, Pressler SJ, Sellke FW, Shen WK, Smith SC, Tomaselli GF (2014) 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 129(25 (Suppl 2)):S76–S99
de Oliveira Otto MC, Mozaffarian D, Kromhout D, Bertoni AG, Sibley CT, Jacobs DR, Nettleton JA (2012) Dietary intake of saturated fat by food source and incident cardiovascular disease: the Multi-Ethnic Study of Atherosclerosis. Am J Clin Nutr 96(2):397–404
Chowdhury R, Warnakula S, Kunutsor S, Crowe F, Ward HA, Johnson L, Franco OH, Butterworth AS, Forouhi NG, Thompson SG, Khaw KT, Mozaffarian D, Danesh J (2014) Di Angelantonio E (2014) Association of dietary, circulating, and supplement fatty acids with coronary risk: a systematic review and meta-analysis. Ann Intern Med 160(6):398–406
Bolland MJ, Avenell A, Baron JA, Grey A, MacLennan GS, Gamble GD, Reid IR (2010) Effect of calcium supplements on risk of myocardial infarction and cardiovascular events: meta-analysis. BMJ 341:c3691. doi:10.1136/bmj.c3691
Kanis JA, Johansson H, Oden A, De Laet C, Johnell O, Eisman JA, Mc Closkey E, Mellstrom D, Pols H, Reeve J, Silman A, Tenenhouse A (2005) A meta-analysis of milk intake and fracture risk: low utility for case finding. Osteoporosis Int 16(7):799–804
Owusu W, Willett WC, Feskanich D, Ascherio A, Spiegelman D, Colditz GA (1997) Calcium intake and the incidence of forearm and hip fractures among men. J Nutr 127(9):1782–1787
Cumming RG, Cummings SR, Nevitt MC, Scott J, Ensrud KE, Vogt TM, Fox K (1997) Calcium intake and fracture risk: results from the study of osteoporotic fractures. Am J Epidemiol 145(10):926–934
Meyer HE, Pedersen JI, Løken EB, Tverdal A (1997) Dietary factors and the incidence of hip fracture in middle-aged Norwegians. A prospective study. Am J Epidemiol 145(2):117–123
Fujiwara S, Kasagi F, Yamada M, Kodama K (1997) Risk factors for hip fracture in a Japanese cohort. J Bone Miner Res 12(7):998–1004
Feskanich D, Bischoff-Ferrari HA, Frazier AL, Willett WC (2014) Milk consumption during teenage years and risk of hip fractures in older adults. JAMA pediatrics 168(1):54–60. doi:10.1001/jamapediatrics.2013.3821
Feart C, Lorrain S, Ginder Coupez V, Samieri C, Letenneur L, Paineau D, Barberger-Gateau P (2013) Adherence to a Mediterranean diet and risk of fractures in French older persons. Osteoporos Int 24(12):3031–3041
Benetou V, Orfanos P, Zylis D, Sieri S, Contiero P, Tumino R, Giurdanella MC, Peeters PH, Linseisen J, Nieters A, Boeing H, Weikert C, Pettersson U, Johansson I, Bueno-de-Mesquita HB, Dorronsoro M, Boffetta P, Trichopoulou A (2011) Diet and hip fractures among elderly Europeans in the EPIC cohort. Eur J Clin Nutr 65(1):132–139
Díez-Pérez A, González-Macías J, Marín F, Abizanda M, Alvarez R, Gimeno A, Pegenaute E, Vila J (2007) Prediction of absolute risk of non-spinal fractures using clinical risk factors and heel quantitative ultrasound. Osteoporos Int 18(5):629–639
Roy DK, O’Neill TW, Finn JD, Lunt M, Silman AJ, Felsenberg D, Armbrecht G, Banzer D, Benevolenskaya LI, Bhalla A, Bruges Armas J, Cannata JB, Cooper C, Dequeker J, Diaz MN, Eastell R, Yershova OB, Felsch B, Gowin W, Havelka S, Hoszowski K, Ismail AA, Jajic I, Janott I, Johnell O, Kanis JA, Kragl G, Lopez Vaz A, Lorenc R, Lyritis G, Masaryk P, Matthis C, Miazgowski T, Gennari C, Pols HA, Poor G, Raspe HH, Reid DM, Reisinger W, Scheidt-Nave C, Stepan JJ, Todd CJ, Weber K, Woolf AD, Reeve J (2003) Determinants of incident vertebral fracture in men and women: results from the European Prospective Osteoporosis Study (EPOS). Osteoporos Int 14(1):19–26. doi:10.1007/s00198-002-1317-8
Looker AC, Harris TB, Madans JH, Sempos CT (1993) Dietary calcium and hip fracture risk: the NHANES I Epidemiologic Follow-Up Study. Osteoporos Int 3(4):177–184
Acknowledgments
Writing and editorial assistance were provided by Dr. Kim Russell (PhD) and Dr. Lisa Buttle (PhD) of Chill Pill Media LLP, whose contribution was covered by a grant from the Danone Company and from the Centre National de l’Economie Laitière (CNIEL).
Author Contribution
SR, OB, CC, RR and JYR originated the project and defined the methodology. JJB, PB, MLB, EG, SG and JMK identified and selected the sections to be covered. JJB, OB, RR and JYR were responsible for the literature search. SR, JJB, OB, PB, MLB, CC, JPD, EG, SG and JMK were involved in the selection of the articles eventually analysed and quoted. SR, JJB, OB, PB, JPD, EG, SG, JMK and JYR participated in the first two face-to-face experts consensus meetings. SR, JJB, OB, PB, MLB, CC, JPD, EG, SG and JMK participated in the third face-to-face experts consensus meeting. SR, JJB, OB, MLB, JPD, RR and JYR were responsible for the drafting of the first version. SR, JJB, OB, PB, MLB, CC, JPD, EG, SG and JMK reviewed the first draft of the manuscript after it was edited and corrected by the medical writers. JJB, OB, RR and JYR were responsible to finalize, with the help of the medical writers, the final version of the manuscript which was approved by all co-authors. Drs. Russell and Buttle were of major help in preparing the final version of the manuscript. All authors agree to be accountable for the work and to ensure that any questions relating to the accuracy and integrity of the paper are investigated and properly resolved.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
SR has, or has had, during the last 3 years grants or honoraria as consultant or speaker bureau from the following pharmaceutical companies: Abbott, MSD, Amgen, Will Pharma, Pfizer. JJB declares Danone Institute (Belgium) (Section 2) and Amgen and Bayer (Section 3). OB reports personal fees from Bayer, grants from Genevrier, grants and personal fees from IBSA, grants from MSD, grants from Novartis, grants from Nutraveris, grants from Pfizer, grants and personal fees from Rottapharm, grants and personal fees from Servier, grants and personal fees from SMB, grants from Theramex, outside the submitted work. PB has no conflict of interest. MLB has received board membership from Alexion and Servier. She has received consultancy fees from Servier, MSD, Amgen, NPS, SPA, Abiogen, Alexion and Shire. She has received grants from Servier, MSD, Eli Lilly, Amgen, NPS, SPA, Abiogen, Bruno Farmaceutici, Alexion and Shire. She has received payment for lectures including service on speakers’ bureaux from NPS and payment for manuscript preparation from Bruno Farmaceutici. CC has received consultancy, lecture fees and honoraria from AMGEN, GSK, Alliance for Better Bone Health, MSD, Eli Lilly, Pfizer, Novartis, Servier, Merck, Medtronic and Roche. JPD has no conflict of interest. EG has no conflict of interest. SG has received grants and/or fees from Merck Sharp & Dohme, support for congress preparation from Novartis, grants, fees and non-financial support (support for congress preparation) from Amgen, fees from Takeda and and fees and non-financial support (support for congress preparation) from Madaus Rottapharma. JMK has no conflict of interest. RR has received fees from Amgen, Danone, GSK, MSD and Servier. JYR has received grants from Bristol Myers Squibb, Merck Sharp & Dohme, Rottapharm, Teva, Lilly, Novartis, Roche, GlaxoSmithKline, Amgen and Servier. He has received lecture fees from Merck Sharp & Dohme, Lilly, Rottapharm, IBSA, Genevrier, Novartis, Servier, Roche, GlaxoSmithKline, Teijin, Teva, Ebewee Pharma, Zodiac, Analuis, Theramex, Nycomed, Novo-Nordisk and Nolver.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Rozenberg, S., Body, JJ., Bruyère, O. et al. Effects of Dairy Products Consumption on Health: Benefits and Beliefs—A Commentary from the Belgian Bone Club and the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases. Calcif Tissue Int 98, 1–17 (2016). https://doi.org/10.1007/s00223-015-0062-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-015-0062-x