Abstract
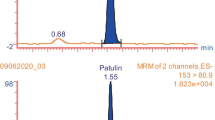
The advantages of using smart materials as immunosorbents in the analysis of complex matrices by ion mobility spectrometry (IMS) have been highlighted in this study. A novel analytical method has been proposed for the sensitive, selective, and fast determination of residues of the plant growth regulator forchlorfenuron in fruit juices. Three different monoclonal antibodies (s3#22, p2#21, and p6#41) were employed for the production of immunosorbents, based on Sepharose gel beads, which were characterized in terms of loading capacity, solvent resistance, and repeatability for its use in solid-phase extraction (SPE). Immunosorbents that were prepared with antibody p6#44 provided the best performance, with a loading capacity of 0.97 μg, a 10% (v/v) 2-propanol tolerance, and a reusability of at least eight uses. The SPE procedure involved the use of a column with 0.15 g Sepharose beads, containing 0.5 mg antibody, which was loaded to 20 mL of the sample, washed with 2 mL of water plus 2 mL of 10% (v/v) 2-propanol, and eluted with 2 mL of 2-propanol. The cleaned extract was directly analyzed by IMS, giving a limit of detection of 2 μg L−1 with a relative standard deviation of 7.6%. Trueness was assessed by the analysis of blank grape and kiwifruit juice samples spiked with forchlorfenuron concentrations from 10 to 400 μg L−1, with recoveries from 80 to 115%. The analytical performance of the proposed immunosorbent was compared with conventional extraction and cleanup methods, such as QuEChERS and C18-based SPE, giving the cleanest extracts for accurate determinations of forchlorfenuron by IMS.

ᅟ



Similar content being viewed by others
References
Reynolds AG, Wardle DA, Zusowski C, Looney NF. Phenyl ureas forchlorfenuron and thiadiazuron affect yield components, fruit composition and storage potential of four seedless grape selections. J Am Soc Hortic Sci. 1992;117:85–6.
Sharma D, Awasthi MD. Behaviour of forchlorfenuron residues in grape, soil and water. Chemosphere. 2003;50:589–94.
European Food Safety Authority. Peer review of the pesticide risk assessment of the active substance forchlorfenuron. EFSA J. 2017;15:4874.
U.S. Environmental Protection Agency. Pesticide fact sheet, forchlorfenuron. 2004.
Commission Regulation (EU) No 398/2014 of 22 April 2014 amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for benthiavalicarb, cyazofamid, cyhalofop-butyl, forchlorfenuron, pymetrozine and silthiofam in or on certain products. Off J Eur Union L 23.4.2014 119/3.
U.S. Code of Federal Regulations, Title 40—protection of environment, part 180—tolerances and exemptions for pesticide chemical residues in food, 180.569 forchlorfenuron; tolerances for residues.
Liu S, He H, Huang X, Jin Q, Zhu G. Comparison of extraction solvents and sorbents in the quick, easy, cheap, effective, rugged, and safe method for the determination of pesticide multiresidue in fruits by ultra high performance liquid chromatography with tandem mass spectrometry. J Sep Sci. 2015;38:3525–32.
Yan Z, Nie JY, Xu GF, Li HF, Li J, Li ZX, et al. Simultaneous determination of plant growth regulators in fruits using a modified QuEChERS procedure and UPLC–MS/MS. Hortic Plat J. 2016;2(4):203–8.
Suárez-Pantaleón C, Esteve-Turrillas FA, Mercader JV, Agulló C, Abad-Somovilla A, Abad-Fuentes A. Development and validation of a direct competitive monoclonal antibody-based immunoassay for the sensitive and selective analysis of the phytoregulator forchlorfenuron. Anal Bioanal Chem. 2012;403:2019–26.
Suárez-Pantaleón C, Wichers J, Abad-Somovilla A, Van Amerongen A, Abad-Fuentes A. Development of an immunochromatographic assay based on carbon nanoparticles for the determination of the phytoregulator forchlorfenuron. Biosens Bioelectron. 2013;42:170–6.
Xu Q, Cai L, Zhao H, Tang J, Shen Y, Hu X, et al. Forchlorfenuron detection based on its inhibitory effect towards catalase immobilized on boron nitride substrate. Biosens Bioelectron. 2015;63:294–300.
Chen X, Yan K, Xiao X, Li G. Analysis of forchlorfenuron and thidiazuron in fruits and vegetables by surface. J Sep Sci. 2016;39:2340–6.
Hu JY, Li JZ. Determination of forchlorfenuron residues in watermelon by solid-phase extraction and high-performance liquid chromatography. J AOAC Int. 2006;89(6):1635–40.
Wang F, Zhang WF, Chen YL, Xu GG, Deng ZF, Du HF, et al. Simultaneous determination of six plant growth regulators in fruits using high performance liquid chromatography based on solid phase extraction and cleanup with a novel mixed-mode functionalized calixarene sorbent. Anal Methods. 2015;7(15):6365–71.
Cheng Y, Nie J, Li Z, Yan Z, Xu G, Li H, et al. A molecularly imprinted polymer synthesized using β-cyclodextrin as the monomer for the efficient recognition of forchlorfenuron in fruits. Anal Bioanal Chem. 2017;409(21):5065–72.
Eiceman GA, Stone JA. Peer reviewed: ion mobility spectrometers in national defense. New uses of previously unheralded analytical instruments. Anal Chem. 2004;76:390A–7A.
Szymańska E, Davies AN, Buydens LMC. Chemometrics for ion mobility spectrometry data: recent advances and future prospects. Analyst. 2016;141:5689–708.
Płotka-Wasylka J, Szczepanska N, de la Guardia M, Namiesnik J. Modern trends in solid phase extraction: new sorbent media. TRAC Trends Anal Chem. 2016;77:23–43.
Pichon V, Bouzige M, Mieége CC, Hennion MC. Immunosorbents: natural molecular recognition materials for sample preparation of complex environmental matrices. TRAC Trends Anal Chem. 1999;18:219–35.
Senyuva HZ, Gilbert J. Immunoaffinity column clean-up techniques in food analysis: a review. J Chromatogr B. 2010;878:115–32.
Martín-Esteban A. Molecularly-imprinted polymers as a versatile, highly selective tool in sample preparation. TrAC Trends Anal Chem. 2013;45:169–81.
Du F, Guo L, Qin Q, Zheng X, Ruan G, Li J, et al. Recent advances in aptamer-functionalized materials in sample preparation. TRAC Trends Anal Chem. 2015;67:134–46.
Rocío-Bautista P, González-Hernández P, Pino V, Pas J, Afonso AM. Metal-organic frameworks as novel sorbents in dispersive-based microextraction approaches. TRAC Trends Anal Chem. 2017;90:114–34.
Wang C, Li M, Xu H, Wei Y. Preparation of an internal surface reversed-phase restricted-access material for the analysis of hydrophobic molecules in biological matrices. J Chromatogr A. 2014;1343:195–9.
Khalesi M, Sheikh-Zeinoddin M, Tabrizchi M. Determination of ochratoxin A in licorice root using inverse ion mobility spectrometry. Talanta. 2011;83:988–93.
Armenta S, de la Guardia M, Abad-Fuentes A, Abad-Somovilla A, Esteve-Turrillas FA. Off-line coupling of multidimensional immunoaffinity chromatography and ion mobility spectrometry: a promising partnership. J Chromatogr A. 2015;1426:110–7.
Armenta S, de la Guardia M, Abad-Fuentes A, Abad-Somovilla A, Esteve-Turrillas FA. Highly selective solid-phase extraction sorbents for chloramphenicol determination in food and urine by ion mobility spectrometry. Anal Bioanal Chem. 2016;408:8559–67.
Esteve-Turrillas FA, Mercader JV, Agulló C, Abad-Somovilla A, Abad-Fuentes A. Development of immunoaffinity columns for pyraclostrobin extraction from fruit juices and analysis by liquid chromatography with UV detection. J Chromatogr A. 2011;1218:4902–9.
Suárez-Pantaleón C, Mercader JV, Agulló C, Abad-Somovilla A, Abad-Fuentes A. Production and characterization of monoclonal and polyclonal antibodies to forchlorfenuron. J Agric Food Chem. 2008;56:11122–31.
Suárez-Pantaleón C, Mercader JV, Agulló C, Abad-Somovilla A, Abad-Fuentes A. Forchlorfenuron-mimicking haptens: from immunogen design to antibody characterization by hierarchical clustering analysis. Org Biomol Chem. 2011;9:4863–72.
Valverde A, Aguilera A, Ferrer C, Camacho F, Cammarano A. Analysis of forchlorfenuron in vegetables by LC/TOF-MS after extraction with the buffered QuEChERS method. J Agric Food Chem. 2010;58:2818–23.
Delaunay N, Pichon V, Hennion MC. Immunoaffinity solid-phase extraction for the trace-analysis of low-molecular-mass analytes in complex sample matrices. J Chromatogr B. 2000;745:15–37.
Funding
The authors gratefully acknowledge the financial support of the Ministerio de Economía y Competitividad (AGL2012-39965-C02-01-02/ALI and CTQ-2014-52841-P) and Generalitat Valenciana (PROMETEO-II 2014-077).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Orellana-Silla, A., Armenta, S., de la Guardia, M. et al. Development of immunosorbents for the analysis of forchlorfenuron in fruit juices by ion mobility spectrometry. Anal Bioanal Chem 410, 5961–5967 (2018). https://doi.org/10.1007/s00216-018-1213-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-1213-5