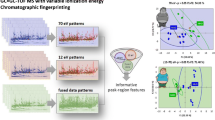
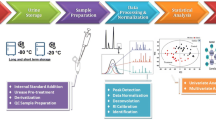
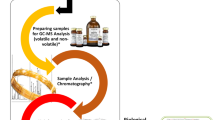
Abstract
This study exploits the information potential of comprehensive two-dimensional gas chromatography configured with a parallel dual secondary column-dual detection by mass spectrometry and flame ionization (GC×2GC-MS/FID) to study changes in urinary metabolic signatures of mice subjected to high-fructose diets. Samples are taken from mice fed with normal or fructose-enriched diets provided either in aqueous solution or in solid form and analyzed at three stages of the dietary intervention (1, 6, and 12 weeks). Automated Untargeted and Targeted fingerprinting for 2D data elaboration is adopted for the most inclusive data mining of GC×GC patterns. The UT fingerprinting strategy performs a fully automated peak-region features fingerprinting and combines results from pre-targeted compounds and unknowns across the sample-set. The most informative metabolites, with statistically relevant differences between sample groups, are obtained by unsupervised multivariate analysis (MVA) and cross-validated by multi-factor analysis (MFA) with external standard quantitation by GC-MS. Results indicate coherent clustering of mice urine signatures according to dietary manipulation. Notably, the metabolite fingerprints of mice fed with liquid fructose exhibited greater derangement in fructose, glucose, citric, pyruvic, malic, malonic, gluconic, cis-aconitic, succinic and 2-keto glutaric acids, glycine acyl derivatives (N-carboxy glycine, N-butyrylglycine, N-isovaleroylglycine, N-phenylacetylglycine), and hippuric acid. Untargeted fingerprinting indicates some analytes which were not a priori pre-targeted which provide additional insights: N-acetyl glucosamine, N-acetyl glutamine, malonyl glycine, methyl malonyl glycine, and glutaric acid. Visual features fingerprinting is used to track individual variations during experiments, thereby extending the panorama of possible data elaboration tools.

ᅟ







Similar content being viewed by others
References
Malik VS, Hu FB. Fructose and cardiometabolic health what the evidence from sugar-sweetened beverages tells us. J Am Coll Cardiol. 2015;66:1615–24. https://doi.org/10.1016/j.jacc.2015.08.025.
Rizkalla SW. Health implications of fructose consumption: a review of recent data.(review)(report). Nutr Metab (Lond). 2010;7:82. https://doi.org/10.1186/1743-7075-7-82.
Lim JS, Mietus-Snyder M, Valente A, Schwarz J-M, Lustig RH. The role of fructose in the pathogenesis of NAFLD and the metabolic syndrome. Nat Rev Gastroenterol Hepatol. 2010;7:251–64.
Teff KL, Grudziak J, Townsend RR, Dunn TN, Grant RW, Adams SH, et al. Endocrine and metabolic effects of consuming fructose- and glucose-sweetened beverages with meals in obese men and women: influence of insulin resistance on plasma triglyceride responses. J Clin Endocrinol Metab. 2009;94:1562–9. https://doi.org/10.1210/jc.2008-2192.
Lê KA, Ith M, Kreis R, Faeh D, Bortolotti M, Tran C, et al. Fructose overconsumption causes dyslipidemia and ectopic lipid deposition in healthy subjects with and without a family history of type 2 diabetes. Am J Clin Nutr. 2009;89:1760–5. https://doi.org/10.3945/ajcn.2008.27336.
Schwarz JM, Noworolski SM, Wen MJ, Dyachenko A, Prior JL, Weinberg ME, et al. Effect of a high-fructose weight-maintaining diet on lipogenesis and liver fat. J Clin Endocrinol Metab. 2015;100:2434–42. https://doi.org/10.1210/jc.2014-3678.
Ritze Y, Bárdos G, D’Haese JG, Ernst B, Thurnheer M, Schultes B, et al. Effect of high sugar intake on glucose transporter and weight regulating hormones in mice and humans. PLoS One. 2014;9:1–9. https://doi.org/10.1371/journal.pone.0101702.
Mastrocola R, Ferrocino I, Liberto E, Chiazza F, Cento AS, Querio G., Nigro D, Bitonto V, Cutrin JC, Rantsiou K, Durante M, Masini E, Aragno M, Cordero C, Cocolin L CM. Fructose liquid and solid formulations differently affect gut integrity, microbiota composition and related liver toxicity: a comparative in vivo study. J Biochem Nutr. https://doi.org/10.1016/j.jnutbio.2018.02.003.
Hunter P. Reading the metabolic fine print. EMBO Rep. 2009;10:20–3. https://doi.org/10.1038/embor.2008.236.
Almstetter MF, Oefner PJ, Dettmer K. Comprehensive two-dimensional gas chromatography in metabolomics. Anal Bioanal Chem. 2012;402:1993–2013. https://doi.org/10.1007/s00216-011-5630-y.
Reichenbach SE, Tian X, Tao Q, Ledford EB, Wu Z, Fiehn O. Informatics for cross-sample analysis with comprehensive two-dimensional gas chromatography and high-resolution mass spectrometry (GCxGC-HRMS). Talanta. 2011;83:1279–88. https://doi.org/10.1016/j.talanta.2010.09.057.
Koek MM, Muilwijk B, van Stee LLP, Hankemeier T. Higher mass loadability in comprehensive two-dimensional gas chromatography-mass spectrometry for improved analytical performance in metabolomics analysis. J Chromatogr A. 2008;1186:420–9. https://doi.org/10.1016/j.chroma.2007.11.107.
Nicolotti L, Cordero C, Bressanello D, Cagliero C, Liberto E, Magagna F, et al. Parallel dual secondary column-dual detection: a further way of enhancing the informative potential of two-dimensional comprehensive gas chromatography. J Chromatogr A. 2014;1360:264–74. https://doi.org/10.1016/j.chroma.2014.07.081.
Reichenbach SE, Rempe DW, Tao Q, Bressanello D, Liberto E, Bicchi C, et al. Alignment for comprehensive two-dimensional gas chromatography with dual secondary columns and detectors. Anal Chem. 2015;87:10056–63. https://doi.org/10.1021/acs.analchem.5b02718.
Sgorbini B, Cagliero C, Boggia L, Liberto E, Reichenbach SE, Rubiolo P, et al. Parallel dual secondary-column-dual detection comprehensive two-dimensional gas chromatography: a flexible and reliable analytical tool for essential oils quantitative profiling. Flavour Fragr J. 2015;30:366–80. https://doi.org/10.1002/ffj.3255.
Cordero C, Rubiolo P, Cobelli L, Stani G, Miliazza A, Giardina M, et al. Potential of the reversed-inject differential flow modulator for comprehensive two-dimensional gas chromatography in the quantitative profiling and fingerprinting of essential oils of different complexity. J Chromatogr A. 2015;1417:79–95. https://doi.org/10.1016/j.chroma.2015.09.027.
Cordero C, Rubiolo P, Reichenbach SE, Carretta A, Cobelli L, Giardina M, et al. Method translation and full metadata transfer from thermal to differential flow modulated comprehensive two dimensional gas chromatography: profiling of suspected fragrance allergens. J Chromatogr A. 2016; https://doi.org/10.1016/j.chroma.2016.12.011.
Magagna F, Valverde-Som L, Ruíz-Samblás C, Cuadros-Rodríguez L, Reichenbach SE, Bicchi C, et al. Combined untargeted and targeted fingerprinting with comprehensive two-dimensional chromatography for volatiles and ripening indicators in olive oil. Anal Chim Acta. 2016;936:245–58. https://doi.org/10.1016/j.aca.2016.07.005.
Zhang Q, Wang G, Du Y, Zhu L, A J. GC/MS analysis of the rat urine for metabonomic research. J Chromatogr B 2007;854:20–5. https://doi.org/10.1016/j.jchromb.2007.03.048.
Reichenbach SE, Carr PW, Stoll DR, Tao Q. Smart templates for peak pattern matching with comprehensive two-dimensional liquid chromatography. J Chromatogr A. 2009;1216:3458–66. https://doi.org/10.1016/j.chroma.2008.09.058.
Sgorbini B, Cagliero C, Boggia L, Liberto E, Reichenbach SE, Rubiolo P, Cordero C, Bicchi C. Parallel dual secondary-column-dual detection comprehensive two-dimensional gas chromatography: a flexible and reliable analytical tool for essential oils quantitative profiling. Flavour Fragr J. 2015; https://doi.org/10.1002/ffj.3255.
Reichenbach SE, Tian X, Boateng AA, Mullen CA, Cordero C, Tao Q. Reliable peak selection for multisample analysis with comprehensive two-dimensional chromatography. Anal Chem. 2013;85:4974–81. https://doi.org/10.1021/ac303773v.
Reichenbach SE, Tian X, Cordero C, Tao Q. Features for non-targeted cross-sample analysis with comprehensive two-dimensional chromatography. J Chromatogr A. 2012;1226:140–8. https://doi.org/10.1016/j.chroma.2011.07.046.
Bressanello D, Liberto E, Collino M, Reichenbach SE, Benetti E, Chiazza F, et al. Urinary metabolic fingerprinting of mice with diet-induced metabolic derangements by parallel dual secondary column-dual detection two-dimensional comprehensive gas chromatography. J Chromatogr A. 2014;1361:265–76. https://doi.org/10.1016/j.chroma.2014.08.015.
Hollingsworth B V., Reichenbach SE, Tao Q, Visvanathan A. Comparative visualization for comprehensive two-dimensional gas chromatography. 2006; 51–58.
Magnusson B, Örnemark U. (eds.) Eurachem Guide: The Fitness for Purpose of Analytical Methods – A Laboratory Guide to Method Validation and Related Topics, (2nd ed. 2014). Available from www.eurachem.org. Last accessed February the 1st, 2018.
Barwick V. (Ed), Eurachem/CITAC Guide: Guide to Quality in Analytical Chemistry: An Aid to Accreditation (3 rd ed. 2016). Available from www.eurachem.org. Last accessed on February the 1st, 2018.
Commission E. Commission Decision 2002/657/EC implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Off J Eur Union. 2002;L221:8–36.
Zhao X, Han Q, Liu Y, Sun C, Gang X, Wang G. The relationship between branched-chain amino acid related metabolomic signature and insulin resistance: a systematic review. J Diabetes Res. 2016; https://doi.org/10.1155/2016/2794591.
Fontana L, Cummings NE, Apelo SIA, Joshua C, Kasza I, Schmidt BA, et al. HHS public access. Cell Rep. 2016;16:520–30. https://doi.org/10.1016/j.celrep.2016.05.092.Decreased.
Tulipani S, Palau-Rodriguez M, Miñarro Alonso A, Cardona F, Marco-Ramell A, Zonja B, et al. Biomarkers of morbid obesity and prediabetes by metabolomic profiling of human discordant phenotypes. Clin Chim Acta. 2016;463:53–61. https://doi.org/10.1016/j.cca.2016.10.005.
McCarty MF, DiNicolantonio JJ. The cardiometabolic benefits of glycine: is glycine an “antidote” to dietary fructose? Open Heart. 2014;1:e000103. https://doi.org/10.1136/openhrt-2014-000103.
Cordero C, Zebelo SA, Gnavi G, Griglione A, Bicchi C, Maffei ME, et al. HS-SPME-GC×GC-qMS volatile metabolite profiling of Chrysolina herbacea frass and Mentha spp. leaves. Anal Bioanal Chem. 2012;402:1941–52. https://doi.org/10.1007/s00216-011-5600-4.
Cordero C, Liberto E, Bicchi C, Rubiolo P, Reichenbach SE, Tian X, et al. Targeted and non-targeted approaches for complex natural sample profiling by GCxGC-qMS. J Chromatogr Sci. 2010;48:251–61. https://doi.org/10.1093/chromsci/48.4.251.
Purcaro G, Cordero C, Liberto E, Bicchi C, Conte LS. Toward a definition of blueprint of virgin olive oil by comprehensive two-dimensional gas chromatography. J Chromatogr A. 2014;1334:101–11. https://doi.org/10.1016/j.chroma.2014.01.067.
Acknowledgements
Authors are indebted with Dr. Martina Carbone for the technical help.
Funding
This work was supported and funded by the University of Turin (Ricerca Locale 2016 and 2017), Regione Toscana (Bando Nutraceutica 2014), Project TAGIDISFRU, ERA-HDHL-ERANET Biomarkers for Nutrition and Health Implementing the JPG HDHL objectives”—Project “SALIVAGES.”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethics approval
The research was conducted in accordance with the European Directive 2010/63/EU on the protection of animals used for scientific purposes as well as the International Guiding Principles for Biomedical Research Involving Animals, issued by the Council for the International Organizations of Medical Sciences. Both the Turin University Ethics Committee (Prot. 6437 22/02/2016) and the Italian Ministry of Health (Prot. 42/2017-PR) approved the experimental protocol. All efforts were made to minimize animal suffering and to reduce the number of animals used.
Electronic supplementary material
ESM 1
(PDF 554 kb)
Rights and permissions
About this article
Cite this article
Bressanello, D., Liberto, E., Collino, M. et al. Combined untargeted and targeted fingerprinting by comprehensive two-dimensional gas chromatography: revealing fructose-induced changes in mice urinary metabolic signatures. Anal Bioanal Chem 410, 2723–2737 (2018). https://doi.org/10.1007/s00216-018-0950-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-0950-9