Abstract
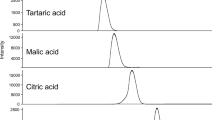
The analysis of flavonoids in unifloral honeys by high-performance liquid chromatography (HPLC) coupled with coulometric electrode array detection (CEAD) is described. The compounds were extracted by a nonionic polymeric resin (Amberlite XAD-2) and then separated on a reversed phase column using gradient elution. Quercetin, naringenin, hesperetin, luteolin, kaempferol, isorhamnetin, and galangin were detected in a coulometric electrode array detection system between +300 and +800 mV against palladium reference electrodes, and their presence was additionally confirmed by HPLC coupled with electrospray ionization mass spectrometry. The method was applied to analysis of 19 honeys of different varieties and origin. The limits of detection and quantitation ranged between 1.6 and 8.3 μg/kg and 3.9 and 27.4 μg/kg, respectively. The recoveries were above 96% in fluid and above 89% in creamy honeys. Some of these honeys (melon, pumpkin, cherry blossom, dandelion, maple, and pine tree honey) were investigated for their flavonoid content and profile for the first time. Differences between honeys were observed both in flavonoid concentrations and in the flavonoid profiles. The flavonoid concentrations ranged from 0.015 to 3.4 mg/kg honey. Galangin, kaempferol, quercetin, isorhamnetin, and luteolin were detected in all investigated honeys, whereas hesperetin occurred only in lemon and orange honeys and naringenin in lemon, orange, rhododendron, rosemary, and cherry blossom honeys.

Electrode array detection



Similar content being viewed by others
References
Amiot MJ, Aubert S, Gonnet M, Tacchini M (1989) Apidologie 20:115–125
Trautvetter S, Koelling-Speer I, Speer K (2009) Apidologie 40:140–150
Pichichero E, Canuti L, Canini A (2009) J Sci Food Agric 89:609–616
Pulcini P, Allegrini F, Festuccia N (2006) Apiacta 41:21–27
Michalkiewicz A, Biesaga M, Pyrzynska K (2008) J Chromatogr A 1187:18–24
Gheldof N, Wang X-H, Engeseth NJ (2002) J Agric Food Chem 50:5870–5877
Yao L, Jiang Y, D’Arcy B, Singanusong R, Datta N, Caffin N, Raymont K (2004) J Agric Food Chem 52:210–214
Ferreres F, Garcia-Viguera C, Tomás-Lorente F, Tomás-Barberán FA (1993) J Sci Food Agric 61:121–123
Gil MI, Ferreres F, Ortiz A, Subra E, Tomás-Barberán FA (1995) J Agric Food Chem 43:2833–2838
Martos I, Cossentini M, Ferreres F, Tomás-Barberán FA (1997) J Agric Food Chem 45:2824–2829
Martos I, Ferreres F, Yao L, D’Arcy B, Caffin N, Tomás-Barberán FA (2000) J Agric Food Chem 48:4744–4748
Tomás-Barberán FA, Martos I, Ferreres F, Radovic BS, Anklam E (2001) J Sci Food Agric 81:485–496
Iurlina MO, Saiz AI, Fritz R, Manrique GD (2009) Food Chem 115:1141–1149
Weston RJ, Mitchell KR, Allen KL (1999) Food Chem 64:295–301
Ferreres F, Tomás-Barberán FA, Gil MI, Tomás-Lorente F, Tomás-Lorente F (1991) J Sci Food Agric 56:49–56
Soler C, Gil MI, Garcia-Viguera C, Tomás-Barberán FA (1995) Apidologie 26:53–60
Weston RJ, Brocklebank LK, Lu Y (2000) Food Chem 70:427–435
Vit P, Soler C, Tomás-Barberán FA (1997) Z Lebensm Unters Forsch A 204:43–47
Biesaga M, Pyrzynska K (2009) J Chromatogr A 1216:6620–6626
Liang Y, Cao W, Chen W-J, Xiao X-H, Zheng J-B (2009) Food Chem 114:1537–1541
Truchado P, Ferreres F, Tomás-Barberán FA (2009) J Chromatogr A 1216:7241–7248
Truchado P, Ferreres F, Bortolotti L, Sabatini AG, Tomás-Barberán FA (2008) J Agric Food Chem 56:8815–8824
Berahia T, Cerrati C, Sabatier S, Amiot M (1993) Sciences des Aliments 13:15–24
Sabatier S, Amiot MJ, Tacchini M, Aubert S (1992) J Food Sci 57:773–774, 777
Delgado C, Tomás-Barberán FA, Talou T, Gaset A (1994) Chromatographia 38:71–78
Andrade P, Ferreres F, Gil MI, Tomás-Barberán FA (1997) Food Chem 60:79–84
Gómez-Caravaca AM, Segura-Carretera A, Fernandez-Gutiérrez A (2006) Agro Food Industry Hi-Tech 17:68–72
Arraez-Román D, Gómez-Caravaca AM, Gómez-Romero M, Segura-Carretero A, Fernandez-Guitiérrez A (2006) J Pharmaceut Biomed Analysis 41:1648–1656
Volpi N (2004) Electrophoresis 25:1872–1878
Kenjeric D, Mandic ML, Primorac L, Cacic F (2008) Food Chem 110:187–192
Ferreres F, Tomás-Barberán FA, Soler C, Garcia-Viguera C, Ortiz A, Tomás-Lorente F (1994) Apidologie 25:21–30
Yao L, Datta FA, Tomás-Barberán FA, Ferreres F, Martos I, Singanusong R (2004) Food Chem 81:159–168
Yao L, Jiang Y, Singanusong R, D’Arcy B, Datta N, Caffin N, Raymont K (2004) Food Res Int 37:166–174
Kenjeric D, Mandic ML, Primorac L, Bubalo D, Perl A (2007) Food Chem 102:683–690
Ferreres F, Juan T, Perez-Arquillue C, Herrera-Marteache A, Garcia-Viguera C, Tomás-Barberán FA (1998) J Sci Food Agric 77:506–510
Čeksterytė V, Kazlauskas S, Racys J (2006) Biologija 2:28–33
Vit P, Tomás-Barberán FA (1998) Z Lebensm Unters Forsch A 206:288–293
Alabdeen Makawi SZ, Gadkariem EA, Ayoub SMH (2009) E-J Chem 6:S429–S437
Fiorani M, Accorsi A, Blasa M, Diamantini G, Piatti E (2006) J Agric Food Chem 54:8328–8334
Martos I, Ferreres F, Tomás-Barberán FA (2000) J Agric Food Chem 48:1498–1502
Baltrušaitytė V, Venskutonis PR, Čeksterytė V (2007) Food Chem 101:502–514
Volpi N, Bergonzini G (2006) J Pharmaceut Biomed Analysis 42:354–361
Inoue K, Murayama S, Seshimo F, Takeba K, Yoshimura Y, Nakazawa H (2005) J Sci Food Agric 85:872–878
Joerg E, Sontag G (1993) J Chromatogr A 635:137–142
Pohl P (2009) TRAC 28:117–128
Cuevas-Glory LF, Pino JA, Santiago LS, Sauri-Duch E (2007) Food Chem 103:1032–1043
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the special issue Analytical Sciences in Austria with Guest Editors G. Allmaier, W. Buchberger, and K. Francesconi.
Rights and permissions
About this article
Cite this article
Petrus, K., Schwartz, H. & Sontag, G. Analysis of flavonoids in honey by HPLC coupled with coulometric electrode array detection and electrospray ionization mass spectrometry. Anal Bioanal Chem 400, 2555–2563 (2011). https://doi.org/10.1007/s00216-010-4614-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-010-4614-7