Abstract
Objective
Cannabinoid receptor agonists such as delta-9-tetrahydrocannabinol (Δ9-THC) enhance the antinociceptive potency of mu opioid receptor agonists such as morphine, indicating that opioid/cannabinoid mixtures might be effective for treating pain. However, such enhancement will be beneficial only if cannabinoids do not also enhance adverse effects of opioids, including those related to abuse. In rhesus monkeys, cannabinoids fail to enhance and often decrease self-administration of the mu opioid receptor agonist heroin, suggesting that opioid/cannabinoid mixtures do not have greater reinforcing effects (abuse potential) compared with opioids alone. Previous studies on the self-administration of opioid/cannabinoid mixtures used single-response procedures, which do not easily differentiate changes in reinforcing effects from other effects (e.g., rate decreasing).
Methods
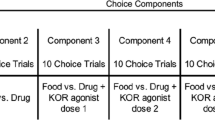
In this study, rhesus monkeys (n = 4) responded under a choice procedure wherein responding on one lever delivered sucrose pellets and responding on the other lever delivered intravenous infusions of the mu opioid receptor agonist remifentanil (0.032–1.0 μg/kg/infusion) alone or in combination with either Δ9-THC (10–100 μg/kg/infusion) or the synthetically derived cannabinoid receptor agonist CP55940 (3.2–10 μg/kg/infusion).
Results
Remifentanil dose-dependently increased choice of drug over food, whether available alone or in combination with a cannabinoid, and the potency of remifentanil was not significantly altered by coadministration with a cannabinoid. Mixtures containing the largest doses of cannabinoids decreased response rates in most subjects, confirming that behaviorally active doses were studied.
Conclusion
Overall, these results extend previous studies to include choice behavior and show that cannabinoids do not substantially enhance the reinforcing effects of mu opioid receptor agonists.


Similar content being viewed by others
References
Abel EL (1975) Cannabis: effects on hunger and thirst. Behav Bio 15:255–281. https://doi.org/10.1016/S0091-6773(75)91684-3
Bachhuber MA, Saloner B, Cunningham CO, Barry CL (2014) Medical cannabis laws and opioid analgesic overdose mortality in the United States, 1999-2010. JAMA Intern Med 174:1668–1673. https://doi.org/10.1001/jamainternmed.2014.4005
Banks ML, Negus SS (2017) Insights from preclinical choice models on treating drug addiction. Trends Pharmacol Sci 38:181–194. https://doi.org/10.1016/j.tips.2016.11.002
Bellocchio L, Lafenêtre P, Cannich A, Cota D, Puente N, Grandes P, Chaouloff F, Piazza PV, Marsicano G (2010) Bimodal control of stimulated food intake by the endocannabinoid system. Nat Neurosci 13:281–283. https://doi.org/10.1038/nn.2494
Boehnke KF, Litinas E, Clauw DJ (2016) Medical cannabis use is associated with decreased opiate medication use in a retrospective cross-sectional survey of patients with chronic pain. J Pain 17:739–774. https://doi.org/10.1016/j.jpain.2016.03.002
Bradford AC, Bradford WD, Abraham A, Bagwell Adams G (2018) Association between US state medical cannabis laws and opioid prescribing in the Medicare part D population. JAMA Intern Med. 178:667-672. https://doi.org/10.1001/jamainternmed.2018.0266
Catania AC (1966) Concurrent operants. In: Honig WK (ed) Operant behavior: areas of research and application, Appleton-Century-Crofts, New York, pp 213–270
Cooper ZD, Bedi G, Ramesh D, Balter R, Comer SD, Haney M (2018) Impact of co-administration of oxycodone and smoked cannabis on analgesia and abuse liability. Neuropsychopharmacology. https://doi.org/10.1038/s41386-018-0011-2 [Epub ahead of print]
Dowell D, Haegerich TM, Chou R (2016) CDC guideline for prescribing opioids for chronic pain—United States. JAMA 315:1624–1645. https://doi.org/10.1001/jama.2016.1464
Elsmore TF, Fletcher GV (1972) Δ9-tetrahydrocannabinol: aversive effects in rat at high doses. Science 175:911–912. https://doi.org/10.1126/science.175.4024.911
Freeman KB, Woolverton WL (2011) Self-administration of cocaine and remifentanil by monkeys: choice between single drugs and mixtures. Psychopharmacology 215:281–290. https://doi.org/10.1007/s00213-010-2131-1
Grotenhermen F (2003) Pharmacokinetics and pharmacodynamics of cannabinoids. Clin Pharmacokinet 42:327–360. https://doi.org/10.2165/00003088-200342040-00003
Hall W, West R, Marsden J, Humphreys K, Neale J, Petry N (2018) It is premature to expand access to medicinal cannabis in hopes of solving the US opioid crisis. Addiction 113:987–988. https://doi.org/10.1111/add.14139
Hayes MJ, Brown MS (2014) Legalization of medical marijuana and incidence of opioid mortality. JAMA Intern Med 174:1673–1674. https://doi.org/10.1001/jamainternmed.2014.2716
Hruba L, McMahon LR (2014) The cannabinoid agonist HU-210: Pseudo-irreversible discriminative stimulus effects in rhesus monkeys. Eur J Pharmacol 727:35-42. https://doi.org/10.1016/j.ejphar.2014.01.041
Katz JL (1989) Drugs as reinforcers: pharmacological and behavioural factors. In: JM Liebman JM, Cooper SJ (eds) The Neuropharmacological basis of reward. Oxford University Press, Oxford, pp 164–213
Kirkham TC, Williams CM (2001) Endogenous cannabinoids and appetite. Nutr Res Rev 14:65–86. https://doi.org/10.1079/NRR200118
Ko MC, Terner J, Hursh S, Woods JH, Winger G (2002) Relative reinforcing effects of three opioids with different durations of action. J Pharmacol Exp Ther 301:698–704. https://doi.org/10.1124/jpet.301.2.698
Koffarnus MN, Woods JH (2008) Quantification of drug choice with the generalized matching law in rhesus monkeys. J Exp Anal Behav 89:209–224. https://doi.org/10.1901/jeab.2008.89-209
Koffarnus MN, Hall A, Winger G (2012) Individual differences in rhesus monkeys’ demand for drugs of abuse. Addict Biol 17:887–896. https://doi.org/10.1111/j.1369-1600.2011.00335.x
Lagorio CH, Winger G (2014) Random-ratio schedules produce greater demand for iv drug administration than fixed-ratio schedules in rhesus monkeys. Psychopharmacology 231:2981–2988. https://doi.org/10.1007/s00213-014-3477-6
Lamb RJ, Ginsburg BC (2018) Addiction as a BAD, a behavioral allocation disorder. Pharmacol Biochem Behav 164:62–70. https://doi.org/10.1016/j.pbb.2017.05.002
Lamb RJ, Maguire DR, Ginsburg BC, Pinkston JW, France CP (2016) Determinants of choice, and vulnerability and recovery in addiction. Behav Process 127:35–42. https://doi.org/10.1016/j.beproc.2016.04.001
Li JX, McMahon LR, Gerak LR, Becker GL, France CP (2008) Interactions between Δ 9-tetrahydrocannabinol and μ opioid receptor agonists in rhesus monkeys: discrimination and antinociception. Psychopharmacology 199:199–208. https://doi.org/10.1007/s00213-008-1157-0
Li JX, Koek W, France CP (2012) Interactions between delta9-tetrahydrocannabinol and heroin: self-administration in rhesus monkeys. Behav Pharmacol 23:754–761. https://doi.org/10.1097/FBP.0b013e32835a3907
Maguire DR, France CP (2014) Impact of efficacy at the μ-opioid receptor on antinociceptive effects of combinations of μ-opioid receptor agonists and cannabinoid receptor agonists. J Pharmacol Exp Ther 351:383–389. https://doi.org/10.1124/jpet.114.216648
Maguire DR, France CP (2016a) Interactions between cannabinoid receptor agonists and mu opioid receptor agonists in rhesus monkeys discriminating fentanyl. Eur J Pharmacol 784:199–206. https://doi.org/10.1016/j.ejphar.2016.05.018
Maguire DR, France CP (2016b) Effects of daily delta-9-tetrahydrocannabinol treatment on heroin self-administration in rhesus monkeys. Behav Pharmacol 27:249–257. https://doi.org/10.1097/FBP.0000000000000192
Maguire DR, Gerak LR, France CP (2013a) Delay discounting of food and remifentanil in rhesus monkeys. Psychopharmacology 229:323–330. https://doi.org/10.1007/s00213-013-3121-x
Maguire DR, Yang W, France CP (2013b) Interactions between μ-opioid receptor agonists and cannabinoid receptor agonists in rhesus monkeys: antinociception, drug discrimination, and drug self-administration. J Pharmacol Exp Ther 345:354–362. https://doi.org/10.1124/jpet.113.204099
Maguire DR, Gerak LR, France CP (2013c) Effect of delay on self-administration of remifentanil under a drug versus drug choice procedure in rhesus monkeys. J Pharmacol Exp Ther 347:557–563. https://doi.org/10.1124/jpet.113.208355
Maguire DR, Gerak LR, France CP (2016) Delay discounting of the mu opioid receptor agonist remifentanil in rhesus monkeys. Behav Pharmacol 27:148–154. https://doi.org/10.1097/FBP.0000000000000193
McGregor IS, Issakidis CN, Prior G (1996) Aversive effects of the synthetic cannabinoid CP 55,940 in rats. Pharmacol Biochem Behav 53:657–664. https://doi.org/10.1016/0091-3057(95)02066-7
McMahon LR (2006) Characterization of cannabinoid agonists and apparent pA2 analysis of cannabinoid antagonists in rhesus monkeys discriminating Δ9-tetrahydrocannabinol. J Pharmacol Exp Ther 319:1211–1218. https://doi.org/10.1124/jpet.106.107110
McMahon LR (2011) Chronic Δ9-tetrahydrocannabinol treatment in rhesus monkeys: differential tolerance and cross-tolerance among cannabinoids. Br J Pharmacol 162:1060–1073. https://doi.org/10.1111/j.1476-5381.2010.01116.x
Nader MA, Woolverton WL (1991) Effects of increasing the magnitude of an alternative reinforcer on drug choice in a discrete-trials choice procedure. Psychopharmacology 105:169–174. https://doi.org/10.1007/BF02244304
Nader MA, Woolverton WL (1992) Choice between cocaine and food by rhesus monkeys: effects of conditions of food availability. Behav Pharmacol 3:635-638. https://doi.org/10.1097/00008877-199212000-00010
National Research Council (2011). Guide for the care and use of laboratory animals, 8th ed. National Academies Press, Washington, D.C.
Negus SS (2005a) Interactions between the reinforcing effects of cocaine and heroin in a drug-vs-food choice procedure in rhesus monkeys: a dose-addition analysis. Psychopharmacology 180:115–124. https://doi.org/10.1007/s00213-004-2133-y
Negus SS (2005b) Effects of punishment on choice between cocaine and food in rhesus monkeys. Psychopharmacology 181:244–252. https://doi.org/10.1007/s00213-005-2266-7
Negus SS (2006) Choice between heroin and food in nondependent and heroin-dependent rhesus monkeys: effects of naloxone, buprenorphine, and methadone. J Pharmacol Exp Ther 317:711–723. https://doi.org/10.1124/jpet.105.095380
Nielsen S, Sabioni P, Trigo JM, Ware MA, Betz-Stablein BD, Murnion B, Lintzeris N, Khor KE, Farrell M, Smith A, Le Foll B (2017) Opioid-sparing effect of cannabinoids: a systematic review and meta-analysis. Neuropsychopharmacology 42:1752–1765. https://doi.org/10.1038/npp.2017.51
Ohlsson A, Lindgren JE, Wahlen A, Agurell S, Hollister LE, Gillespie HK (1980) Plasma delta-9-tetrahydrocannabinol concentrations and clinical effects after oral and intravenous administration and smoking. Clin Pharmacol Ther 28:409–416. https://doi.org/10.1038/clpt.1980.181
Perkins FN, Freeman KB (2018) Pharmacotherapies for decreasing maladaptive choice in drug addiction: targeting the behavior and the drug. Pharmacol Biochem Behav 164:40–49. https://doi.org/10.1016/j.pbb.2017.06.015
Podlesnik CA, Ko MC, Winger G, Wichmann J, Prinssen EP, Woods JH (2011) The effects of nociceptin/orphanin FQ receptor agonist Ro 64-6198 and diazepam on antinociception and remifentanil self-administration in rhesus monkeys. Psychopharmacology 213:53–60. https://doi.org/10.1007/s00213-010-2012-7
Riley AL (2011) The paradox of drug taking: the role of the aversive effects of drugs. Physiol Behav 103:69–78. https://doi.org/10.1016/j.physbeh.2010.11.021
Stevenson GW, Folk JE, Rice KC, Negus SS (2005) Interactions between δ and μ opioid agonists in assays of schedule-controlled responding, thermal nociception, drug self-administration, and drug versus food choice in rhesus monkeys: studies with SNC80 [(+)-4-[(αR)-α-((2S, 5R)-4-allyl-2, 5-dimethyl-1-piperazinyl)-3-methoxybenzyl]-N, N-diethylbenzamide] and heroin. J Pharmacol Exp Ther 314:221–231. https://doi.org/10.1124/jpet.104.082685
Wade-Galuska T, Winger G, Woods JH (2007) A behavioral economic analysis of cocaine and remifentanil self-administration in rhesus monkeys. Psychopharmacology 194:563–572. https://doi.org/10.1007/s00213-007-0858-0
Wade-Galuska T, Galuska CM, Winger G (2011) Effects of daily morphine administration and deprivation on choice and demand for remifentanil and cocaine in rhesus monkeys. J Exp Anal Behav 95:75–89. https://doi.org/10.1901/jeab.2011.95-75
Wen H, Hockenberry JM (2018) Association of medical and adult-use marijuana laws with opioid prescribing for medicaid enrollees. JAMA Intern Med 178:673-679. https://doi.org/10.1001/jamainternmed.2018.1007
Winger G, Galuska CM, Hursh SR, Woods JH (2006) Relative reinforcing effects of cocaine, remifentanil, and their combination in rhesus monkeys. J Pharmacol Exp Ther 318:223–229. https://doi.org/10.1124/jpet.105.100461
Woods JH, Winger G (2002) Observing responses maintained by stimuli associated with cocaine or remifentanil reinforcement in rhesus monkeys. Psychopharmacology 163:345–351. https://doi.org/10.1007/s00213-002-1201-4
Woolverton WL, Wang Z, Vasterling T, Tallarida R (2008) Self-administration of cocaine–remifentanil mixtures by monkeys: an isobolographic analysis. Psychopharmacology 198:387–394. https://doi.org/10.1007/s00213-008-1152-5
Acknowledgements
The authors thank Eli Desarno, Steven Garza, Sarah Howard, Jade Juarez, Krissian Martinez, Emily Spolarich, and Samuel Womack for excellent technical assistance. Special thanks to Drs. Gail Winger and Yonggong Shi for technical assistance. This work was supported by the National Institutes of Health (R01DA005018) and the Welch Foundation (AQ-0039). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the National Institute on Drug Abuse.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Maguire, D.R., France, C.P. Reinforcing effects of opioid/cannabinoid mixtures in rhesus monkeys responding under a food/drug choice procedure. Psychopharmacology 235, 2357–2365 (2018). https://doi.org/10.1007/s00213-018-4932-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4932-6