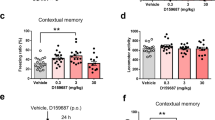
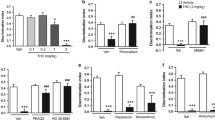
Abstract
Rationale and objectives
Cyclic nucleotide phosphodiesterase-2 (PDE2) is a potential therapeutic target for the treatment of cognitive dysfunction. Using the object recognition test (ORT), this study assessed the effects of two PDE2 inhibitors, Bay 60-7550 and ND7001, on learning and memory, and examined underlying mechanisms.
Methods
To assess the role of PDE2 inhibition on phases of memory, Bay 60-7550 (3 mg/kg) was administered: 30 min prior to training; 0, 1, or 3 h after training; or 30 min prior to recall testing. To assess cyclic nucleotide involvement in PDE2 inhibitor–enhanced memory consolidation, either the nitric oxide synthase inhibitor NG-nitro-L-arginine methyl ester (L-NAME; 20 mg/kg; intraperitoneal (IP)), soluble guanylyl cyclase inhibitor 1H-[−1,2,4]oxadiazolo-[4,3-a]quinoxalin-1-one (ODQ; 20 mg/kg; IP), protein kinase G inhibitor KT5823 (2.5 μg; intracerebroventricular (ICV)), or protein kinase A inhibitor H89 (1 μg; ICV) was administered 30 min prior to the PDE2 inhibitor Bay 60-7550 (3 mg/kg) or ND7001 (3 mg/kg). Changes in the phosphorylation of 3’5’-cyclic adenosine monophosphate (cAMP) response element binding protein (CREB) at Ser-133 and vasodilator-stimulated phosphoprotein (VASP) at Ser-239 were determined to confirm activation of cAMP and 3’5’-cyclic guanosine monophosphate (cGMP) signaling.
Results
Bay 60-7550 (3 mg/kg) enhanced memory of mice in the ORT when given 30 min prior to training, immediately after training, or 30 min prior to recall. Inhibitors of the cGMP pathway blocked the memory-enhancing effects of both Bay 60-7550 (3 mg/kg) and ND7001 (3 mg/kg) on early consolidation processes. Bay 60-7550 (3 mg/kg) enhanced phosphorylation of CREB and VASP, both targets of cGMP-dependent protein kinase (PKG).
Conclusions
These results confirm a potential of PDE2, or components of its signaling pathway, as a therapeutic target for drug discovery focused on restoring memory function.





Similar content being viewed by others
References
Akkerman S, Blokland A, Prickaerts J (2014a) Mind the gap: delayed manifestation of long-term object memory improvement by phosphodiesterase inhibitors. Neurobiol Learn Mem 109:139–43. doi:10.1016/j.nlm.2014.01.006
Akkerman S, Blokland A, van Goethem NP et al (2015) PDE5 inhibition improves acquisition processes after learning via a central mechanism. Neuropharmacology 97:233–239. doi:10.1016/j.neuropharm.2015.04.019
Akkerman S, Prickaerts J, Bruder AK et al (2014b) PDE5 inhibition improves object memory in standard housed rats but not in rats housed in an enriched environment: implications for memory models? PLoS One 9, e111692. doi:10.1371/journal.pone.0111692
Almeida RC, Souza DG, Soletti RC et al (2006) Involvement of PKA, MAPK/ERK and CaMKII, but not PKC in the acute antidepressant-like effect of memantine in mice. Neurosci Lett 395:93–7. doi:10.1016/j.neulet.2005.10.057
Antunes M, Biala G (2012) The novel object recognition memory: neurobiology, test procedure, and its modifications. Cogn Process 13:93–110. doi:10.1007/s10339-011-0430-z
Balderas I, Rodriguez-Ortiz CJ, Bermudez-Rattoni F (2013) Retrieval and reconsolidation of object recognition memory are independent processes in the perirhinal cortex. Neuroscience 253:398–405. doi:10.1016/j.neuroscience.2013.09.001
Beavo J, Brunton LL (2002) Cyclic nucleotide research -- still expanding after half a century. Nat Rev Mol Cell Biol 3:710–8. doi:10.1038/nrm911
Bernabeu R, Schmitz P, Faillace MP et al (1996) Hippocampal cGMP and cAMP are differentially involved in memory processing of inhibitory avoidance learning. Neuroreport 7:585–8
Bianchi M, Fone KFC, Azmi N et al (2006) Isolation rearing induces recognition memory deficits accompanied by cytoskeletal alterations in rat hippocampus. Eur J Neurosci 24:2894–902. doi:10.1111/j.1460-9568.2006.05170.x
Boess FG, Hendrix M, van der Staay F-J et al (2004) Inhibition of phosphodiesterase 2 increases neuronal cGMP, synaptic plasticity and memory performance. Neuropharmacology 47:1081–92. doi:10.1016/j.neuropharm.2004.07.040
Bolger GB, Rodgers L, Riggs M (1994) Differential CNS expression of alternative mRNA isoforms of the mammalian genes encoding cAMP-specific phosphodiesterases. Gene 149:237–44
Bollen E, Akkerman S, Puzzo D et al (2015) Object memory enhancement by combining sub-efficacious doses of specific phosphodiesterase inhibitors. Neuropharmacology 95:361–366. doi:10.1016/j.neuropharm.2015.04.008
Bollen E, Puzzo D, Rutten K et al (2014) Improved long-term memory via enhancing cGMP-PKG signaling requires cAMP-PKA signaling. Neuropsychopharmacology 4:1–9. doi:10.1038/npp.2014.106
Bourtchouladze R, Abel T, Berman N et al (1998) Different training procedures recruit either one or two critical periods for contextual memory consolidation, each of which requires protein synthesis and PKA. Learn Mem 5:365–374. doi:10.1101/lm.5.4.365
Bourtchuladze R, Frenguelli B, Blendy J et al (1994) Deficient long-term memory in mice with a targeted mutation of the cAMP-responsive element-binding protein. Cell 79:59–68. doi:10.1016/0092-8674(94)90400-6
Brown MW, Barker GRI, Aggleton JP, Warburton EC (2012) What pharmacological interventions indicate concerning the role of the perirhinal cortex in recognition memory. Neuropsychologia 50:3122–3140. doi:10.1016/j.neuropsychologia.2012.07.034
Carlezon WA, Duman RS, Nestler EJ (2005) The many faces of CREB. Trends Neurosci 28:436–445. doi:10.1016/j.tins.2005.06.005
Castellucci VF, Kandel ER, Schwartz JH et al (1980) Intracellular injection of the catalytic subunit of cyclic AMP-dependent protein kinase simulates facilitation of transmitter release underlying behavioral sensitization in Aplysia. Proc Natl Acad Sci 77:7492–7496. doi:10.1073/pnas.77.12.7492
Chien W-L, Liang K-C, Teng C-M et al (2005) Enhancement of learning behaviour by a potent nitric oxide-guanylate cyclase activator YC-1. Eur J Neurosci 21:1679–88. doi:10.1111/j.1460-9568.2005.03993.x
Chijiwa T, Mishima A, Hagiwara M et al (1990) Inhibition of forskolin-induced neurite outgrowth and protein phosphorylation by a newly synthesized selective inhibitor of cyclic AMP-dependent protein kinase, N-[2-(p-bromocinnamylamino)ethyl]-5-isoquinolinesulfonamide (H-89), of PC12D pheochromocytoma. J Biol Chem 265:5267–72
Cunha MP, Budni J, Pazini FL et al (2014) Involvement of PKA, PKC, CAMK-II and MEK1/2 in the acute antidepressant-like effect of creatine in mice. Pharmacol Reports 66:653–659. doi:10.1016/j.pharep.2014.03.004
Dere E, Huston JP, De Souza Silva MA (2007) The pharmacology, neuroanatomy and neurogenetics of one-trial object recognition in rodents. Neurosci Biobehav Rev 31:673–704. doi:10.1016/j.neubiorev.2007.01.005
Ding L, Zhang C, Masood A et al (2014) Protective effects of phosphodiesterase 2 inhibitor on depression- and anxiety-like behaviors: Involvement of antioxidant and anti-apoptotic mechanisms. Behav Brain Res 268:150–8. doi:10.1016/j.bbr.2014.03.042
Domek-Łopacińska K, Strosznajder JB (2008) The effect of selective inhibition of cyclic GMP hydrolyzing phosphodiesterases 2 and 5 on learning and memory processes and nitric oxide synthase activity in brain during aging. Brain Res 1216:68–77. doi:10.1016/j.brainres.2008.02.108
Douglas LA, Varlinskaya EI, Spear LP (2003) Novel-object place conditioning in adolescent and adult male and female rats: effects of social isolation. Physiol Behav 80:317–25
Ennaceur A, Delacour J (1988) A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behav Brain Res 31:47–59
Greengard P (1976) Possible role for cyclic nucleotides and phosphorylated membrane proteins in postsynaptic actions of neurotransmitters. Nature 260:101–8
Hall FS (1998) Social deprivation of neonatal, adolescent, and adult rats has distinct neurochemical and behavioral consequences. Crit Rev Neurobiol 12:129–62
Johannessen M, Moens U (2007) Multisite phosphorylation of the cAMP response element-binding protein (CREB) by a diversity of protein kinases. Front Biosci 12:1814–1832. doi:10.2741/2190
Keravis T, Lugnier C (2012) Cyclic nucleotide phosphodiesterase (PDE) isozymes as targets of the intracellular signalling network: benefits of PDE inhibitors in various diseases and perspectives for future therapeutic developments. Br J Pharmacol 165:1288–305. doi:10.1111/j.1476-5381.2011.01729.x
Kleppisch T (2009) Phosphodiesterases in the central nervous system. Handb Exp Pharmacol 71–92. doi: 10.1007/978-3-540-68964-5_5
Lapiz MDS, Fulford A, Muchimapura S et al (2003) Influence of postweaning social isolation in the rat on brain development, conditioned behavior, and neurotransmission. Neurosci Behav Physiol 33:13–29. doi:10.1023/A:1021171129766
Lee B, Butcher GQ, Hoyt KR et al (2005) Activity-dependent neuroprotection and cAMP response element-binding protein (CREB): kinase coupling, stimulus intensity, and temporal regulation of CREB phosphorylation at serine 133. J Neurosci 25:1137–1148. doi:10.1523/JNEUROSCI.4288-04.2005
Lee MC, Smith FL, Stevens DL, Welch SP (2003) The role of several kinases in mice tolerant to delta 9-tetrahydrocannabinol. J Pharmacol Experamental Ther 305:593–9. doi:10.1124/jpet.102.044446
Leger M, Quiedeville A, Bouet V et al (2013) Object recognition test in mice. Nat Protoc 8:2531–7. doi:10.1038/nprot.2013.155
Liu P, Smith PF, Appleton I et al (2003) Nitric oxide synthase and arginase in the rat hippocampus and the entorhinal, perirhinal, postrhinal, and temporal cortices: Regional variations and age-related changes. Hippocampus 13:859–867. doi:10.1002/hipo.10138
Masood A, Huang Y, Hajjhussein H et al (2009) Anxiolytic effects of phosphodiesterase-2 inhibitors associated with increased cGMP signaling. J Pharmacol Exp Ther 331:690–699. doi:10.1124/jpet.109.156729.1993
Masood A, Nadeem A, Mustafa SJ et al (2008) Reversal of oxidative stress-induced anxiety by inhibition of phosphodiesterase-2 in mice. J Pharmacol Experamental Ther 326:369–379. doi:10.1124/jpet.108.137208.be
Nonaka M, Kim R, Fukushima H et al (2014) Region-specific activation of CRTC1-CREB signaling mediates long-term fear memory. Neuron 84:92–106. doi:10.1016/j.neuron.2014.08.049
Omori K, Kotera J (2007) Overview of PDEs and their regulation. Circ Res 100:309–27. doi:10.1161/01.RES.0000256354.95791.f1
Paxinos G, Franklin KB (2008) The mouse brain in stereotaxic coordinates, compact, third edition: the coronal plates and diagrams, 3rd edn. Academic Press
Prickaerts J, De Vente J, Honig W et al (2002) cGMP, but not cAMP, in rat hippocampus is involved in early stages of object memory consolidation. Eur J Pharmacol 436:83–87. doi:10.1016/S0014-2999(01)01614-4
Reneerkens O, Rutten K, Bollen E, et al. (2013) Inhibition of phoshodiesterase type 2 or type 10 reverses object memory deficits induced by scopolamine or MK-801. Behav Brain Res 236:16–22. doi:10.1016/j.bbr.2012.08.019
Rodefer JS, Saland SK, Eckrich SJ (2012) Selective phosphodiesterase inhibitors improve performance on the ED/ID cognitive task in rats. Neuropharmacology 62:1182–90. doi:10.1016/j.neuropharm.2011.08.008
Rutten K, Prickaerts J, Blokland A (2006) Rolipram reverses scopolamine-induced and time-dependent memory deficits in object recognition by different mechanisms of action. Neurobiol Learn Mem 85:132–138. doi:10.1016/j.nlm.2005.09.002
Rutten K, Prickaerts J, Hendrix M et al (2007) Time-dependent involvement of cAMP and cGMP in consolidation of object memory: studies using selective phosphodiesterase type 2, 4 and 5 inhibitors. Eur J Pharmacol 558:107–12. doi:10.1016/j.ejphar.2006.11.041
Rutten K, Van Donkelaar EL, Ferrington L et al (2009) Phosphodiesterase inhibitors enhance object memory independent of cerebral blood flow and glucose utilization in rats. Neuropsychopharmacology 34:1914–25. doi:10.1038/npp.2009.24
Seoane A, Tinsley CJ, Brown MW (2012) Interfering with Fos expression in rat perirhinal cortex impairs recognition memory. Hippocampus 22:2101–2113. doi:10.1002/hipo.22028
Stephenson DT, Coskran TM, Wilhelms MB et al (2009) Immunohistochemical localization of phosphodiesterase 2A in multiple mammalian species. J Histochem Cytochem 57:933–49. doi:10.1369/jhc.2009.953471
Suvarna NU, O’Donnell JM (2002) Hydrolysis of N-methyl-D-aspartate receptor-stimulated cAMP and cGMP by PDE4 and PDE2 phosphodiesterases in primary neuronal cultures of rat cerebral cortex and hippocampus. J Pharmacol Experamental Ther 302:249–56
Suzuki W (1996) The anatomy, physiology and functions of the perirhinal cortex. Curr Opin Neurobiol 6:179–186. doi:10.1016/S0959-4388(96)80071-7
Thorsell A, Slawecki CJ, El Khoury A et al (2006) The effects of social isolation on neuropeptide Y levels, exploratory and anxiety-related behaviors in rats. Pharmacol Biochem Behav 83:28–34. doi:10.1016/j.pbb.2005.12.005
van Donkelaar EL, Rutten K, Blokland A et al (2008) Phosphodiesterase 2 and 5 inhibition attenuates the object memory deficit induced by acute tryptophan depletion. Eur J Pharmacol 600:98–104. doi:10.1016/j.ejphar.2008.10.027
van Goethem NP, Rutten K, van der Staay FJ et al (2012) Object recognition testing: Rodent species, strains, housing conditions, and estrous cycle. Behav Brain Res 232:323–334. doi:10.1016/j.bbr.2012.03.023
van Groen T (2001) Entorhinal cortex of the mouse: cytoarchitectonical organization. Hippocampus 11:397–407. doi:10.1002/hipo.1054
van Staveren WC, Markerink-van Ittersum M, Steinbusch HW, de Vente J (2001) The effects of phosphodiesterase inhibition on cyclic GMP and cyclic AMP accumulation in the hippocampus of the rat. Brain Res 888:275–286
Van Staveren WCG, Steinbusch HWM, Markerink-Van Ittersum M et al (2003) mRNA expression patterns of the cGMP-hydrolyzing phosphodiesterases types 2, 5, and 9 during development of the rat brain. J Comp Neurol 467:566–80. doi:10.1002/cne.10955
Wan H, Aggleton JP, Brown MW (1999) Different contributions of the hippocampus and perirhinal cortex to recognition memory. J Neurosci 19:1142–1148
Weiss IC, Pryce CR, Jongen-Rêlo AL et al (2004) Effect of social isolation on stress-related behavioural and neuroendocrine state in the rat. Behav Brain Res 152:279–295. doi:10.1016/j.bbr.2003.10.015
Winters BD, Saksida LM, Bussey TJ (2008) Object recognition memory: neurobiological mechanisms of encoding, consolidation and retrieval. Neurosci Biobehav Rev 32:1055–1070. doi:10.1016/j.neubiorev.2008.04.004
Wu Z-Q, Li M, Chen J et al (2006) Involvement of cAMP/cAMP-dependent protein kinase signaling pathway in regulation of Na+, K + −ATPase upon activation of opioid receptors by morphine. Mol Pharmacol 69:866–76. doi:10.1124/mol.105.016501
Xu Y, Pan J, Chen L et al (2013) Phosphodiesterase-2 inhibitor reverses corticosterone-induced neurotoxicity and related behavioural changes via cGMP/PKG dependent pathway. Int J Neuropsychopharmacol 16:835–47. doi:10.1017/S146114571200065X
Xu Y, Pan J, Sun J et al (2015) Inhibition of phosphodiesterase 2 reverses impaired cognition and neuronal remodeling caused by chronic stress. Neurobiol Aging 36:955–970. doi:10.1016/j.neurobiolaging.2014.08.028
Zhang H-T, Huang Y, Jin S-L et al (2002) Antidepressant-like profile and reduced sensitivity to rolipram in mice deficient in the PDE4D phosphodiesterase enzyme. Neuropsychopharmacology 27:587–95. doi:10.1016/S0893-133X(02)00344-5
Acknowledgments
This research was supported by a grant from the National Institute of Mental Health (MH088480).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lueptow, L.M., Zhan, CG. & O’Donnell, J.M. Cyclic GMP–mediated memory enhancement in the object recognition test by inhibitors of phosphodiesterase-2 in mice. Psychopharmacology 233, 447–456 (2016). https://doi.org/10.1007/s00213-015-4129-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-015-4129-1