Abstract
Introduction
The α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA) receptor potentiator Org 26576 represents an interesting pharmacological tool to evaluate the utility of glutamatergic enhancement towards the treatment of psychiatric disorders. In this study, a rat–human translational pharmacokinetic–pharmacodynamic (PK–PD) model of AMPA receptor modulation was used to predict human target engagement and inform dose selection in efficacy clinical trials.
Methods
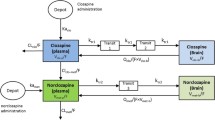
Modelling and simulation was applied to rat plasma and cerebrospinal fluid (CSF) pharmacokinetic and pharmacodynamic measurements to identify a target concentration (EC80) for AMPA receptor modulation. Human plasma pharmacokinetics was determined from 33 healthy volunteers and eight major depressive disorder patients. From four out of these eight patients, CSF PK was also determined. Simulations of human CSF levels were performed for several doses of Org 26576.
Results
Org 26576 (0.1–10 mg/kg, i.v.) potentiated rat hippocampal AMPA receptor responses in an exposure-dependant manner. The rat plasma and CSF PK data were fitted by one-compartment model each. The rat CSF PK–PD model yielded an EC80 value of 593 ng/ml (90% confidence interval 406.8, 1,264.1). The human plasma and CSF PK data were simultaneously well described by a two-compartment model. Simulations showed that in humans at 100 mg QD, CSF levels of Org 26576 would exceed the EC80 target concentration for about 2 h and that 400 mg BID would engage AMPA receptors for 24 h.
Conclusion
The modelling approach provided useful insight on the likely human dose–molecular target engagement relationship for Org 26576. Based on the current analysis, 100 and 400 mg BID would be suitable to provide ‘phasic’ and ‘continuous’ AMPA receptor engagement, respectively.








Similar content being viewed by others
References
Arai AC, Kessler M (2007) Pharmacology of ampakine modulators: from AMPA receptors to synapses and behaviour. Curr Drug Targets 8:583–602
Bigge CF (1999) Ionotropic glutamate receptors. Curr Opin Chem Biol 3:441–447
Bowie D (2008) Ionotropic glutamate receptors & CNS disorders. CNS Neurol Disord Drug Targets 7(2):129–143
Broberg BV, Glenthøj BY, Dias R, Larsen DB, Olsen CK (2009) Reversal of cognitive deficits by an ampakine (CX516) and sertindole in two animal models of schizophrenia-sub-chronic and early postnatal PCP treatment in attentional set-shifting. Psychopharmacology 206(4):631–640
Damgaard T, Larsen DB, Hansen SL, Grayson B, Neill JC, Plath N (2010) Positive modulation of alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors reverses sub-chronic PCP-induced deficits in the novel object recognition task in rats. Behav Brain Res 207(1):144–150
Danhof M, de Lange ECM, Della Pasqua OE, Ploeger BA, Voskuyl RA (2008) Mechanism-based pharmacokinetic–pharmacodynamic (PK-PD) modeling in translational drug research. Trends Pharmacol Sci 29:186–191
de Lange ECM, Danhof M (2002) Considerations in the use of cerebrospinal fluid pharmacokinetics to predict brain target concentrations in the clinical setting: implications of the barriers between blood and brain. Clin Pharmacokinet 41:691–703
Dingledine R, Borges K, Bowie D, Traynelis SF (1999) The glutamate receptor ion channels. Pharmacol Rev 51(1):7–61
Erdemli G, Smith LH, Sammons M, Jeggo R, Shahid M (2007) Org 26576, a novel positive allosteric modulator, potentiates AMPA receptor responses in hippocampal neurones. J Neuropsychopharmacol 21:A51
Goff DC, Leahy L, Berman I, Posever T, Herz L, Leon AC, Johnson SA, Lynch G (2001) A placebo-controlled pilot study of the ampakine CX516 added to clozapine in schizophrenia. J Clin Psycopharmacol 21:484–487
Goff DC, Lambert JS, Leon AC, Green MF, Miller AL, Patel J, Manschreck T, Freudenreich O, Johnson SA (2008) A placebo-controlled add-on trial of the ampakine, CX516, for cognitive deficits in schizophrenia. Neuropsychopharmacol 33:465–472
Hamlyn E, Brand L, Shahid M, Harvey BH (2009) The ampakine, Org 26576, bolsters early spatial reference learning and retrieval in the Morris water maze: a subchronic, dose-ranging study in rats. Behav Pharmacol 20(7):662–667
Hampson RE, Rogers G, Lynch G, Deadwyler SA (1998a) Facilitative effects of the ampakine CX516 on short-term memory in rats: enhancement of delayed-no match-to-sample performance. J Neurosci 18:2740–2747
Hampson RE, Rogers G, Lynch G, Deadwyler SA (1998b) Facilitative effects of the ampakine CX516 on short-term memory in rats: correlations with hippocampal neuronal activity. J Neurosci 18:2748–2763
Hutmacher MM, Bursi R, Chapel S, Kerbusch T (2008) Implications for animal–human scaling of the parallel elimination profile PK model. Abstract 1374, Population Approach Group Europe, 17th Meeting, 18–20 June, Marseille, France
Ito I, Tanabe S, Kohda A, Sugiyama H (1990) Allosteric potentiation of quisqualate receptors by a nootropic drug aniracetam. J Physiol 424:533–543
Jeggo RD, Wang Y, Jordan D, Ramage AG (2007) Activation of 5-HT1B and 5-HT1D receptors in the rat nucleus tractus solitarius: opposing action on neurones that receive an excitatory vagal C-fibre afferent input. Br J Pharmacol 150:987–995
Johnson SA, Luu NT, Herbst TA, Knapp R, Lutz D, Arai A, Rogers GA, Lynch G (1999) Synergistic interactions between ampakines and antipsychotic drugs. L Pharmacol Exp Ther 289(1):392–397
Jordan GR, McCulloch J, Shahid M, Hill DR, Henry B, Horsburgh K (2005) Regionally selective and dose-dependent effects of the ampakines Org 26576 and Org 24448 on local cerebral glucose utilisation in the mouse as assessed by 14C-2-deoxyglucose autoradiography. Neuropharmacology 49:254–264
Kew JN, Kemp JA (2005) Ionotropic and metabotropic glutamate receptor structure and pharmacology. Psychopharmacology 179(1):4–29
Knapp RJ, Goldenberg R, Shuck C, Cecil A, Watkins J, Miller C, Crites G, Malatynska E (2002) Antidepressant activity of memory-enhancing drugs in the reduction of submissive behaviour model. Eur J Pharmacol 440(1):27–35
Larson J, Quach CN, LeDuc BQ, Nguyen A, Rogers GA, Lynch G (1996) Effects of an AMPA receptor modulator on methamphetamine-induced hyperactivity in rats. Brain Res 738:353–356
Li X, Tizzano JP, Griffey K, Clay M, Lindstrom T, Skolnick P (2001) Antidepressant-like actions of an AMPA receptor potentiator (LY392098). Neuropharmacology 40:1028–1033
Lynch G (2004) AMPA receptor modulators as cognitive enhancers. Curr Opin Pharmacol 4:4–11
Lynch G (2006) Glutamate-based therapeutic approaches: ampakines. Curr Opin Pharmacol 6:82–88
Lynch G, Gall CM (2006) Ampakines and the threefold path to cognitive enhancement. Trends Neurosci 29:554–562
Mager DE, Jusko WJ (2008) Development of translational pharmacokinetic–pharmacodynamic models. Clin Pharm Ther 83:909–912
Mankoff SP, Brander C, Ferrone S, Marincola FM (2004) Lost in translation: obstacles to translational medicine. J Transl Med 2:1–5
Nikisch G, Baumann P, Kiessling B, Reinert M, Wiedemann G, Kehr J, Mathé AA, Piel M, Roesch F, Weisser H, Schneider P, Hertel A (2010) Relationship between dopamine D2 receptor occupancy, clinical response, and drug and monoamine metabolites levels in plasma and cerebrospinal fluid. A pilot study in patients suffering from first-episode schizophrenia treated with quetiapine. J Psychiatr Res 44:754–759
Olsen CK, Kreilgaard M, Didriksen M (2006) Positive modulation of glutamatergic receptors potentiates the suppressive effects of antipsychotics on conditioned avoidance responding in rats. Pharmacol Biochem Behav 84(2):259–265
O’Neill MJ, Bleakman D, Zimmerman DM, Nisenbaum ES (2004a) AMPA receptor potentiators for the treatment of CNS disorders. Curr Drug Targets CNS Neurol Disord 3(3):181–194
O’Neill MJ, Murray TK, Whalley K, Ward MA, Hicks CA, Woodhouse S, Osborne DJ, Skolnick P (2004b) Neurotrophic actions of the novel AMPA receptor potentiator LY404187, in rodent models of Parkinson’s disease. Eur J Pharmacol 486:163–174
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates. Academic Press, Sidney, p 154
Porrino LJ, Daunais JB, Rogers GA, Hampson RE, Deadwyler SA (2005) Facilitation of task performance and removal of the effects of sleep deprivation by an ampakine (CX717) in nonhuman primates. PLoS Biol 3:1639–1652
Sheiner LB, Steimer J-L (2000) Pharmacokinetic/pharmacodynamic modeling in drug development. Annu Rev Pharacol Toxicol 40:67–95
Su XW, Li XY, Banasr M, Koo JW, Shahid M, Henry B, Duman RS (2009) Chronic treatment with AMPA receptor potentiator Org 26576 increases neuronal cell proliferation and survival in adult rodent hippocampus. Psychopharmacology 206(2):215–222
Tierney PL, Thierry AM, Glowinski J, Deniau JM, Gioanni Y (2008) Dopamine modulates temporal dynamics of feedforward inhibition in rat prefrontal cortex in vivo. Cereb Cortex 18:2251–2262
Vandergriff J, Huff K, Bond A, Lodge D (2001) Potentiation of responses to AMPA on central neurones by LY392098 and LY404187 in vivo. Neuropharmacol 40:1003–1009
Venkatakrishnan K, Tseng E, Nelson FR, Rollema H, French JL, Kaplan IV, Horner WE, Gibbs MA (2007) Central nervous system pharmacokinetics of the Mdr1 P-glycoprotein substrate CP-615,003: intersite differences and implications for human receptor occupancy projections from cerebrospinal fluid exposures. Drug Metab Dispos 35:1341–1349
Wählby U, Jonsson EN, Karlsson MO (2001) Assessment of actual significance levels for covariate effects in NONMEM. J Pharmacokinet Pharmacodyn 28(3):231–251
Watson J, Wright S, Lucas A, Clarke KL, Viggers J, Cheetham S, Jeffrey P, Porter R, Read KD (2009) Receptor occupancy and brain free fraction. Drug Metab Dispos 37:753–760
Wezenberg E, Verkes RJ, Ruigt GSF, Hulstijn W, Sabbe BGC (2007) Acute effects of the ampakine farampator on memory and information processing in healthy elderly volunteers. Neuropsychopharmacol 32:1272–1283
Woolley ML, Waters KA, Gartlon JE, Lacroix LP, Jennings C, Shaughnessy F, Ong A, Pemberton DJ, Harries MH, Southam E, Jones DN, Dawson LA (2009) Evaluation of the pro-cognitive effects of the AMPA receptor positive modulator, 5-(1-piperidinylcarbonyl)-2,1,3-benzoxadiazole (CX691), in the rat. Psychopharmacology 202(1–3):343–354
Acknowledgements
This study was funded by Schering-Plough Corporation, now Merck (Whitehouse Station, NJ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bursi, R., Erdemli, G., Campbell, R. et al. Translational PK–PD modelling of molecular target modulation for the AMPA receptor positive allosteric modulator Org 26576. Psychopharmacology 218, 713–724 (2011). https://doi.org/10.1007/s00213-011-2365-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-011-2365-6