Abstract
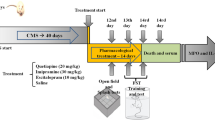
Major depressive disorder (or depression) is one of the most frequent psychiatric illnesses in the population, with chronic stress being one of the main etiological factors. Studies have shown that cholecalciferol supplementation can lead to attenuation of the depressive state; however, the biochemical mechanisms involved in the relationship between cholecalciferol and depression are not very well known. The objective of this study was to investigate the effects of the administration of cholecalciferol on behavioral parameters (tail suspension test (TST), open field test (OFT), splash test (ST)) and redox state (dichlorofluorescein (DCF)) in adult female Swiss mice subjected to a model of depression induced by chronic corticosterone treatment. Corticosterone (20 mg/kg, p.o.) was administered once a day for 21 days. For investigation of the antidepressant-like effect, cholecalciferol (100 IU/kg) or fluoxetine (10 mg/kg, positive control) was administered p.o. within the last 7 days of corticosterone administration. After the treatments, the behavioral tests and biochemical analyses in the hippocampus and prefrontal cortex of the rodent samples were performed. Animals submitted to repeated corticosterone administration showed a depressive-like behavior, evidenced by a significant increase in the immobility time in the TST, which was significantly reduced by the administration of cholecalciferol or fluoxetine. In addition, the groups treated with cholecalciferol and fluoxetine showed a significant decrease in the production of reactive oxygen species (ROS) in the hippocampus. These results show that cholecalciferol, similar to fluoxetine, has a potential antidepressant-like effect, which may be related to the lower ROS production.




Similar content being viewed by others
Abbreviations
- DCF:
-
Dichlorofluorescein
- DCFH:
-
2,7 Dichlorofluorescein
- DMSO:
-
Dimethylsulfoxide
- ROS:
-
Reactive oxygen species
- HPA:
-
Hypothalamic-pituitary-adrenal
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- OFT:
-
Open field test
- ST:
-
Splash test
- TNF:
-
Forced swim test
- TST:
-
Tail suspension test
- VDR:
-
Vitamin D receptor
References
Alekhya P, Sriharsha M, Ramudu VR et al (2015) Adherence to antidepressant therapy: sociodemographic factor wise distribution. Int J Pharm Clin Res 7:180–184
Alrefaie Z, Alhayani A (2015) Vitamin D improves decline in cognitive function and cholinergic transmission in prefrontal cortex of streptozotocin-induced diabetic rats. Behav Brain Res 287:156–162
Anglin RE, Samaan Z, Walter SD, McDonald SD (2013) Vitamin D deficiency and depression in adults: systematic review and meta-analysis. Br J Psychiatry 202:100–107
Balcombe JP, Barnard ND, Sandusky C (2004) Laboratory routines cause animal stress. Contemp Top Lab Anim Sci 43:42–51
Bet PM, Hugtenburg JG, Penninx BW, Hoogendijk WJ (2013) Side effects of antidepressants during long-term use in a naturalistic setting. Eur Neuropsychopharmacol 23:1443–1451
Beyer JL, Payne ME (2016) Nutrition and bipolar depression. Psychiatr Clin North Am 39:75–86
Brouwer-Brolsma EM, Dhonukshe-Rutten RA, Van Wijngaarden JP et al (2016) Low vitamin D status is associated with more depressive symptoms in Dutch older adults. Eur J Nutr 55:1525–1534
Camargo A, Dalmagro AP, Rikel L, da Silva EB, Simão da Silva KAB, Zeni ALB (2018) Cholecalciferol counteracts depressive-like behavior and oxidative stress induced by repeated corticosterone treatment in mice. Eur J Pharmacol 833:451–461
Casseb GAS, Kaster MP, Rodrigues ALS (2019) Potential role of vitamin D for the management of depression and anxiety. CNS Drugs 33:619–637
Chen KB, Lin AM, Chiu TH (2003) Systemic vitamin D3 attenuated oxidative injuries in the locus coeruleus of rat brain. Ann N Y Acad Sci 993:313–249
Circu ML, Aw TY (2010) Reactive oxygen species, cellular redox systems and apoptosis. Sciences (New York) 48:749–762
Cui C, Song S, Cui J, Feng Y, Gao J, Jiang P (2017) Vitamin D receptor activation influences NADPH oxidase (NOX2) activity and protects against neurological deficits and apoptosis in a rat model of traumatic brain injury. Oxidative Med Cell Longev 2017:9245702
Curtis KS, Davis LM, Johnson AL, Therrien KL, Contreras RJ (2004) Sex differences in behavioral taste responses to and ingestion of sucrose and NaCl solutions by rats. Physiol Behav 80:657–664
Erbaş O, Solmaz V, Aksoy D, Yavaşoğlu A, Sağcan M, Taşkıran D (2014) Cholecalciferol (vitamin D 3) improves cognitive dysfunction and reduces inflammation in a rat fatty liver model of metabolic syndrome. Life Sci 103:68–72
Eyles DW, Burne TH, McGrath JJ (2013) Vitamin D, effects on brain development, adult brain function and the links between low levels of vitamin D and neuropsychiatric disease. Front Neuroendocrinol 34:47–64
Fedotova J, Dudnichenko T, Kruzliak P, Puchavskaya Z (2016) Different effects of vitamin D hormone treatment on depression-like behavior in the adult ovariectomized female rats. Biomed Pharmacother 84:1865–1872
Gupta D, Radhakrishnan M, Kurhe Y (2015) Effect of a novel 5-HT 3 receptor antagonist 4i , in corticosterone-induced depression-like behavior and oxidative stress in mice. Steroids 96:95–102
Hallgren M, Stubbs B, Vancampfort D, Lundin A, Jääkallio P, Forsell Y (2017) Treatment guidelines for depression: greater emphasis on physical activity is needed. Eur Psychiatry 40:1–3
Han C, Lim YH, Honget YC (2016) The association between oxidative stress and depressive symptom scores in elderly population : a repeated panel study. J Prev Med Public Health 49:260–274
Hempel SL, Buettner GR, O'Malley YQ, Wessels DA, Flaherty DM (1999) Dihydrofluorescein diacetate is superior for detecting. Free Radic Biol Med 27:146–159
Ibi M, Sawada H, Nakanishi M, Kume T, Katsuki H, Kaneko S, Shimohama S, Akaike A (2001) Protective effects of 1α,25–(OH)2D3 against the neurotoxicity of glutamate and reactive oxygen species in mesencephalic culture. Neuropharmacology 40:761–771
Jin J, Maren S (2015) Prefrontal-hippocampal interactions in memory and emotion. Front Syst Neurosci 9:170
Joseph JJ, Golden SH (2016) Cortisol dysregulation: the bidirectional link between stress, depression, and type 2 diabetes mellitus. Ann N Y Acad Sci 1391:20-34
Kalueff AV, Lou YR, Laaksi I, Tuohimaa P (2004) Increased grooming behavior in mice lacking vitamin D receptors. Physiol Behav 82:405–409
Khawam EA, Laurencic G, Malone DA Jr (2006) Side effects of antidepressants: an overview. Cleve Clin J Med 73:1–9
Kim GH, Kim JE, Rhie SJ, Yoon S (2015) The role of oxidative stress in neurodegenerative diseases. Exp Neurobiol 24:325–340
Lardner AL (2015) Vitamin D and hippocampal development-the story so far. Front Mol Neurosci 8:58
Liu T, Zhong S, Liao X, Chen J, He T, Lai S, Jia Y (2015) A meta-analysis of oxidative stress markers in depression. PLoS One 10:1–17
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folinphenol reagent. J Biol Chem 193:265–267
Lucca G, Comim CM, Valvassori SS, Réus GZ, Vuolo F, Petronilho F, Dal-Pizzol F, Gavioli EC, Quevedo J (2009) Effects of chronic mild stress on the oxidative parameters in the rat brain. Neurochem Int 54:358–362
Manna P, Achari AE, Jain SK (2017) Vitamin D supplementation inhibits oxidative stress and upregulate SIRT1/AMPK/GLUT4 cascade in high glucose-treated 3T3L1 adipocytes and in adipose tissue of high fat diet-fed diabetic mice. Arch Biochem Biophys 615:22–34
Manoharan S, Guillemin GJ, Abiramasundari RS, Essa MM, Akbar M, Akbar MD (2016) The role of reactive oxygen species in the pathogenesis of Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease: a mini review. Oxidative Med Cell Longev 2016:1–15
Manosso LM, Moretti M, Rodrigues ALS (2013) Nutritional strategies for dealing with depression. Food Funct 4:1776–1793
Maurya PK, Noto C, Rizzo LB, Rios AC, Nunes SO, Barbosa DS et al (2016) The role of oxidative and nitrosative stress in accelerated aging and major depressive disorder. Prog Neuro-Psychopharmacol Biol Psychiatry 65:134–144
Moniczewski A, Gawlik M, Smaga I, Niedzielska E, Krzek J, Przegaliński E, Pera J, Filip M (2015) Oxidative stress as an etiological factor and a potential treatment target of psychiatric disorders. Part 1. Chemical aspects and biological sources of oxidative stress in the brain. Pharmacol Rep 67:560–568
Moretti M, Colla A, de Oliveira BG, dos Santos DB, Budni J, de Freitas AE, Farina M, Rodrigues ALS (2012) Ascorbic acid treatment, similarly to fluoxetine, reverses depressive-like behavior and brain oxidative damage induced by chronic unpredictable stress. J Psychiatr Res 46:331–340
Moretti M, Budni J, Dos Santos DB, Antunes A, Daufenbach JF, Manosso LM, Farina M, Rodrigues ALS (2013) Protective effects of ascorbic acid on behavior and oxidative status of restraint-stressed mice. J Mol Neurosci 46:68–79
Morimoto M, Morita N, Ozawa H, Yokoyama K, Kawata M (1996) Distribution of glucocorticoid receptor immunoreactivity and mRNA in the rat brain: an immunohistochemical and in situ hybridization study. Neurosci Res 26(3):235–269
Mozaffari-Khosravi H, Nabizade L, Yassini-Ardakani SM, Hadinedoushan H, Barzegar K (2013) The effect of 2 different single injections of high dose of vitamin D on improving the depression in depressed patients with vitamin D deficiency: a randomized clinical trial. J Clin Psychopharmacol 33:378–385
Mpandzou G, Aït Ben Haddou E, Regragui W, Benomar A, Yahyaoui M (2016) Vitamin D deficiency and its role in neurological conditions: a review. Rev Neurol (Paris) 172:109–22
Olescowicz G, Neis VB, Fraga DB, Rosa PB, Azevedo DP, Melleu FF, Brocardo PS, Gil-Mohapel J, Rodrigues ALS (2017) Antidepressant and pro-neurogenic effects of agmatine in a mouse model of stress induced by chronic exposure to corticosterone. Prog Neuro-Psychopharmacol Biol Psychiatry 81:395–407
Otte C, Gold SM, Penninx BW, Pariante CM, Etkin A, Fava M, Mohr DC, Schatzberg AF (2016) Major depressive disorder. Nat Rev Dis Primers 2:1065
Parker GB, Brotchie H, Graham RK (2017) Vitamin D and depression. J Affect Disord 208:56–61
Rodrigues AL, Rocha JB, Mello CF, Souza DO (1996) Effect of perinatal lead exposure on rat behaviour in open-field and two-way avoidance tasks. Pharmacol Toxicol 79:150–156
Rosa PB, Ribeiro CM, Bettio LE, Colla A, Lieberknecht V, Moretti M, Rodrigues AL (2014) Folic acid prevents depressive-like behavior induced by chronic corticosterone treatment in mice. Pharmacol Biochem Behav 127:1–6
Sanacora G, Treccani G, Popoli M (2012) Towards a glutamate hypothesis of depression: an emerging frontier of neuropsychopharmacology for mood disorders. Neurophamarcology 62:63–77
Sato H, Takahashi T, Sumitani K, Takatsu H, Urano S (2010) Glucocorticoid generates ROS to induce oxidative injury in the hippocampus, leading to impairment of cognitive function of rats. J Clin Biochem Nutr 47(3):224–232
Sepehrmanesh Z, Kolahdooz F, Abedi F, Mazroii N, Assarian A, Asemi Z, Esmaillzadeh A (2016) Vitamin D supplementation affects the Beck depression inventory, insulin resistance, and biomarkers of oxidative stress in patients with major depressive disorder: a randomized, controlled clinical trial. J Nutr 46:243–248
Shin YC, Jung CH, Kim HJ, Kim EJ, Lim SW (2016) The associations among vitamin D deficiency, C-reactive protein, and depressive symptoms. J Psychosom Res 90:98–104
Silva MC, de Sousa CN, Gomes PX, de Oliveira GV, Araújo FY, Ximenes NC, da Silva JC et al (2016) Evidence for protective effect of lipoic acid and desvenlafaxine on oxidative stress in a model depression in mice. Prog Neuro-Psychopharmacol Biol Psychiatry 64:142–148
Smaga I, Niedzielska E, Gawlik M, Moniczewski A, Krzek J, Przegaliński E, Pera J, Filip M (2015) Oxidative stress as an etiological factor and a potential treatment target of psychiatric disorders. Part 2. Depression, anxiety, schizophrenia and autism. Pharmacol Rep 67:569–580
Spiers JG, Chen HJ, Bradley AJ, Anderson ST, Sernia C, Lavidis NA (2013) Acute restraint stress induces rapid and prolonged changes in erythrocyte and hippocampal redox status. Psychoneuroendocrinology 38:2511–2519
Spiers JG, Chen HJ, Sernia C, Lavidis NA (2015) Activation of the hypothalamic-pituitary-adrenal stress axis induces cellular oxidative stress. Front Neurosci 8:456
Steru L, Chermat R, Thierry B, Simon P (1985) The tail suspension test - a new method for screening antidepressants in mice. Psychopharmacology 85:367–370
Tarbali S, Khezri S (2016) Vitamin D3 attenuates oxidative stress and cognitive deficits in a model of toxic demyelination. Iran J Basic Med Sci 19:80–88
Velimirović M, Dožudić GJ, Selaković V, Stojković T, Puškaš N, Zaletel I, Živković M, Dragutinović V, Nikolić T, Jelenković A, Djorović D, Mirčić A, Petronijević ND (2018) Effects of vitamin D3 on the NADPH oxidase and matrix metalloproteinase in an animal model of global cerebral ischemia. Oxidative Med Cell Longev 2018:3273654
World Health Organization (WHO) (2018a) Depression. http://www.who.int/en/news-room/fact-sheets/detail/depression. Accessed 03 october 2018
World Health Organization (WHO) (2018b) Mental Health Gap Action Programme (mhGAP) Intervention Guide. http://apps.who.int/iris/bitstream/handle/10665/250239/9789241549790-eng.pdf;jsessionid=F7475276A6CADF44441D68B715E1B4DC?sequence=1. Accessed 03 october 2018
Wrzosek M, Łukaszkiewicz J, Wrzosek M, Jakubczyk A, Matsumoto H, Piątkiewicz P et al (2013) Vitamin D and the central nervous system. Pharmacol Rep 65:271–278
Yalcin I, Belzung C, Surget A (2008) Mouse strain differences in the unpredictable chronic mild stress: a four-antidepressant survey. Behav Brain Res 193:140–143
Yang L, Wu L, Du S, Hu Y, Fan Y, Ma J (2016) 1,25(OH)2D3 inhibits high glucose-induced apoptosis and ROS production in human peritoneal mesothelial cells via the MAPK/P38 pathway. Mol Med Rep 14:839–844
Yau WY, Chan MC, Wing YK, Lam HB, Lin W, Lam SP, Lee CP (2014) Noncontinuous use of antidepressant in adults with major depressive disorders – a retrospective cohort study. Brain Behav 4:390–397
Zhang Y, Su WJ, Chen Y, Wu TY, Gong H, Shen XL, Wang YX, Sun XJ, Jiang CL (2016) Effects of hydrogen-rich water on depressive-like behavior in mice. Sci Rep 6:2374
Zhao Y, Ma R, Shen J, Su H, Xing D, Du L (2008) A mouse model of depression induced by repeated corticosterone injections. Eur J Pharmacol 581:113–120
Zhao J, Jung YH, Jin Y, Kang S, Jang CG, Lee J (2019) A comprehensive metabolomics investigation of hippocampus, serum, and feces affected by chronic fluoxetine treatment using the chronic unpredictable mild stress mouse model of depression. Sci Rep 9(1):7566
Author contribution statement
MM and ALSR conceived and designed research. SVSS, PBR, VBN, and MM conducted experiments. SVSS, PBR, JDM, and MM analyzed data. SVSS, JDM, and MM wrote the manuscript. All authors read and approved the manuscript.
Funding
This study was financially supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [grant numbers 150082/2018-5 and 310113/2017-2], and Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES). ALSR is CNPq Research Fellow.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted (Ethics Committee CEUA PP00795).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva Souza, S.V., da Rosa, P.B., Neis, V.B. et al. Effects of cholecalciferol on behavior and production of reactive oxygen species in female mice subjected to corticosterone-induced model of depression. Naunyn-Schmiedeberg's Arch Pharmacol 393, 111–120 (2020). https://doi.org/10.1007/s00210-019-01714-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-019-01714-2