Abstract
Purpose
To elucidate the effects of various tibial and femoral attachment locations on the theoretical length changes and isometry of PCL grafts in healthy knees during in vivo weightbearing motion.
Methods
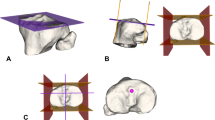
The intact knees of 14 patients were imaged using a combined magnetic resonance and dual fluoroscopic imaging technique while the patient performed a quasi-static lunge (0°–120° of flexion). The theoretical end-to-end distances of the 3-dimensional wrapping paths between 165 femoral attachments, including the anatomic anterolateral bundle (ALB), central attachment and posteromedial bundle (PMB) of the PCL, connected to an anterolateral, central, and posteromedial tibial attachment were simulated and measured. A descriptive heatmap was created to demonstrate the length changes on the medial condyle and formal comparisons were made between the length changes of the anatomic PCL and most isometric grafts.
Results
The most isometric graft, with approximately 3% length change between 0° and 120° of flexion, was located proximal to the anatomic femoral PCL attachments. Grafts with femoral attachments proximal to the isometric zone decreased in length with increasing flexion angles, whereas grafts with more distal attachments increased in length with increasing flexion angles. The ALB and central single-bundle graft demonstrated a significant elongation from 0° to 120° of flexion (p < 0.001). The PMB decreased in length between 0° and 60° of flexion after which the bundle increased in length to its maximum length at 120° (p < 0.001). No significant differences in length changes were found between either the ALB or PMB and the central graft, and between the ALB and PMB at flexion angles ≥ 60° (n.s.).
Conclusions
The most isometric attachment was proximal to the anatomic PCL footprint and resulted in non-physiological length changes. Moving the femoral attachment locations of the PCL significantly affected length change patterns, whereas moving the tibia locations did not. The importance of anatomically positioned (i.e., distal to the isometric area) femoral PCL reconstruction locations to replicate physiological length changes is highlighted. These data can be used to optimize tunnel positioning in either single- or double-bundle and primary or revision PCL reconstruction cases.
Level of evidence
IV.





Similar content being viewed by others
References
Abrahams M (1967) Mechanical behaviour of tendon in vitro. Med Biol Eng 5:433–443
Ahmad CS, Cohen ZA, Levine WN, Gardner TR, Ateshian GA, Mow VC (2003) Codominance of the individual posterior cruciate ligament bundles. An analysis of bundle lengths and orientation. Am J Sports Med 31:221–225
Amis AA, Gupte CM, Bull AM, Edwards A (2006) Anatomy of the posterior cruciate ligament and the meniscofemoral ligaments. Knee Surg Sports Traumatol Arthrosc 14:257–263
Bach BR, Daluga DJ, Mikosz R, Andriacchi TP, Seidl R (1992) Force displacement characteristics of the posterior cruciate ligament. Am J Sports Med 20:67–71 (discussion 71–72)
Bernard M, Hertel P, Hornung H, Cierpinski T (1997) Femoral insertion of the ACL. Radiographic quadrant method. Am J Knee Surg 10:14–21 (discussion 21–12)
Burns WC 2nd, Draganich LF, Pyevich M, Reider B (1995) The effect of femoral tunnel position and graft tensioning technique on posterior laxity of the posterior cruciate ligament-reconstructed knee. Am J Sports Med 23:424–430
Butler DL, Grood ES, Noyes FR, Zernicke RF (1978) Biomechanics of ligaments and tendons. Exerc Sport Sci Rev 6:125–181
Chan YS, Yang SC, Chang CH, Chen AC, Yuan LJ, Hsu KY et al (2006) Arthroscopic reconstruction of the posterior cruciate ligament with use of a quadruple hamstring tendon graft with 3- to 5-year follow-up. Arthroscopy 22:762–770
Defrate LE, Papannagari R, Gill TJ, Moses JM, Pathare NP, Li G (2006) The 6 degrees of freedom kinematics of the knee after anterior cruciate ligament deficiency: an in vivo imaging analysis. Am J Sports Med 34:1240–1246
DeFrate LE, van der Ven A, Gill TJ, Li G (2004) The effect of length on the structural properties of an Achilles tendon graft as used in posterior cruciate ligament reconstruction. Am J Sports Med 32:993–997
Galloway MT, Grood ES, Mehalik JN, Levy M, Saddler SC, Noyes FR (1996) Posterior cruciate ligament reconstruction. An in vitro study of femoral and tibial graft placement. Am J Sports Med 24:437–445
Giffin JR, Stabile KJ, Zantop T, Vogrin TM, Woo SL, Harner CD (2007) Importance of tibial slope for stability of the posterior cruciate ligament deficient knee. Am J Sports Med 35:1443–1449
Gill TJ, Van de Velde SK, Wing DW, Oh LS, Hosseini A, Li G (2009) Tibiofemoral and patellofemoral kinematics after reconstruction of an isolated posterior cruciate ligament injury: in vivo analysis during lunge. Am J Sports Med 37:2377–2385
Grood ES, Hefzy MS, Lindenfield TN (1989) Factors affecting the region of most isometric femoral attachments. Part I: the posterior cruciate ligament. Am J Sports Med 17:197–207
Jain V, Goyal A, Mohindra M, Kumar R, Joshi D, Chaudhary D (2016) A comparative analysis of arthroscopic double-bundle versus single-bundle posterior cruciate ligament reconstruction using hamstring tendon autograft. Arch Orthop Trauma Surg 136:1555–1561
Johannsen AM, Anderson CJ, Wijdicks CA, Engebretsen L, LaPrade RF (2013) Radiographic landmarks for tunnel positioning in posterior cruciate ligament reconstructions. Am J Sports Med 41:35–42
Kennedy NI, LaPrade RF, Goldsmith MT, Faucett SC, Rasmussen MT, Coatney GA et al (2014) Posterior cruciate ligament graft fixation angles, part 1: biomechanical evaluation for anatomic single-bundle reconstruction. Am J Sports Med 42:2338–2345
Kennedy NI, LaPrade RF, Goldsmith MT, Faucett SC, Rasmussen MT, Coatney GA et al (2014) Posterior cruciate ligament graft fixation angles, part 2: biomechanical evaluation for anatomic double-bundle reconstruction. Am J Sports Med 42:2346–2355
Kennedy NI, Wijdicks CA, Goldsmith MT, Michalski MP, Devitt BM, Aroen A et al (2013) Kinematic analysis of the posterior cruciate ligament, part 1: the individual and collective function of the anterolateral and posteromedial bundles. Am J Sports Med 41:2828–2838
Kim SJ, Jung M, Moon HK, Kim SG, Chun YM (2011) Anterolateral transtibial posterior cruciate ligament reconstruction combined with anatomical reconstruction of posterolateral corner insufficiency: comparison of single-bundle versus double-bundle posterior cruciate ligament reconstruction over a 2- to 6-year follow-up. Am J Sports Med 39:481–489
Kozanek M, Fu EC, Van de Velde SK, Gill TJ, Li G (2009) Posterolateral structures of the knee in posterior cruciate ligament deficiency. Am J Sports Med 37:534–541
Lee DY, Park YJ, Kim DH, Kim HJ, Nam DC, Park JS et al (2017) The role of isolated posterior cruciate ligament reconstruction in knees with combined posterior cruciate ligament and posterolateral complex injury. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-017-4672-x
Li G, DeFrate LE, Sun H, Gill TJ (2004) In vivo elongation of the anterior cruciate ligament and posterior cruciate ligament during knee flexion. Am J Sports Med 32:1415–1420
Li G, Gill TJ, DeFrate LE, Zayontz S, Glatt V, Zarins B (2002) Biomechanical consequences of PCL deficiency in the knee under simulated muscle loads—an in vitro experimental study. J Orthop Res 20:887–892
Li G, Papannagari R, Li M, Bingham J, Nha KW, Allred D et al (2008) Effect of posterior cruciate ligament deficiency on in vivo translation and rotation of the knee during weightbearing flexion. Am J Sports Med 36:474–479
Li G, Van de Velde SK, Bingham JT (2008) Validation of a non-invasive fluoroscopic imaging technique for the measurement of dynamic knee joint motion. J Biomech 41:1616–1622
Li G, Wuerz TH, DeFrate LE (2004) Feasibility of using orthogonal fluoroscopic images to measure in vivo joint kinematics. J Biomech Eng 126:314–318
Logan M, Williams A, Lavelle J, Gedroyc W, Freeman M (2004) The effect of posterior cruciate ligament deficiency on knee kinematics. Am J Sports Med 32:1915–1922
MacGillivray JD, Stein BE, Park M, Allen AA, Wickiewicz TL, Warren RF (2006) Comparison of tibial inlay versus transtibial techniques for isolated posterior cruciate ligament reconstruction: minimum 2-year follow-up. Arthroscopy 22:320–328
Mariani PP, Adriani E, Santori N, Maresca G (1997) Arthroscopic posterior cruciate ligament reconstruction with bone-tendon-bone patellar graft. Knee Surg Sports Traumatol Arthrosc 5:239–244
Markolf KL, Feeley BT, Jackson SR, McAllister DR (2006) Where should the femoral tunnel of a posterior cruciate ligament reconstruction be placed to best restore anteroposterior laxity and ligament forces? Am J Sports Med 34:604–611
Markolf KL, McAllister DR, Young CR, McWilliams J, Oakes DA (2003) Biomechanical effects of medial-lateral tibial tunnel placement in posterior cruciate ligament reconstruction. J Orthop Res 21:177–182
Norbakhsh ST, Zafarani Z, Najafi A, Aslani H (2014) Arthroscopic posterior cruciate ligament reconstruction by using hamstring tendon autograft and transosseous screw fixation: minimal 3 years follow-up. Arch Orthop Trauma Surg 134:1723–1730
Noyes FR, Barber-Westin SD (2005) Posterior cruciate ligament revision reconstruction, part 1: causes of surgical failure in 52 consecutive operations. Am J Sports Med 33:646–654
Ogata K, McCarthy JA (1992) Measurements of length and tension patterns during reconstruction of the posterior cruciate ligament. Am J Sports Med 20:351–355
Osti M, Tschann P, Kunzel KH, Benedetto KP (2012) Anatomic characteristics and radiographic references of the anterolateral and posteromedial bundles of the posterior cruciate ligament. Am J Sports Med 40:1558–1563
Papannagari R, DeFrate LE, Nha KW, Moses JM, Moussa M, Gill TJ et al (2007) Function of posterior cruciate ligament bundles during in vivo knee flexion. Am J Sports Med 35:1507–1512
Parolie JM, Bergfeld JA (1986) Long-term results of nonoperative treatment of isolated posterior cruciate ligament injuries in the athlete. Am J Sports Med 14:35–38
Patel DV, Allen AA, Warren RF, Wickiewicz TL, Simonian PT (2007) The nonoperative treatment of acute, isolated (partial or complete) posterior cruciate ligament-deficient knees: an intermediate-term follow-up study. HSS J 3:137–146
Petersen W, Lenschow S, Weimann A, Strobel MJ, Raschke MJ, Zantop T (2006) Importance of femoral tunnel placement in double-bundle posterior cruciate ligament reconstruction: biomechanical analysis using a robotic/universal force-moment sensor testing system. Am J Sports Med 34:456–463
Race A, Amis AA (1998) PCL reconstruction. In vitro biomechanical comparison of ‘isometric’ versus single and double-bundled ‘anatomic’ grafts. J Bone Joint Surg Br 80:173–179
Sekiya JK, West RV, Ong BC, Irrgang JJ, Fu FH, Harner CD (2005) Clinical outcomes after isolated arthroscopic single-bundle posterior cruciate ligament reconstruction. Arthroscopy 21:1042–1050
Shearn JT, Grood ES, Noyes FR, Levy MS (2004) Two-bundle posterior cruciate ligament reconstruction: how bundle tension depends on femoral placement. J Bone Joint Surg Am 86-a:1262–1270
Sidles JA, Larson RV, Garbini JL, Downey DJ, Matsen FA 3rd (1988) Ligament length relationships in the moving knee. J Orthop Res 6:593–610
Song EK, Park HW, Ahn YS, Seon JK (2014) Transtibial versus tibial inlay techniques for posterior cruciate ligament reconstruction: long-term follow-up study. Am J Sports Med 42:2964–2971
Strobel MJ, Weiler A, Schulz MS, Russe K, Eichhorn HJ (2003) Arthroscopic evaluation of articular cartilage lesions in posterior-cruciate-ligament-deficient knees. Arthroscopy 19:262–268
Tajima G, Nozaki M, Iriuchishima T, Ingham SJ, Shen W, Smolinski P et al (2009) Morphology of the tibial insertion of the posterior cruciate ligament. J Bone Joint Surg Am 91:859–866
Taylor KA, Cutcliffe HC, Queen RM, Utturkar GM, Spritzer CE, Garrett WE et al (2013) In vivo measurement of ACL length and relative strain during walking. J Biomech 46:478–483
Torg JS, Barton TM, Pavlov H, Stine R (1989) Natural history of the posterior cruciate ligament-deficient knee. Clin Orthop Relat Res 208–216
Trus P, Petermann J, Gotzen L (1994) Posterior cruciate ligament (PCL) reconstruction—an in vitro study of isometry. Part I. Tests using a string linkage model. Knee Surg Sports Traumatol Arthrosc 2:100–103
Van de Velde SK, Bingham JT, Gill TJ, Li G (2009) Analysis of tibiofemoral cartilage deformation in the posterior cruciate ligament-deficient knee. J Bone Joint Surg Am 91:167–175
Van de Velde SK, DeFrate LE, Gill TJ, Moses JM, Papannagari R, Li G (2007) The effect of anterior cruciate ligament deficiency on the in vivo elongation of the medial and lateral collateral ligaments. Am J Sports Med 35:294–300
von Eisenhart-Rothe R, Lenze U, Hinterwimmer S, Pohlig F, Graichen H, Stein T et al (2012) Tibiofemoral and patellofemoral joint 3D-kinematics in patients with posterior cruciate ligament deficiency compared to healthy volunteers. BMC Musculoskelet Disord 13:231
Wang CJ, Chan YS, Weng LH, Yuan LJ, Chen HS (2004) Comparison of autogenous and allogenous posterior cruciate ligament reconstructions of the knee. Injury 35:1279–1285
Wijdicks CA, Kennedy NI, Goldsmith MT, Devitt BM, Michalski MP, Aroen A et al (2013) Kinematic analysis of the posterior cruciate ligament, part 2: a comparison of anatomic single- versus double-bundle reconstruction. Am J Sports Med 41:2839–2848
Wong T, Wang CJ, Weng LH, Hsu SL, Chou WY, Chen JM et al (2009) Functional outcomes of arthroscopic posterior cruciate ligament reconstruction: comparison of anteromedial and anterolateral trans-tibia approach. Arch Orthop Trauma Surg 129:315–321
Wu CH, Chen AC, Yuan LJ, Chang CH, Chan YS, Hsu KY et al (2007) Arthroscopic reconstruction of the posterior cruciate ligament by using a quadriceps tendon autograft: a minimum 5-year follow-up. Arthroscopy 23:420–427
Funding
This work was supported by the National Institutes of Health (NIH) Grant R-01 AR055612.
Author information
Authors and Affiliations
Contributions
WAK: Conception and design of the work, acquisition, analysis and interpretation of data, drafting the work, revising the work critically for important intellectual content, final approval of the version to be published. AJTJ: Conception and design of the work, acquisition of data, revising the work critically for important intellectual content, final approval of the version to be published. NHV: Analysis and interpretation of data, revising the work critically for important intellectual content, final approval of the version to be published. ERAA: Conception and design of the work, acquisition, interpretation of data, revising the work critically for important intellectual content, final approval of the version to be published. RGHHN: Conception and design of the work, acquisition, interpretation of data, revising the work critically for important intellectual content, final approval of the version to be published. PDA: Conception and design of the work, interpretation of data, revising the work critically for important intellectual content, final approval of the version to be published. RFL: Conception and design of the work, interpretation of data, revising the work critically for important intellectual content, final approval of the version to be published. SKV: Conception and design of the work, and interpretation of data for the work, revising the work critically for important intellectual content, final approval of the version to be published. GL: Conception and design of the work, interpretation of data for the work, revising the work critically for important intellectual content, final approval of the version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest related to the submitted work.
Ethical approval
Study protocol was approved by the Partners Human Research Committee (Protocol #: 2003P000337/PHS).
Electronic supplementary material
Below is the link to the electronic supplementary material.
167_2018_5233_MOESM1_ESM.mov
Lateral view of a 3D femur model in 90° of flexion, with the quadrant applied as described by Bernard et al [5]. The “heat map” illustrates the normalized graft length changes (relative to their length at 60° of flexion, i.e., zero point) of the 165 theoretical grafts when connected to the central tibial attachment for the intact knees during the quasi-static lunge (0°–120° of flexion). Attachments located proximal to the isometric zone resulted in decreased graft lengths with increasing flexion angles, whereas distal grafts increased in length with increasing flexion angles. Dashed lines show the outline of the anatomical posterior cruciate ligament footprint. The black x on the femur shows the center of the anterolateral bundle; the black dot shows the center of the posteromedial bundle (MOV 5316 KB)
Rights and permissions
About this article
Cite this article
Kernkamp, W.A., Jens, A.J.T., Varady, N.H. et al. Anatomic is better than isometric posterior cruciate ligament tunnel placement based upon in vivo simulation. Knee Surg Sports Traumatol Arthrosc 27, 2440–2449 (2019). https://doi.org/10.1007/s00167-018-5233-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-018-5233-7