Abstract
Purpose
Septic shock is a leading cause of death among critically ill patients, in particular when complicated by acute kidney injury (AKI). Small experimental and human clinical studies have suggested that high-volume haemofiltration (HVHF) may improve haemodynamic profile and mortality. We sought to determine the impact of HVHF on 28-day mortality in critically ill patients with septic shock and AKI.
Methods
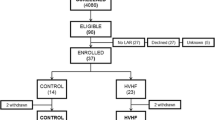
This was a prospective, randomized, open, multicentre clinical trial conducted at 18 intensive care units in France, Belgium and the Netherlands. A total of 140 critically ill patients with septic shock and AKI for less than 24 h were enrolled from October 2005 through March 2010. Patients were randomized to either HVHF at 70 mL/kg/h or standard-volume haemofiltration (SVHF) at 35 mL/kg/h, for a 96-h period.
Results
Primary endpoint was 28-day mortality. The trial was stopped prematurely after enrolment of 140 patients because of slow patient accrual and resources no longer being available. A total of 137 patients were analysed (two withdrew consent, one was excluded); 66 patients in the HVHF group and 71 in the SVHF group. Mortality at 28 days was lower than expected but not different between groups (HVHF 37.9 % vs. SVHF 40.8 %, log-rank test p = 0.94). There were no statistically significant differences in any of the secondary endpoints between treatment groups.
Conclusions
In the IVOIRE trial, there was no evidence that HVHF at 70 mL/kg/h, when compared with contemporary SVHF at 35 mL/kg/h, leads to a reduction of 28-day mortality or contributes to early improvements in haemodynamic profile or organ function. HVHF, as applied in this trial, cannot be recommended for treatment of septic shock complicated by AKI.


Similar content being viewed by others
References
Angus DC, Linde-Zwirble WT, Lidicker J et al (2001) Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 29:1303–1310
Bagshaw SM, Uchino S, Bellomo R et al (2007) Septic acute kidney injury in critically ill patients: clinical characteristics and outcomes. Clin J Am Soc Nephrol 2:431–439
Bernard GR, Vincent JL, Laterre PF et al (2001) Efficacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med 344:699–709
Opal SM, Laterre P-F, Francois B et al (2013) Effect of eritoran, an antagonist of MD2-TLR4, on mortality in patients with severe sepsis: the ACCESS randomized trial. JAMA J Am Med Assoc 309:1154–1162
Finfer S, Chittock DR, Su SY-S et al (2009) Intensive versus conventional glucose control in critically ill patients. N Engl J Med 360:1283–1297
Hoffmann JN, Hartl WH, Deppisch R et al (1996) Effect of hemofiltration on hemodynamics and systemic concentrations of anaphylatoxins and cytokines in human sepsis. Intensive Care Med 22:1360–1367
Heering P, Morgera S, Schmitz FJ et al (1997) Cytokine removal and cardiovascular hemodynamics in septic patients with continuous venovenous hemofiltration. Intensive Care Med 23:288–296
Bellomo R, Tipping P, Boyce N (1993) Continuous veno-venous hemofiltration with dialysis removes cytokines from the circulation of septic patients. Crit Care Med 21:522–526
Honore PM, Joannes-Boyau O (2004) High volume hemofiltration (HVHF) in sepsis: a comprehensive review of rationale, clinical applicability, potential indications and recommendations for future research. Int J Artif Organs 27:1077–1082
Rimmelé T, Kellum JA (2012) High-volume hemofiltration in the intensive care unit: a blood purification therapy. Anesthesiology 116:1377–1387
Rogiers P, Zhang H, Smail N et al (1999) Continuous venovenous hemofiltration improves cardiac performance by mechanisms other than tumor necrosis factor-alpha attenuation during endotoxic shock. Crit Care Med 27:1848–1855
Yekebas EF, Eisenberger CF, Ohnesorge H et al (2001) Attenuation of sepsis-related immunoparalysis by continuous veno-venous hemofiltration in experimental porcine pancreatitis. Crit Care Med 29:1423–1430
Wang H, Zhang ZH, Yan XW et al (2005) Amelioration of hemodynamics and oxygen metabolism by continuous venovenous hemofiltration in experimental porcine pancreatitis. World J Gastroenterol 11:127–131
Rimmelé T, Assadi A, Cattenoz M et al (2009) High-volume haemofiltration with a new haemofiltration membrane having enhanced adsorption properties in septic pigs. Nephrol Dial Transplant 24:421–427
Bellomo R, Kellum JA, Gandhi CR et al (2000) The effect of intensive plasma water exchange by hemofiltration on hemodynamics and soluble mediators in canine endotoxemia. Am J Respir Crit Care Med 161:1429–1436
Honore PM, Jamez J, Wauthier M et al (2000) Prospective evaluation of short-term, high-volume isovolemic hemofiltration on the hemodynamic course and outcome in patients with intractable circulatory failure resulting from septic shock. Crit Care Med 28:3581–3587
Joannes-Boyau O, Rapaport S, Bazin R et al (2004) Impact of high volume hemofiltration on hemodynamic disturbance and outcome during septic shock. ASAIO J 50:102–109
Cornejo R, Downey P, Castro R et al (2006) High-volume hemofiltration as salvage therapy in severe hyperdynamic septic shock. Intensive Care Med 32:713–722
Cole L, Bellomo R, Hart G et al (2002) A phase II randomized, controlled trial of continuous hemofiltration in sepsis. Crit Care Med 30:100–106
Cole L, Bellomo R, Journois D et al (2001) High-volume haemofiltration in human septic shock. Intensive Care Med 27:978–986
Ratanarat R, Brendolan A, Piccinni P et al (2005) Pulse high-volume haemofiltration for treatment of severe sepsis: effects on hemodynamics and survival. Crit Care 9:R294–R302
Boussekey N, Chiche A, Faure K et al (2008) A pilot randomized study comparing high and low volume hemofiltration on vasopressor use in septic shock. Intensive Care Med 34:1646–1653
Uchino S, Bellomo R, Morimatsu H et al (2007) Continuous renal replacement therapy: a worldwide practice survey. The beginning and ending supportive therapy for the kidney (B.E.S.T. kidney) investigators. Intensive Care Med 33:1563–1570
Vesconi S, Cruz DN, Fumagalli R et al (2009) Delivered dose of renal replacement therapy and mortality in critically ill patients with acute kidney injury. Crit Care 13:R57
Lee K, Lee SR, Mun CH, Min BG (2008) Pulse push/pull hemodialysis: in vitro study on new dialysis modality with higher convective efficiency. Artif Organs 32:406–411
Churchwell MD, Pasko DA, Btaiche IF et al (2007) Trace element removal during in vitro and in vivo continuous haemodialysis. Nephrol Dial Transplant 22:2970–2977
Bouman CS, van Kan HJ, Koopmans RP et al (2006) Discrepancies between observed and predicted continuous venovenous hemofiltration removal of antimicrobial agents in critically ill patients and the effects on dosing. Intensive Care Med 32:2013–2019
Klouche K, Cavadore P, Portales P et al (2002) Continuous veno-venous hemofiltration improves hemodynamics in septic shock with acute renal failure without modifying TNFalpha and IL6 plasma concentrations. J Nephrol 15:150–157
Kellum JA, Venkataraman R (2002) Blood purification in sepsis: an idea whose time has come? Crit Care Med 30:1387–1388
Joannes-Boyau O, Honore PM, Boer W (2006) Hemofiltration: the case for removal of sepsis mediators from where they do harm. Crit Care Med 34:2244–2246
Levy MM, Fink MP, Marshall JC et al (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med 31:1250–1256
Bellomo R, Ronco C, Kellum JA et al (2004) Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 8:R204–R212
Pottecher T, Calvat S, Dupont H et al (2006) Haemodynamic management of severe sepsis: recommendations of the French Intensive Care Societies (SFAR/SRLF) Consensus Conference, 13 October 2005, Paris, France. Crit Care 10:311
Vincent JL, Moreno R, Takala J et al (1996) The SOFA (Sepsis-Related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22:707–710
Le Gall JR, Lemeshow S, Saulnier F (1993) A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 270:2957–2963
Shore S, Nelson DP, Pearl JM et al (2001) Usefulness of corticosteroid therapy in decreasing epinephrine requirements in critically ill infants with congenital heart disease. Am J Cardiol 88:591–594
Wernovsky G, Wypij D, Jonas RA et al (1995) Postoperative course and hemodynamic profile after the arterial switch operation in neonates and infants. A comparison of low-flow cardiopulmonary bypass and circulatory arrest. Circulation 92:2226–2235
Zuppa AF, Nadkarni V, Davis L et al (2004) The effect of a thyroid hormone infusion on vasopressor support in critically ill children with cessation of neurologic function. Crit Care Med 32:2318–2322
Tsujimoto H, Ono S, Hiraki S et al (2004) Hemoperfusion with polymyxin B-immobilized fibers reduced the number of CD16+ CD14+ monocytes in patients with septic shock. J Endotoxin Res 10:229–237
Cruz DN, Antonelli M, Fumagalli R et al (2009) Early use of polymyxin B hemoperfusion in abdominal septic shock: the EUPHAS randomized controlled trial. JAMA 301:2445–2452
Parrillo JE (1993) Pathogenetic mechanisms of septic shock. N Engl J Med 328:1471–1477
Metnitz PG, Krenn CG, Steltzer H et al (2002) Effect of acute renal failure requiring renal replacement therapy on outcome in critically ill patients. Crit Care Med 30:2051–2058
Schrier RW, Wang W (2004) Acute renal failure and sepsis. N Engl J Med 351:159–169
Pocock S (1996) Clinical trials: a practical approach. Wiley, Canada
Hernandez AV, Eijkemans MJ, Steyerberg EW (2006) Randomized controlled trials with time-to-event outcomes: how much does prespecified covariate adjustment increase power? Ann Epidemiol 16:41–48
Zhang P, Yang Y, Lv R et al (2012) Effect of the intensity of continuous renal replacement therapy in patients with sepsis and acute kidney injury: a single-center randomized clinical trial. Nephrol Dial Transpl 27:967–973
Ronco C, Bellomo R, Homel P et al (2000) Effects of different doses in continuous veno-venous haemofiltration on outcomes of acute renal failure: a prospective randomised trial. Lancet 356:26–30
Bellomo R, Cass A, Cole L et al (2009) Intensity of continuous renal-replacement therapy in critically ill patients. N Engl J Med 361:1627–1638
Palevsky PM, Zhang JH, O’Connor TZ et al (2008) Intensity of renal support in critically ill patients with acute kidney injury. N Engl J Med 359:7–20
Bellomo R, Lipcsey M, Calzavacca P et al (2013) Early acid-base and blood pressure effects of continuous renal replacement therapy intensity in patients with metabolic acidosis. Intensive Care Med 39:429–436
Demirjian S, Teo BW, Guzman JA et al (2011) Hypophosphatemia during continuous hemodialysis is associated with prolonged respiratory failure in patients with acute kidney injury. Nephrol Dial Transpl 26:3508–3514
Roberts DM, Roberts JA, Roberts MS et al (2012) Variability of antibiotic concentrations in critically ill patients receiving continuous renal replacement therapy: a multicentre pharmacokinetic study. Crit Care Med 40:1523–1528
Payen D, Mateo J, Cavaillon JM et al (2009) Impact of continuous venovenous hemofiltration on organ failure during the early phase of severe sepsis: a randomized controlled trial. Crit Care Med 37:803–810
Acknowledgments
A special thanks to the members of the Data Safety and Monitoring Committee (J.P. Pignon, Villejuif; F. Sztark, Bordeaux; G. Boulard, Bordeaux; L. Trinquard, Paris; G. Hanique, Brussels) who helped us to steer the study and gave valuable comments on the manuscript. Thanks to the Hospital Pharmacovigilance Unit which reviews all the serious adverse events for classification. A big thanks to all the participants and in particular the nurses that worked continuously for the success of this study. The study was supported by a grant from the French Health Ministry (Hospital Clinical Research Program—PHRC).
Conflicts of interest
Dr. Bagshaw is supported by a Canada Research Chair in Critical Care Nephrology and Clinical Investigator Award from Alberta Innovates—Health Solutions. Other authors do not have any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
On behalf of the IVOIRE Study Group.
Trial registration: ClinicalTrials.gov; Identifier: NCT00241228.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix: Participating centres and investigators
Appendix: Participating centres and investigators
University Hospital of Bordeaux, Hopital Haut-Lévèque (France): Joannes-Boyau, Fleureau, Dewitte, Coquin, Rozé, Janvier, Ouattara; St-Pierre Para-University Hospital (Belgium): Honoré; Hopital R. Boulin, Libourne (France): Grand, Gauche; University Hospital of Liege (Belgium): Canivet, Wiesen, Dubois; Hôpital Cardiovasculaire et Pneumologique Louis Pradel, Lyon (France): Flamens, Bastien; Clinique Bordeaux Nord Aquitaine, Bordeaux (France): Pujol, Perdrix, Clement; Hopital Européen Georges Pompidou (HEGP), Paris (France): Journois; Albert Michallon Hospital, Grenoble (France): Broux, Robin, Durand; Hôpital de Cavale Blanche, CHU de Brest (France): Floch; Hôpital Tivoli, Université Libre de Bruxelles, La Louvière (Belgium): Franck, Bouckaert; Cliniques de l’Europe-Site St Michel, Brussels (Belgium): Collin.
Rights and permissions
About this article
Cite this article
Joannes-Boyau, O., Honoré, P.M., Perez, P. et al. High-volume versus standard-volume haemofiltration for septic shock patients with acute kidney injury (IVOIRE study): a multicentre randomized controlled trial. Intensive Care Med 39, 1535–1546 (2013). https://doi.org/10.1007/s00134-013-2967-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-013-2967-z