Abstract
Aims/hypothesis
HbA1c, expressed as the percentage of adult haemoglobin that is glycated, is the most widely used measure of chronic glycaemia. Achieving near-normal HbA1c levels has been shown to reduce long-term complications and the HbA1c assay is recommended to determine whether treatment is adequate and to guide adjustments. However, daily adjustments of therapy are guided by capillary glucose levels (mmol/l). We determined the relationship between an accurate measure of mean glucose levels over time and the HbA1c level, and whether HbA1c can be expressed in the same units as self-monitoring results.
Methods
Twenty-two participants with diabetes and three non-diabetic participants were included in this longitudinal observational study. Mean glucose levels were measured by continuous glucose monitoring (CGM), which measures interstitial glucose levels every 5 min, for 12 weeks. Capillary measurements were obtained four times per day to confirm the accuracy of CGM. HbA1c was measured at baseline and every 4 weeks.
Results
The HbA1c results at weeks 8 and 12 correlated strongly (r = 0.90) with the CGM results during the preceding 8 and 12 weeks. A curvilinear (exponential) relationship and a linear regression captured the relationship with similarly high correlations, which allowed transformation of HbA1c values to a calculated mean glucose level.
Conclusions and interpretation
HbA1c correlates closely with a complete measure of average glycaemia over the preceding 8–12 weeks. The translation of HbA1c to an average glucose level for reporting and management purposes is feasible.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The HbA1c assay has become the most commonly used measure of chronic glycaemia in epidemiological studies, clinical trials and the management of diabetes since its introduction more than 25 years ago [1–3]. The concentration of HbA1c, formed through the non-enzymatic attachment of glucose to haemoglobin, is commonly considered to reflect the integrated mean glucose level over the previous 8–12 weeks, the time period being dictated by the 120 day lifespan of the erythrocyte. The relationship between mean blood glucose and HbA1c has been suggested by old studies that used a variety of measures of outpatient and inpatient plasma and capillary glucose concentrations, all of which suffered from relatively infrequent sampling of glucose levels, infrequent sampling of HbA1c, or both [4–8]. Arguably, the two most convincing studies measured four to six fingerstick blood glucose levels per day for 6–8 weeks and compared the calculated mean blood glucose levels with the HbA1c levels measured at the end of the study period [9, 10]. The clinical role of the assay was subsequently established by demonstrating that it provided information regarding mean blood glucose that could not be provided by any other available measures [10]. The pre-eminent role of the assay was cemented by the Diabetes Control and Complications Trial (DCCT), and others, which used the assay to measure chronic glycaemia and demonstrated its central role in the pathogenesis of long-term complications [11–13]. The recommended goals for metabolic control of diabetes are based on the HbA1c assay [2, 14, 15], the vast majority of methods now being standardised to the DCCT assay through an international programme [16].
Despite the popularity and central role of the HbA1c assay, its true relationship to mean blood glucose remains poorly understood. HbA1c appears to reach a new steady state 3–4 months after glucose changes [17]. All of the previous studies that suggested a relationship between HbA1c level and mean blood glucose [4–10] used relatively infrequent monitoring during the daytime, which is prone to sampling error, and did not capture glucose levels overnight or true 24 h glycaemia. In addition, the limited time period of the previous studies and limited number of HbA1c assays performed precluded an analysis of the time period of mean glycaemia that the HbA1c reflects best.
We used continuous glucose monitoring (CGM), which measures interstitial glucose levels every 5 min, for 3 months in patients with diabetes with relatively stable glycaemia, and in non-diabetic individuals, to determine true mean glycaemia. HbA1c assays were measured monthly and best-fit correlations were determined over time.
Methods
Participants
Patients with type 1 (n = 15) or type 2 (n = 7) diabetes mellitus were recruited from the Massachusetts General Hospital Diabetes Center. Three non-diabetic volunteers were also recruited. The diabetic participants were required to have relatively stable glycaemic control, manifest by at least two HbA1c assay results in the previous 6 months that were no more than 1% different. We tried to recruit approximately similar numbers of diabetic participants with HbA1c <7, 7–8.5 and >8.5%. The non-diabetic participants had no history of diabetes and had HbA1c levels in the non-diabetic range (<6.1%) in the assays used for the study. Exclusion criteria included any condition that would probably cause a change in glucose control during the 3 month period of study or that would change erythrocyte turnover. Potential participants with known haemoglobinopathies were excluded to avoid any possible interference with the HbA1c assays [18]. All participants signed an informed consent form approved by the Human Research Committee.
Measurements
CGM was performed with the Continuous Glucose Monitoring System (CGMS Medtronic Minimed, Northridge, CA, USA), which measures interstitial glucose levels every 5 min through a catheter inserted subcutaneously. Participants changed the sensor units every 3 days, as instructed by the manufacturer, and returned the monitors to the Diabetes Center every 2 weeks so that they could be downloaded. Diabetes therapy was not adjusted on the basis of the CGM data, unless otherwise unrecognised periods of hypoglycaemia, such as periods of nocturnal glucose levels <3.33 mmol/l, were noted. When this occurred, the patient’s physician was notified so that appropriate treatment changes could be made. In addition to the CGM, participants were requested to perform self-monitoring of capillary glucose (OneTouch Ultra; Lifescan, Milipitas, CA, USA) at least four times per day, usually before meals and at bedtime. These measurements were used to calibrate the CGMS and adjust therapy, and provided an independent measure of glycaemia. Capillary glucose levels are calibrated by Lifescan to provide values equivalent to plasma glucose levels. Glucose levels were also measured with a laboratory-based hexokinase assay (Hitachi Model 917 autoanalyser; Hitachi, Tokyo, Japan) at each 2 week visit to the Diabetes Center, contemporaneously with a self-glucose monitoring test. The capillary glucose values correlated closely with the simultaneously measured laboratory values (n = 146 pairs, capillary value = 0.8876 × laboratory value + 21.4, R 2 = 0.9629, p < 0.001), validating the accuracy of the self-monitoring. The mean absolute difference between the self-monitored and laboratory results was <0.11 mmol/l.
Two different assays were used to measure HbA1c: an HPLC method that is the primary reference method for the National Glycohemoglobin Standardization Program (NGSP) [15] and a boronate affinity assay (Primus PDQ, Primus Diagnostics, Kansas City, MO, USA) that measures HbA1c independently of any haemoglobin abnormalities. Because of technical issues (modestly elevated fetal haemoglobin levels in five participants and missing samples at several time points for two participants), several participants’ samples could not be assayed accurately with the NGSP reference assay [18]. Therefore, the primary results presented here are from the boronate affinity assay. Both HbA1c assays have CVs <2% at low (5%) and high (10%) HbA1c values in our laboratory. Long-term calibration standards are used to prevent assay drift in the NGSP HPLC method and the boronate affinity assay is calibrated against it. The correlation of the boronate affinity assay with the primary HPLC assay is r = 0.99 (p < 0.001).
Statistical analyses
The demographic data are expressed as mean ± SD. Correlations between mean glucose levels derived from the CGM results and HbA1c levels were analysed using linear regression, exponential and quadratic equations, with Spearman correlations. The best fits, judged by the r values closest to 1.0, were determined. Analyses were performed using Stata statistical software (Stata Statistical Software Release 8.0, 2003; Stata Corporation, College Station, TX, USA).
Results
Twenty-nine eligible volunteers were recruited to the study. Three volunteers withdrew within the first few weeks because of their inability to carry out various elements of the study. A fourth volunteer was hospitalised for medical reasons unrelated to the study and could not complete the study in a timely fashion. Baseline characteristics of the 25 participants who completed the 12 week study are shown in Table 1.
The participants had HbA1c measured four times, at baseline and monthly for 3 months. HbA1c levels during the study changed minimally from baseline (7.39 ± 1.4%) to study end (7.16 ± 1.3%), with the greatest absolute difference <1%. CGM was performed on average for 81.8 ± 4 days of the 12-week period for the diabetic participants, representing 97% of the possible 84 day period and approximately 24,000 glucose measurements per participant. Self-monitoring by capillary fingerstick was performed an average of 3.73 ± 1.93 times per day by the diabetic participants and 2.68 ± 1.08 times per day by the non-diabetic participants. Almost 6000 paired (simultaneous capillary and interstitial CGMS tests) assays were performed.
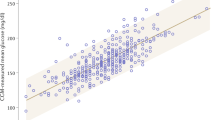
The correlations between mean CGM glucose levels and HbA1c levels at the different time points during the study are shown in Table 2. The strongest correlations (Spearman r = 0.89–90, p < 0.001) were between the mean CGM glucose levels measured during the entire 12-week period and the HbA1c level at weeks 8 or 12.
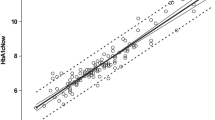
The linear and exponential models provided similarly close correlations between the CGM during the 12 weeks of the study and the HbA1c value at 12 weeks: linear model, mean CGM concentration (mean CGM) = HbA1c × 1.75–3.81 (r = 0.89, p < 0.001) (Fig. 1a); exponential model, mean CGM = 1.28HbA1c + 0.000136exp(HbA1c)–0.92 (r = 0.89, p < 0.001) (Fig. 1b). The correlations of mean CGM levels with the HbA1c values assayed with the NGSP reference HPLC method had an r value for the linear regression of 0.90 (p < 0.001).
Relationship between HbA1c at month 3 and mean glucose level calculated from CGM during 12 previous weeks according to (a) linear regression mean CGM = HbA1c × 1.75–3.81 (r = 0.89, p < 0.001), and (b) the exponential mean CGM = 1.28HbA1c + 0.000136exp(HbA1c)–0.92 (r = 0.89, p < 0.001). Continuous line, fitted values; diamonds, observed values
The HbA1c values were transformed into their equivalent mean CGM values using the exponential and linear regression equations (Table 3). Similar transformations with the HPLC assay results provided similar mean glucose values (not shown).
Discussion
The HbA1c assay is widely accepted as the best means of retrospectively capturing mean glycaemia. It is the basis of treatment guidelines and is used universally to adjust therapy [2, 14, 15]. In order for patients to achieve HbA1c goals, they adjust their day-to-day therapies based on fingerstick capillary glucose levels, measured with meters that are adjusted to provide values comparable to venous plasma levels [3]. However, the relationship between HbA1c values and mean glucose levels has never been carefully explored, owing in great part to the absence of means to measure glucose levels frequently enough to reveal a complete description of mean glucose levels over time. Previous studies that have been performed have used relatively infrequent glucose testing and have not been able to measure mean blood glucose levels with confidence because of potential sampling errors [4–10]. For example, the DCCT data set included a seven-point glucose profile performed every 3 months [8]. The intra-individual variations in mean glucose profiles and HbA1c were 29.9 and 9.7% respectively, suggesting the relatively high degree of variability of glucose profiles collected intermittently, and reinforcing the need for frequent measurements to capture mean glycaemia.
We took advantage of the development of CGM devices, which measure interstitial glucose levels every 3–5 min. By using these devices over a 3 month period, we provided a more complete measure, including approximately 24,000 glucose tests per participant, of the average glucose levels in 25 diabetic and non-diabetic individuals and determined their relationship with HbA1c. Although some studies have suggested less than acceptable correlations between CGMS interstitial glucose results and venous plasma or capillary glucose levels, most recent studies, including the present study, have noted generally strong correlations [19, 20]. The results showed that HbA1c is tightly correlated with mean interstitial glucose levels, with exponential and linear regression equations providing similarly tight fits for the data. Previous studies with less detailed measures of chronic glucose values, including one using intermittent CGM, have also suggested a curvilinear relationship with HbA1c [9, 21]. The limited number of HbA1c assay results in the higher range limits our ability to choose between the linear and exponential models. Although the choice of research participants with stable HbA1c levels and the decision not to adjust therapy during the study limited our ability to examine the dynamics of glucose change and HbA1c levels, the strongest correlations between HbA1c and mean glucose levels appeared to be over the preceding 12 weeks, consistent with prior studies [17].
Of note, although there was less than a perfect correlation between the measured mean glucose levels and the HbA1c levels, the relatively high r values, especially given the limited sample size and range of values, suggests that there are probably no extraneous factors that affect the relationship between MBG and HbA1c in a substantive way. Some studies have suggested that variable red cell turnover, which may be affected by hyperglycaemia, may alter the relationship between mean blood glucose and HbA1c values [22]. Other studies have proposed that glycation rates may differ among individuals at the same mean glucose levels [23, 24]. However, the studies that have supported differential rates of glycation (high vs low glycators) have generally been performed in non-diabetic individuals with very limited glucose testing. A study by Hempe and colleagues in 128 children with type 1 diabetes suggested that differential glycation occurred since mean blood glucose levels during a 1 month period, assessed with approximately 80 self-monitored values, did not correlate tightly with the HbA1c at the end of the month (r = 0.71) [25]. However, the present study and others [17] strongly suggest that a 1-month period is inadequate for HbA1c to equilibrate and that if more complete glucose monitoring is performed (8000 assays in 1 month in our study compared with 80 in the Hempe study) the correlations become substantially tighter. The absence of any major outliers in the present study suggest that if differential glycation exists it is relatively rare, or does not affect the relationship between blood glucose values and HbA1c levels enough to disturb a predictable relationship between the two. However, considering the small sample size in the present study and the relatively homogeneous study population, most of the participants being white and with type 1 diabetes, larger studies using comparable assessment of chronic glycaemia will need to be performed to determine conclusively whether differential glycation exists.
The relatively small number of participants precludes any expansive conclusions regarding the relationship between HbA1c and mean glucose levels in the larger diabetic population. Similarly, the limited range of HbA1c levels in the present study (the highest HbA1c level was <11%) does not allow extrapolation of our results to patients with HbA1c levels that are very high. However, we are confident of our findings in the range of glycaemia studied, and have therefore restricted the conversion table (Table 3) to the values included in the study. While inclusion of patients with higher HbA1c levels will be required to determine the conversion equations covering a wider range of glycaemia, and whether an exponential model fits better than a linear one, the range of HbA1c levels in the present study applies to the vast majority of diabetic patients. Of note, similar correlations between average chronic glycaemia and HbA1c were obtained with all of the HbA1c assays performed; thus, the relationship is not limited to only one HbA1c assay.
The mathematical relationship established between HbA1c and mean blood glucose in this study should allow the expression of HbA1c as an equivalent mean glucose level. This is likely to be beneficial to patients and care providers alike as the assay of chronic glucose control would be in the same units as the patients’ self-monitoring. However, before this transformation can take place, the mathematical relationship between HbA1c and mean glucose levels should be confirmed in a larger study with a more diverse population to ensure that the relationship applies for all patients with diabetes. An international study that is examining the relationship between mean glucose levels and HbA1c levels in a large (n = 700), multinational study population including non-diabetic participants and type 1 and type 2 diabetic participants is ongoing. Like the present study, the international study is employing CGMS and frequent self-monitored capillary profiles to capture average glucose levels as completely as possible.
Abbreviations
- CGM:
-
continuous glucose monitoring
- CGMS:
-
Continuous Glucose Monitoring System
- MBG:
-
mean blood glucose concentration
- NGSP:
-
National Glycohemoglobin Standardization Program
References
Saudek CD, Derr RL, Kalyani RR (2006) Assessing glycemia in diabetes using self-monitoring blood glucose and hemoglobin A1c. JAMA 295:1688–1697
American Diabetes Association (2007) Standards of medical care of diabetes. Diabetes Care 30(Suppl 1):S4–S41
Goldstein DE, Little R, Lorenz RA et al (2004) Tests of glycemia in diabetes. Diabetes Care 27:1761–1773
Koenig RJ, Peterson CM, Jones RL, Saudek C, Lehrman M, Cerami A (1976) Correlation of glucose regulation and hemoglobin A1c in diabetes mellitus. N Engl J Med 295:417–420
Koenig RJ, Peterson CM, Kilo C, Cerami A, Williamson JR (1976) Hemoglobin A1c as an indicator of the degree of glucose intolerance in diabetes. Diabetes 24:230–232
Peterson CM, Jones RL, Dupuis A, Bernstein R, O’Shea M (1979) Feasibility of improved glucose control in patients with insulin dependent diabetes mellitus. Diabetes Care 2:329–335
Murata GH, Hoffman RM, Duckworth WC, Wendel CS, Shah JH (2004) Contributions of weekly mean blood glucose values to hemoglobin A1c in insulin-treated type 2 diabetes: the Diabetes Outcomes in Veterans Study (DOVES). Am J Med Sci 327:319–323
Rohlfing CL, Wiedmeyer HM, Little R, England JD, Tennill A, Goldstein DE (2002) Defining the relationship between plasma glucose and HbA1c in the diabetes control and complications trial. Diabetes Care 25:275–278
Svendsen PA, Lauritzen T, Soegaard U, Nerup J (1982) Glycosylated haemoglobin and steady-state mean blood glucose concentration in type I (insulin-dependent) diabetes. Diabetologia 23:403–405
Nathan DM, Singer DE, Hurxthal K, Goodson JD (1984) The clinical information value of the glycosylated hemoglobin assay. N Engl J Med 310:341–346
Diabetes Control and Complications Trial Research Group (1993) The effect of intensive diabetes treatment on the development and progression of long-term complications in insulin-dependent diabetes mellitus: the diabetes control and complications trial. N Engl J Med 329:978–986
DCCT Research Group (1995) The association between glycemic exposure and long-term diabetic complications in the diabetes control and complications trial. Diabetes 44:968–983
UK Prospective Diabetes Study Group (1988) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352:837–853
European Diabetes Policy Group (1999) A desk-top guide to type 2 diabetes mellitus. Diabet Med 16:716–730
Nathan DM, Buse JB, Davidson MB et al (2006) Management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy. Diabetelogia 49:1711–1721
Little RR, Rohlfing CL, Wiedmeyer H-M, Myers GL, Sacks DB, Goldstein DE (2001) The National Glycohemoglobin Standardization Program (NGSP): a five-year progress report. Clin Chem 47:1985–1992
Tahara Y, Shima K (1993) The response of GHb to stepwise plasma glucose over time in diabetic patients. Diabetes Care 9:1313–1314
Bry L, Chen PC, Sacks DB (2001) Effects of hemoglobin variants and chemically modified derivatives on assays for glycohemoglobin. Clin Chem 47:153–163
Djakoure-Platonoff C, Radermercker R, Reach G, Slama G, Selam J-L (2003) Accuracy of continuous glucose monitoring system in inpatient and outpatient conditions. Diabetes Metab 29:159–162
Clarke WL, Anderson S, Farhy L (2005) Evaluating the clinical accuracy of two continuous glucose sensors using continuous glucose-error grid analysis. Diabetes Care 28:2412–2417
Hassan Y, Johnson B, Nader N, Gannon MC, Nuttall FQ (2006) The relationship between 24-hour integrated glucose concentrations and % glycohemoglobin. J Lab Clin Med 147:21–26
Virtue MA, Furne JK, Nuttall FQ, Levitt MD (2004) Relationship between GHb concentration and erythrocyte survival determined from breath carbon monoxide concentration. Diabetes Care 27:931–935
Yudkin JS, Forrest RD, Jackson CA, Ryle AJ, Davie SJ, Gould BJ (1990) Unexplained variability of glycated hemoglobin in non-diabetic subjects not related to glycaemia. Diabetologia 33:208–215
Gould BJ, Davie SJ, Yudkin JS (1997) Investigation of the mechanism underlying the variability of glycated haemoglobin in non-diabetic subjects not related to glycaemia. Clin Chim Acta 260:49–64
Hempe JM, Gomez R, McCarter RJ, Chalew SA (2002) High and low hemoglobin glycation phenotypes in type 1 diabetes. A challenge for interpretation of glycemic control. J Diabetes Complications 16:313–320
Acknowledgements
This study was supported by an unrestricted research grant from NovoNordisk and by the contribution of supplies and equipment from Medtronic Minimed and Lifescan, Inc. D.M. Nathan is supported in part by the Earle P. Charlton Fund for Innovative Diabetes Research.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 2.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by-nc/2.0/.
About this article
Cite this article
Nathan, D.M., Turgeon, H. & Regan, S. Relationship between glycated haemoglobin levels and mean glucose levels over time. Diabetologia 50, 2239–2244 (2007). https://doi.org/10.1007/s00125-007-0803-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-007-0803-0