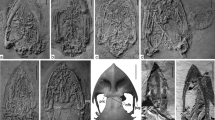
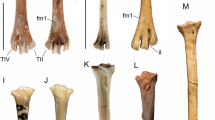
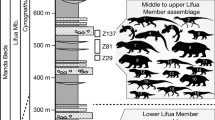
Abstract
The fossil record of Late Cretaceous–Paleogene modern birds in the Southern Hemisphere includes the Maastrichtian Neogaeornis wetzeli from Chile, Polarornis gregorii and Vegavis iaai from Antarctica, and Australornis lovei from the Paleogene of New Zealand. The recent finding of a new and nearly complete Vegavis skeleton constitutes the most informative source for anatomical comparisons among Australornis, Polarornis, and Vegavis. The present contribution includes, for the first time, Vegavis, Polarornis, and Australornis in a comprehensive phylogenetic analysis. This analysis resulted in the recognition of these taxa as a clade of basal Anseriformes that we call Vegaviidae. Vegaviids share a combination of characters related to diving adaptations, including compact and thickened cortex of hindlimb bones, femur with anteroposteriorly compressed and bowed shaft, deep and wide popliteal fossa delimited by a medial ridge, tibiotarsus showing notably proximally expanded cnemial crests, expanded fibular crest, anteroposterior compression of the tibial shaft, and a tarsometatarsus with a strong transverse compression of the shaft. Isolated bones coming from the Cretaceous and Paleogene of South America, Antarctica, and New Zealand are also referred to here to Vegaviidae and support the view that these basal anseriforms were abundant and diverse at high southern latitudes. Moreover, vegaviids represent the first avian lineage to have definitely crossed the K–Pg boundary, supporting the idea that some avian clades were not affected by the end Mesozoic mass extinction event, countering previous interpretations. Recognition of Vegaviidae indicates that modern birds were diversified in southern continents by the Cretaceous and reinforces the hypothesis indicating the important role of Gondwana for the evolutionary history of Anseriformes and Neornithes as a whole.



Similar content being viewed by others
References
Acosta Hospitaleche C, Gelfo JN (2015) New Antarctic findings of Upper Cretaceous and lower Eocene loons (Aves: Gaviiformes). In: Annales de Paléontologie (vol 101, no. 4, pp 315–324). Elsevier Masson, France
Agnolin FL, Ezcurra MD, Pais DF, Salisbury W (2010) A reappraisal of the Cretaceous nonavian dinosaur faunas from Australia and New Zealand, evidence for their Gondwanan affinities. J Syst Palaeontol 8:257–300
Bogan S, Agnolin FL, Novas FE (2016) New Selachian records from the Upper Cretaceous of Southern Patagonia, paleobiogeographical implications and the description of a new taxon. J. Vert. Paleont 36(3):e1105235
Bono RK, Clarke J, Tarduno JA, Brinkman D (2016) A large ornithurine bird (Tingmiatornis arctica) from the Turonian High Arctic: climatic and evolutionary implications. Sci Rep 6:38876
Bourdon E (2005) Osteological evidence for sister group relationship between pseudo-toothed birds (Aves: Odontopterygiformes) and waterfowls (Anseriformes). Naturwissenschaften 92(12):586–591
Case J, Tambussi CP (1999) Maastrichtian record of neornithine birds in Antarctica, comments on a Late Cretaceous radiation of modern birds. J. Vert. Paleont 19(3, Suppl):37A
Case JA, Woodburne MO, Chaney DS (1988) A new genus and species of polydolopid marsupial from the La Meseta Formation, Late Eocene, Seymour Island, Antarctic Peninsula. In: Feldmann RM, Woodburne MO (eds) Geology and paleontology of Seymour Island. Geological Society of America, Boulder, pp 505–521
Case J, Reguero M, Martin J, Cordes-Person A (2006) A cursorial bird from the Maastrichtian of Antarctica. J Vertebr Paleontol 26(3, Supplement):48A
Cenizo MM (2012) Review of the putative Phorusrhacidae from the Cretaceous and Paleogene of Antarctica: new records of ratites and pelagornithid birds. Pol Polar Res 33(3):239–258
Chatterjee S (2002) The morphology and systematics of Polarornis, a Cretaceous loon (Aves, Gaviidae) from Antarctica. In: Zhou Z, Zhang F (eds) Proceedings of the 5th Symposium of the Society of Avian Paleontology and Evolution. Science Press, Beijing, pp 125–155
Chatterjee S, Martinioni D, Novas F, Mussel F, Templin R (2006) A new fossil loon from the Late Cretaceous of Antarctica and early radiation of foot-propelled diving birds. J Vertebr Paleontol 26:49A
Chiappe LM (1996) Early avian evolution in the Southern Hemisphere, the fossil record of birds in the Mesozoic of Gondwana. Mem Queensland Mus 39:533–554
Chiappe LM (2016) Birds of stone, Chinese avian fossils from the age of dinosaurs. 304 pp. Johns Hopkins University Press, Baltimore
Chinsamy A (2002) Bone microstructure of early birds. In: Chiappe LM, Witmer LM (eds) Mesozoic birds, above the heads of dinosaurs. Univ. California Press, Berkeley, pp 421–431
Chinsamy A, Martin LD, Dodson P (1998) Bone microstructure of the diving Hesperornis and the volant Ichthyornis from the Niobrara Chalk of western Kansas. Cretac Res 19:225–235
Clarke JA, Tambussi CP, Noriega JI, Erickson GM, Ketcham RA (2005) Definitive fossil evidence for the extant avian radiation in the Cretaceous. Nature 433:305–308
Clarke JA, Chatterjee S, Li Z, Riede T, Agnolin F, Goller F, Novas FE (2016) Fossil evidence of the avian vocal organ from the Mesozoic. Nature 538(7626):502–505
Cooper A, Penny D (1997) Mass survival of birds across the Cretaceous–Tertiary boundary, Molecular evidence. Science 275:1109–1113
Cordes AH (2002) A new charadriiform avian specimen from the early Maastrichtian of Cape Lamb, Vega Island, Antarctic Peninsula. J Vertebr Paleontol 22:99A
Cracraft J (2001) Avian evolution, Gondwana biogeography, and the Cretaceous–Tertiary mass extinction event. Proc. Roy. Soc. London B 1268:459–469
Cracraft J, Clarke J (2001) The basal clades of modern birds. In: New perspectives on the origin and early evolution of birds: proceedings of the international symposium in honor of John H. Ostrom. Peabody Museum of Natural History, New Haven, pp 143–152
De Mendoza R, Tambussi C (2015) Osteosclerosis in the extinct Cayaoa bruneti (Aves, Anseriformes). Insights on behavior and flightlessness. Ameghiniana 52:305–313
De Pietri VL, Scofield RP, Zelenkov N, Boles WE, Worthy TH (2016) The unexpected survival of an ancient lineage of anseriform birds into the Neogene of Australia: the youngest record of Presbyornithidae. Open Sci 3(2):150635
Dzerzhinsky FY (1995) Evidence for common ancestry of the Galliformes and Anseriformes. Cour Forschungsinst Senck 181:325–336
Ericson PG (1997) Systematic relationships of the Palaeogene family Presbyornithidae (Aves: Anseriformes). Zool J Linnean Soc 121(4):429–483
Ericson PG, Anderson CL, Britton T, Elzanowski A, Johansson US, Källersjö M, Mayr G (2006) Diversification of Neoaves: integration of molecular sequence data and fossils. Biol Lett 2(4):543–547
Feduccia A (1999) The origin and evolution of birds. Yale University Press, New Haven 245 pp
Feduccia A (2003) “Big Bang” for Tertiary birds? Trends Ecol. Evolution 16:172–176
Feduccia A (2014) Avian extinction at the end of the Cretaceous: assessing the magnitude and subsequent explosive radiation. Cretac Res 50:1–15
Garcia Marsà JA, Agnolín FL, Novas F (2017) Bone microstructure of Vegavis iaai (Aves, Anseriformes) from the Upper Cretaceous of Vega Island, Antarctic Peninsula Historical Biology, 1–5
Hedges SB, Parker PH, Sibley CG, Kumar S (1996) Continental breakup and the ordinal diversification of birds and mammals. Nature 381(6579):226
Hope S (2002) The Mesozoic radiation of Neornithes. In: Chiappe LM, Witmer LM (eds) Mesozoic birds, above the heads of dinosaurs. Berkeley University Press, Berkeley, pp 168–218
Humphrey PS, Livezey BC (1982) Flightlessness in flying steamer-ducks. Auk 99:368–372
Ibañez B, Tambussi CP (2012) Foot-propelled aquatic birds, pelvic morphology and locomotor performance. Ital J Zool 79:356–362
Ksepka DT, Cracraft J (2008) An avian tarsometatarsus from near the KT boundary of New Zealand. J Vertebr Paleontol 28(4):1224–1227
Longrich N (2008) An ornithurine-dominated avifauna from the Belly River Group (Campanian, Upper Cretaceous) of Alberta, Canada. Cret Res 30:161–177
Longrich NR, Tokaryk T, Field DJ (2011) Mass extinction of birds at the Cretaceous–Paleogene (K–Pg) boundary. PNAS 108:15253–15257
Mayr G (2008) Phylogenetic affinities and morphology of the late Eocene anseriform bird Romainvillia stehlini Lebedinsky, 1927. Neues Jahrb Geol Paläontol-Abh 248(3):365–380
Mayr G (2009) Paleogene fossil birds. Springer, Berlin 262 pp
Mayr G (2011) Metaves, Mirandornithes, Strisores and other novelties—a critical review of the higher-level phylogeny of neornithine birds. J Zool Syst Evol Res 49(1):58–76
Mayr G, Clarke J (2003) The deep divergences of neornithine birds: a phylogenetic analysis of morphological characters. Cladistics 19:527–553
Mayr G, Poschmann M (2009) A loon leg (Aves, Gaviidae) with crocodilian tooth from the late Oligocene of Germany. Waterbirds 32(3):468–471
Mayr G, Scofield RP (2014) First diagnosable non-sphenisciform bird from the early Paleocene of New Zealand. J R Soc N Z 44(1):48–56
Mayr G, Zvonok E, Gorobets L (2013) The tarsometatarsus of the middle Eocene loon Colymbiculus udovichenkoi. In: Göhlich UB, Kroh A (eds) Paleornithological research 2013—proceedings of the 8th International Meeting of the Society of Avian Paleontology and Evolution. Natural History Museum Vienna, Vienna, pp 17–22 306 pp
Noriega JI, Tambussi CP (1995) A Late Cretaceous Presbyornithidae (Aves: Anseriformes) from Vega Island, Antarctic Peninsula: paleobiogeographic implications. Ameghiniana 32(1):57–61
Noriega JI, Tambussi CP, Cozzuol MA (2008) New material of Cayaoa bruneti Tonni, an early Miocene anseriform (Aves) from Patagonia, Argentina. Neues Jahrb Geol Paläontol-Abh 249(3):271–280
Novas FE, Cambiaso AV, Lirio JM, Núñez HJ (2002) Paleobiogeografía de los dinosaurios polares de Gondwana. Ameghiniana 39:15R
O'Connor PM, Forster CA (2010) A Late Cretaceous (Maastrichtian) avifauna from the Maevarano Formation, Madagascar. J Vertebr Paleontol 30(4):1178–1201
Olson SL (1992) Neogaeornis wetzeli Lambrecht, a Cretaceous loon from Chile (Aves, Gaviidae). J Vert Paleont 12:122–124
Olson SL, Feduccia A (1980) Presbyornis and the origin of the Anseriformes (Aves: Charadriomorphae) (No. 598.2 OLSp). Smithsonian Institution Press
Padian K, Chiappe LM (1998) The origin and early evolution of birds. Biol Rev 73(1):1–42
Padian K, de Ricqlés AJ, Horner JR (2001) Dinosaurian growth rates and bird origins. Nature 412:405–408
Reguero M, Goin F, Acosta Hospitaleche C, Dutra T, Marenssi S (2013) Late Cretaceous/Paleogene West Antarctica terrestrial biota and its intercontinental affinities, p 120
Rozadilla S, Agnolin FL, Novas FE, Aranciaga AR, Motta MJ, Lirio JM, Isasi MP (2016) A new ornithopod (Dinosauria, Ornithischia) from the Upper Cretaceous of Antarctica and its palaeobiogeographical implications. Cretac Res 57:311–324
Smith ND (2010) Phylogenetic analysis of Pelecaniformes (Aves) based on osteological data: implications for waterbird phylogeny and fossil calibration studies. PLoS One 5(10):e13354
Smith NA, Clarke JA (2014) Osteological histology of the Pan-Alcidae (Aves, Charadriiformes), correlates of wing-propelled diving and flightlessness. Anat Rec 297:188–199
Tambussi CP, Degrange FJ (2013) The Paleogene birds of South America. In: South American and Antarctic Continental Cenozoic Birds. Springer Netherlands, pp 29–47
Van Tuinen M, Hedges SB (2004) The effect of external and internal fossil calibrations on the avian evolutionary timescale. J Paleontol 78(1):45–50
Watanabe J, Matsuoka H (2015) Flightless diving duck (Aves, Anatidae) from the Pleistocene of Shiriya, northeast Japan. J Vertebr Paleontol 35(6):e994745
Weber E, Hesse A (1995) The systematic position of Aptornis, a flightless bird from New Zealand. Cour Forschungsinst Senck 181:293–301
Woolfenden GE (1961) Postcranial osteology of the waterfowl. University of Florida, Gainesville
Worthy TH, Tennyson AJ, Jones C, McNamara JA, Douglas BJ (2007) Miocene waterfowl and other birds from Central Otago, New Zealand. J Syst Palaeontol 5(1):1–39
Worthy TH, Mitri M, Handley WD, Lee MS, Anderson A, Sand C (2016) Osteology supports a stem-galliform affinity for the giant extinct flightless bird Sylviornis neocaledoniae (Sylviornithidae, Galloanseres). PLoS One 11(3):e0150871
Yury-Yáñez RE, Otero RA, Soto-Acuña S, Suárez ME, Rubilar-Rogers D, Sallaberry M (2012) First bird remains from the Eocene of Algarrobo, central Chile. Andean Geol 39(3):548–557.
Zelenkov NV (2012) A new duck from the Middle Miocene of Mongolia, with comments on Miocene evolution of ducks. Paleontol J 46(5):520–530
Zinsmeister WJ (1982) Late Cretaceous–Early Tertiary molluscan biogeography of the southern circum-Pacific. J Paleontol 56:84–102
Acknowledgements
Special thanks to Y. Davies and S. Bogan who allowed reviewing material under their care. We are deeply indebted to S. Lucero, S. Rozadilla, G. Lo Coco, M. Motta, M. Aranciaga Rolando, and J. D’Angelo for their comments and discussion about early bird radiations. Julia Clarke and Trevor Worthy made valuable comments on an early draft of this manuscript. Special thanks to T. Worthy for his enlightening comments on Vegavis specimen and discussions regarding its phylogenetic position. We also like to thank the anonymous reviewers who made valuable comments that greatly improved the quality of this paper. We thank M. Isasi who skillfully prepared the specimen MACN-PV 19.748 of Vegavis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Sven Thatje
Electronic supplementary material
ESM 1
(DOC 4726 kb).
Rights and permissions
About this article
Cite this article
Agnolín, F.L., Egli, F.B., Chatterjee, S. et al. Vegaviidae, a new clade of southern diving birds that survived the K/T boundary. Sci Nat 104, 87 (2017). https://doi.org/10.1007/s00114-017-1508-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-017-1508-y