Abstract
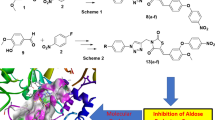
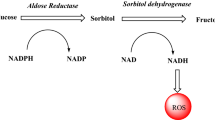
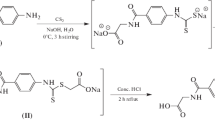
Diabetes is embracing the human population in logarithmic fashion both in developed as well as developing countries. Aldose reductase is one of the important enzymes of polyol pathway of sugar metabolism in humans. Aldose reductase inhibition has been identified as one of the important target for developing novel antidiabetic agents. In this report, we present an effective synthesis of 7-(substituted phenyl) chromeno-pyrano [2,3-d]pyrimidine-6,8,10-(7H,9H,11H)-trione derivatives and demonstrate their aldose reductase inhibition potential in order to identify novel schemes for finding putative aldose reductase inhibitors. The antioxidant activity of all the synthesized compounds with negligible toxicity demonstrates the biological efficacy of the synthesized compounds. The in silico molecular docking and structural analysis of docked poses conducted in the current investigation sheds light on the structural rationale of the observed aldose reductase inhibition by all the newly synthesized compounds.



Similar content being viewed by others
References
Alexiou P, Pegklidou K, Chatzopoulou M, Nicolaou I, Demopoulos VJ (2004) Aldose reductase enzyme and its implication to major health problems of the 21(st) century. Curr Med Chem 16:734–752
Anderson DR, Hegde S, Reinhard E, Gomez L, Vernier WF, Lee L, Liu S, Sambandam A, Sinder PA, Masih L (2005) Aminocyanopyridine inhibitors of mitogen activated protein kinase-activated protein kinase 2 (MK-2). Bioorg Med Chem Lett 15:1587–1590
Ao S, Shingu Y, Kikuchi C, Takano Y, Nomura K, Fujiwara T, Ohkubo Y, Notsu Y, Yamaguchi I (1991) Characterization of a novel aldose reductase inhibitor, FR74366, and its effects on diabetic cataract and neuropathy in the rat. Metabolism 40:77–87
Asano T, Saito Y, Kawakami M, Yamada NJ (2002) Fidarestat (SNK-860), a potent aldose reductase inhibitor, normalizes the elevated sorbitol accumulation in erythrocytes of diabetic patients. J Diabetes Complicat 16:133–138
Balendiran GK, Sawaya MR, Schwarz FP, Ponniah G, Cuckovich R, Verma M, Cascio D (2011) The role of Cys-298 in aldose reductase function. J Biol Chem 286:6336–6344
Bartolome B, Nunez V, Monagas M, Gomez Cordoves C (2004) In vitro antioxidant activity of red grape skins. Eur Food Res Technol 218(2):173–177
Berman HM, Westbrook J, Feng Z, Gilliland GS, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) the protein data bank nucleic. Acids Res 28:235–242
Bhat MA, Siddiqui N, Khan SA (2008) Synthesis of novel 3-(4-acetyl-5H/methyl-5- substituted phenyl-4,5-dihydro-1,3,4-oxadiazol-2-yl)-2H-chromen-2-ones as potential anticonvulsant agents. Acta Pol Pharm 65:235–239
Bianchi G, Tava A (1987) Synthesis of (2R)-(+)-2, 3-dihydro-2, 6-dimethyl-4H-pyran-4- one, a homologue of pheromones of a species in the hepialidae family. Agric Biol Chem 51:2001–2002
Blakeley MP, Ruiz F, Cachau R, Hazemann I, Meilleur F, Mitschler A, Ginell S, Afonine P, Ventura NO, Cousido-Siah A, Haertlein M, Joachimiak A, Myles D, Podjarny A (2008) Quantum model of catalysis based on a mobile proton revealed by subatomic x-ray and neutron diffraction studies of h-aldose reductase. Proc Natl Acad Sci USA 105:1844–1848
Bohren KM, Grimshaw CE, Lai CJ, Harrison DH, Ringe D, Petsko GA, Gabbay KH (1994) Tyrosine-48 is the proton donor and histidine-110 directs substrate stereochemical selectivity in the reduction reaction of human aldose reductase: enzyme kinetics and crystal structure of the Y48H mutant enzyme. Biochemistry 33:2021–2032
Borhani DW, Harter TM, Petrash JM (1992) The crystal structure of the aldose reductase NADPH binary complex. J Biol Chem 267:24841–24847
Brownlee JM, Carlson E, Milne AC, Pape E, Harrison DH (2006) Structural and thermodynamic studies of simple aldose reductase-inhibitor complexes. Bioorg Chem 34:424–444
Brownlee M, Cerami A, Vlassara H (1988) Advanced glycosylation end products in tissue and the biochemical basis of diabetic complications. N Engl J Med 318:1315–1321
Chakrabarti R, Rawat PS, Cooke BM, Coppel RL, Patankar S (2013) Cellular effects of curcumin on Plasmodium falciparum include disruption of microtubules. PLoS One 8:57302
Cheng JF, Ishikawa A, Yoshinori O, Arrhenius T, Nadzan A (2003) Novel chromene derivatives as TNF-alpha inhibitors. Bioorg Med Chem Lett 13:3647–3650
Costantino L, Rastelli G, Gamberini MC, Vinson JA, Bose P, Iannone A, Staffieri M, Antolini L, Del Corso A, Mura U, Albasini A (1999) 1-Benzopyran-4-one antioxidants as aldose reductase inhibitors. J Med Chem 42:1881–1893
Costantino L, Rastelli G, Vianello P, Cignarella G, Barlocco D (1999) Diabetes complications and their potential prevention: aldose reductase inhibition and other approaches. Med Res Rev 1:3–23
Cousido-Siah A, Ruiz FX, Mitschler A, Porté S, De Lera AR, Martín MJ, Manzanaro S, De la Fuente JA, Terwesten F, Betz M, Klebe G, Farrés J, Parés X, Podjarny A (2014) Identification of a novel polyfluorinated compound as a lead to inhibit the human enzymes aldose reductase and AKR1B10: structure determination of both ternary complexes and implications for drug design. Acta Crystallogr Sect A 70:889–903
De Andrade-Neto VF, Goulart MF, Da Silva Filho JF, Da Silva MJ, Pinto MR, Pinto AV, Zalis MG, Carvalho LH, Krettli AU (2004) Antimalarial activity of phenazines from lapachol, beta-lapachone and its derivatives against Plasmodium falciparum in vitro and Plasmodium berghei in vivo. Bioorg Med Chem Lett 14:1145–1149
Ehrig T, Bohren KM, Prendergast FG, Gabbay KH (1994) Mechanism of aldose reductase inhibition: binding of NADP+/NADPH and alrestatin-like inhibitors. Biochemistry 33:7157–7165
Fanfrlík J, Kolar M, Kamlar M, Hurný D, Ruiz FX, Cousido-Siah A, Mitschler A, Rezac J, Munusamy E, Lepsik M, Matejcek P, Vesely J, Podjarny A, Hobza P (2013) Modulation of aldose reductase inhibition by halogen bond tuning. ACS Chem Biol 11:2484–2492
Fernández- Bachiller MI, Pérez C, Monjas L, Rademann J, Rodríguez-Franco MI (2012) New tacrine-4-oxo-4H-chromene hybrids as multifunctional agents for the treatment of Alzheimer’s disease, with cholinergic, antioxidant, and β-amyloid-reducing properties. J Med Chem 55(3):1303–1317
Fukushi S, Merola LO, Kinoshita JH (1980) Altering the course of cataracts in diabetic rats. Invest Ophthalmol Vis Sci 19:313–315
Gacche RN, Dhole NA (2011) Profile of aldose reductase inhibition, anti-cataract and free radical scavenging activity of selected medicinal plants: an attempt to standardize the botanicals for amelioration of diabetes complications. Food Chem Toxicol 49:1806–1813
Gutteridge JM, Halliwell B (1992) Comments on review of free radicals in biology and medicine. Free Radic Biol Med 12:93–95
Harrison DH, Bohren KM, Petsko GA, Ringe D, Gabbay KH (1997) The alrestatin double- decker: binding of two inhibitor molecules to human aldose reductase reveals a new specificity determinant. Biochemistry 36:16134–16140
Howard EI, Sanishvili R, Cachau RE, Mitschler A, Chevrier B, Barth P, Lamour V, Van Zandt M, Sibley E, Bon C, Moras D, Schneider TR, Joachimiak A, Podjamy A (2004) Ultrahigh resolution drug design I: details of interactions in human aldose reductase- inhibitor complex at 0.66 A. Proteins 55:792–804
Huey R, Morris GM, Olson AJ, Goodsell DS (2007) Semiempirical free energy force field with charge-based desolvation. J Comput Chem 28:1145–1652
Kadam AS, Dawane BS, Pawar MP, Shegokar HG, Patil KK, Meshram RJ, Gacche RN (2014) Development of novel pyrazolone derivatives as inhibitors of aldose reductase: an eco-friendly one-pot synthesis, experimental screening and in silico analysis. Bioorg Chem 53:67–74
Kamble RD, Hese SV, Meshram RJ, Kote JR, Gacche RN, Dawane BS (2015) Green synthesis and in silico investigation of dihydro-2H- benzo[1,3]oxazine derivatives as inhibitors of Mycobacterium tuberculosis. Med Chem Res 24:1077–1088
Kamble RD, Meshram RJ, Hese SV, More RA, Kamble SS, Gacche RN, Dawane BS (2016) Synthesis and in silico investigation of thiazoles bearing pyrazoles derivatives as anti-inflammatory agents. Comput Biol Chem 61:86–96
Kamble RD, Yemul OS, Patil SD, Kale AB, Dawane BS (2013) Bleaching earth clay (pH 12.5): a green catalyst for rapid synthesis of pyranopyrazole derivatives via a tandem three-component reaction. Res Chem Intermed 39:3859–3866
Karasu C, Cumaoğlu A, Gürpinar AR, Kartal M, Kovacikova L, Milackova I, Stefek M (2012) Aldose reductase inhibitory activity and antioxidant capacity of pomegranate extracts. Interdiscip Toxicol 54:15–20
Khafagy MM, Abd El-Wahab AHF, Eid FA, El-Agrody AM (2002) Synthesis of halogen derivatives of benzo [h] chromene and benzo [a] anthracene with promising antimicrobial activities. Farmaco 57:715–722
Kinoshita JH, Nishimura C (1988) The involvement of aldose reductase in diabetic complications. Diabetes Metab Res Rev 4:323–337
Koch C, Heine A, Klebe G (2011) Tracing the detail: how mutations affect binding modes and thermodynamic signatures of closely related aldose reductase inhibitors. J Mol Biol 406:700–712
Laskowski RA, Swindells MB (2011) LigPlot+: multiple ligand–protein interaction diagrams for drug discovery. J Chem Inf Model 51(10):2778–2786
Liu K, Zhu HL (2011) Nitroimidazoles as anti-tumor agents anticancer agents. Med Chem 11:687–691
Mandawad GG, Kamble RD, Hese SV, More RA, Gacche RN, Kodam KM, Dawane BS (2014) An efficient synthesis of isoxazoline libraries of thiophene analogs and its antimycobacterial investiga-tion. Med Chem Res. doi:10.1007/s00044-014-1016-y
Malamas MS, Hohman TC, Millen JJ (1994) Novel spirosuccinimide aldose reductase inhibitors derived from isoquinoline-1,3-diones: 2-[(4-Bromo-2- fluorophenyl)methyl]-6-fluorospiro[isoquinoline-4(11H),3′-pyrrolidine]1,2′,3,5′(2H)-tetrone and congeners. Med Chem 37:2043–2058
Memvanga PB, Coco R, Préat V (2013) An oral malaria therapy: curcumin-loaded lipid-based drug delivery systems combined with β-arteether. J Control Release 3:904–913
Miteva MA, Guyon F, Tufféry P (2010) Frog2: efficient 3D conformation ensemblegenerator for small compounds. Nucleic Acids Res 38:W622–W627
Mohr SJ, Chirigios MA, Fuhrman FS, Pryor JW (1975) Pyran copolymer as an effective adjuvant to chemotherapy against a murine leukemia and solid tumor. Cancer Res 35:3750–3754
Moon DO, Choi YH, Kim ND, Park YM, Kim YG (2007) Inhibitory effects of eicosapentaenoic acid on lipopolysaccharide-induced activation in BV2 microglia. Int J Immunopharmacol 7:222–229
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, Olson AJ (1998) Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J Comput Chem 19:1639–1662
Ough M, Lewis A, Bey EA, Gao J, Ritchie JM, Bornmann W, Boothman DA, Oberley LW, Cullen JJ (2005) Efficacy of beta-lapachone in pancreatic cancer treatment: exploiting the novel, therapeutic target NQO1. Cancer Biol Ther 4:95–102
Patel RM, Patel NJ (2011) In vitro antioxidant activity of coumarin compounds by DPPH, super oxide and nitric oxide free radical scavenging methods. Adv Pharm Educ Res 1:52–68
Podjarny A, Cachau RE, Schneider T, Van Zandt M, Joachimiak A (2004) Subatomic and atomic crystallographic studies of aldose reductase: implications for inhibitor binding. Cell Mol Life Sci 61:763–773
Rajput SB, Shinde RB, Routh MM, Karuppayil SM (2013) Anti-candida properties of asaronaldehyde of Acorus gramineus rhizome and three structural isomers. Chin Med 8:18
Ramirez MA, Borja NL (2008) Epalrestat: an aldose reductase inhibitor for the treatment of diabetic neuropathy. Pharmacotherapy 28:646–655
Rappe AK, Casewit CJ, Colwell KS, Goddard WA, Skiff WM (1992) A rule-based full periodic table force field for molecular mechanics and molecular dynamics simulations. J Am Chem Soc 114:10024–10039
Rappe AK, Colwell CK, Casewit J (1993) Application of a universal force field to metallic complexes. Inorg Chem 32:3438–3450
Rastelli G, Antolini L, Benvenuti S, Costantino L (2000) Structural bases for the inhibition of aldose reductase by phenolic compounds. Bioorg Med Chem 8:1151–1158
Rondeau JM, Tete-Favier F, Podjarny A, Reymann JM, Barth P, Biellmann JF, Moras D (1992) Novel NADPH-binding domain revealed by the crystal structure of aldose reductase. Nature 355:469–472
Sadasivam S, Manickman A (1992) Biochemical methods. New Age International Pub. (Pvt) Limited, New Delhi
Sestanj K, Bellini F, Fung S, Abraham N, Treasurywala A, Humber L, Simard-Duquesne N, Dvornik D (1984) N-[5-(trifluoromethyl)-6-methoxy-1-naphthalenyl] thioxomethyl]- N-methylglycine (Tolrestat), a potent, orally active aldose reductase inhibitor. J Med Chem 27:255–256
Smith PW, Sollis SL, Howes PD, Cherry PC, Starkey ID, Cobley KN, Weston H, Scicinski J, Merritt A, Whittington A, Wyatt P, Taylor N, Green D, Bethell R, Madar S, Fenton RJ, Morley PJ, Pateman T, Beresford AJ (1998) Dihydropyrancarboxamides related to zanamivir—a new series of inhibitors of influenza-virus sialidases—1—discovery, synthesis, biological-activity, and structure- activity-relationships of 4-guanidino-4h-pyran-6-carboxamides and 4-amino-4h-pyran-6 carboxamides. J Med Chem 41:787–797
Steuber H (2007) Structural and thermodynamic characterization of inhibitor binding to aldose reductase: insight into binding modes, driving forces and selectivity determinants. PhD thesis, Marburg University, Marburg
Steuber H, Zentgraf M, Gerlach C, Sotriffer CA, Heine A, Klebe G (2006) Expect the unexpected or caveat for drug designers: multiple structure determinations using aldose reductase crystals treated under varying conditions reveal surprising challenges for structure-based ligand design. J Mol Biol 363:174–187
Stribling D, Mirrlees DJ, Harrison HE, Earl DC (1985) Properties of ICI 128,436, a novel aldose reductase inhibitor, and its effects on diabetic complications in the rat. Metabolism 34:336–344
Sudha BN, Sridhar C, Sastry VG, Reddy YR, Sreevidya O, Lavanya S, Jyothi VA, Nagesh V, Sen S, Chakraborty R (2013) Synthesis, characterisation and anthelmintic activity of 3-(4-acetyl-5-phenyl-4,5-dihydro-1,3,4-oxadiazol-2-yl)- 2H- chromen-2-one derivatives. Indian J Chem 52B:422–427
Suryanarayana P, Kumar PA, Saraswat M, Petrash JM, Reddy GB (2004) Inhibition of aldose reductase by tannoid principles of Emblica officinalis: implications for the prevention of sugar cataract. Mol Vis 10:148–154
Tarle I, Borhani DW, Wilson DK, Quiocho FA, Petrash JM (1993) Probing the active site of human aldose reductase. Site-directed mutagenesis of Asp-43, Tyr-48, Lys-77, and His-110. J Biol Chem 268:25687–25693
Urzhumtsev A, Tete-Favier F, Mitschler A, Barbanton J, Barth P, Urzhumtseva L, Biellmann JF, Podjarny A, Moras D (1997) A ‘specificity’ pocket inferred from the crystal structures of the complexes of aldose reductase with the pharmaceutically important inhibitors tolrestat and sorbinil. J Mol Struct 5:601–612
Varnai P, Warshel A (2000) Computer simulation studies of the catalytic mechanism of human aldose reductase. J Am Chem Soc 122:3849–3860
Wallace AC, Laskowski RA, Thornton JM (1995) LIGPLOT: a program to generate schematic diagrams of protein-ligand interactions. Protein Eng 8(2):127–134
Wild S, Roglic G, Green A, Sicree R, King H (2004) Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 27:1047–1053
Zhang L, Zhang H, Zhao Y, Li Z, Chen S, Zhai J, Chen Y, Xie W, Wang Z, Li Q, Zheng X, Hu X (2013) Inhibitor selectivity between aldo-keto reductase superfamily members AKR1B10 and AKR1B1: role of Trp112 (Trp111). FEBS Lett. 22:3681–3686
Zhang Q, Liang XC (2015) Effects of microtubule depolymerization on spontaneous beating and action potential of cardiac myocytes in rats and its mechanism. Zhonghua Shao Shang Za Zhi 3:192–198
Acknowledgments
Author SVH acknowledge the financial support from SRTM University, Nanded.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Shrikant V. Hese and Rohan J. Meshram have contributed equally to this article.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Hese, S.V., Meshram, R.J., Kamble, R.D. et al. Antidiabetic and allied biochemical roles of new chromeno-pyrano pyrimidine compounds: synthesis, in vitro and in silico analysis. Med Chem Res 26, 805–818 (2017). https://doi.org/10.1007/s00044-017-1794-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-017-1794-0