Abstract
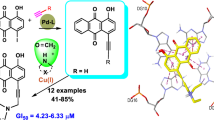
From a partially degraded Diels–Alder adduct of α-myrcene and 1,4-benzoquinone, several model compounds belonging to a new series of 1,4-naphthohydroquinone derivatives have been prepared. Phenyl, pyridyl, imidazolyl and some nucleic base mimic heterocycles have been attached to the naphthohydroquinone system through linkers of different size and type, leading to potentially antineoplastic hybrid structures. The new compounds have been evaluated in vitro for their cytotoxicity against cultured human cancer cells of A-549 lung carcinoma, HT-29 colon adenocarcinoma and MDA-MB-231 breast carcinoma. GI50 values ranged in the μM level.


Similar content being viewed by others
References
Abdel-Hafez NA, Amr AE, Hammam AEG, Mohamed AM, Mohamed SF (2006) Anticancer activities of some newly synthesized pyridine, pyrane, and pyrimidine derivatives. Bioorg Med Chem 14:5481–5488
Aguilera N, Castro MA, García-Grávalos MD, Gordaliza M, Miguel del Corral JM, Molinari A, Oliva A, San Feliciano A (2000) New antineoplastic prenylhydroquinones. Synthesis and evaluation. Bioorg Med Chem 8:1027–1032
Appel PL, Powis G (1980) Relationship of the single-electron reduction potential of quinones to their reduction by flavoproteins. Biochem Pharmacol 29:2567–2572
Araya C, Castro MA, García-Grávalos MD, Miguel del Corral JM, Molinari A, Oliva A, San Feliciano A (2004) Cytotoxic-antineoplastic activity of acetyl derivatives of prenylnaphthohydroquinone. Il Farmaco 59:651–656
Bacherikov VA, Chen CH, Chou TC, Dong HJ, Lin YW, Tsai TJ, Su TL (2004) Potent antitumor N-mustard derivatives of 9-anilinoacridine, synthesis and antitumor evaluation. Bioorg Med Chem Lett 14:4719–4722
Bailly C, Dias N, Goossens JF, Jacquemard U, Lansiaux A, Routier S, Mérour JY (2005) Synthesis of 2,5- and 3,5-diphenylpyridine derivatives for DNA recognition and cytotoxicity. Eur J Med Chem 40:1087–1095
Bokesch H, Boyd MRJ, Kenney S, McMahon J, Monks A, Scudiero D, Skenan P, Storeng R, Vistica D, Waren JT (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82:1107–1112
Brana MF, Cacho M, De Pacual T, Gradillas B, Ramos BA (2001) Intercalators as anticancer drugs. Curr Pharm Des 7:1745–1780
Broughton HB, Castro MA, Chamorro P, García-Grávalos MD, Gordaliza M, Mahiques MM, Miguel del Corral JM, Molinari A, San Feliciano A (2001) New selective cytotoxic diterpenylquinones and diterpenylhydroquinones. J Med Chem 44:1257–1267
Brunmark A, Cadenas E (1989) Oxidation of quinone by hydrogen peroxide, formation of epoxy and hydroquinone adducts and electronically excited states. Basic Life Sci 49:81–86
Castro MA, García-Grávalos MD, Gordaliza M, Mahiques MM, Miguel del Corral JM, San Feliciano A (1996) Synthesis and bioactivity of new antineoplastic terpenylquinones. Bioorg Med Chem Lett 6:1859–1864
Castro MA, García-Grávalos MD, Gordaliza M, Mahiques MM, Miguel del Corral JM, San Feliciano A (1998) Further antineoplastic terpenylquinones and terpenylhydroquinones. Bioorg Med Chem 6:31–41
Castro MA, García-Grávalos MD, Gordaliza M, Gualberto SA, Martin ML, Miguel del Corral JM, Oliveira AB, San Feliciano A (2002a) Synthesis and biological evaluation of cytotoxic 6(7)-alkyl-2-hydroxy-1,4-naphthoquinones. Arch Pharm Pharm Med Chem 9:427–437
Castro MA, Gordaliza M, Gupta M, Miguel del Corral JM, Molinari A, Oliva A, Reinoso P, Solís P, San Feliciano A (2002b) Cytotoxic-antineoplastic activity of hydroquinone derivatives. Eur J Med Chem 37:177–182
Castro MA, Cuevas C, Gamito AM, Gordaliza M, Gualberto A, Martin ML, Miguel del Corral JM, San Feliciano A (2005a) Synthesis and cytotoxicity of new aminoterpenylquinones. Bioorg Med Chem 13:631–644
Castro MA, Cuevas C, Miguel del Corral JM, Molinari A, Ojeda C, Oliva A, San Feliciano A (2005b) New cytotoxic-antineoplastic prenyl-1,2-naphthohydroquinone derivatives. Bioorg Med Chem 13:6645–6650
Castro MA, Cuevas C, Escobar J, Gallardo C, Miguel del Corral JM, Molinari A, Ojeda C, Oliva A, San Feliciano A (2005c) Synthesis, characterization and cytotoxicity of chloro derivatives of prenylnaphthohydroquinone. Bioorg Med Chem 13:3841–3846
Chang HX, Cheng CC, Chou TC, Liu LF, Savaraj N, Yu C (1999) Design of antineoplastic agents based on the “2-phenylnaphthalene-type” structural pattern. 4. Synthesis and biological activity of 2-chloro-3-(substituted phenoxy)-1,4-naphthoquinones and related 5,8-dihydroxy-1,4-naphthoquinones. J Med Chem 42:405–408
Chen Y, Gao H, Li C, Lu W, Pang T, Shen H, Sun J, Xu C, Zhang X (2005) Action of a novel pyrrolo[1,2-c][1.3]benzodiazepine on the viability of Jurkat and neuronal/glial cells. Bioorg Med Chem Lett 15:3233–3236
Chen Y, Gu LQ, Huang ZS, Tan JH, Wang XD, Wu JY, Zhang QX (2006) Synthesis, DNA binding and cytotoxicity of new pyrazole emodin derivatives. Eur J Med Chem 41:1041–1047
Cheng CC (1998) Structural aspects of antineoplastic agents. In: Ellis GP, West GB (eds) A new approach in progress in medicinal chemistry, vol 25. Elsevier, Amsterdam, pp 35–83
Cheng CC, Chou TC, Luo IL (1996) Design of antineoplastic agents on the basis of the 2-phenylnaphthalene-type structural pattern. 3. Synthesis and biological activity evaluation of 5H-benzo[b]naphtho[2,3-d]pyrrole-6,11-dione derivatives. J Heterocycl Chem 33:113–117
Cho BT, Kang SK (2005) Direct and indirect reductive amination of aldehydes and ketones with solid acid-activated sodium borohydride under solvent-free conditions. Tetrahedron 61:5725–5734
Clement JJ, Faircloth GT, Stewart DJ (1988) A simple screening for the quantitative measurement of cytotoxicity, to resting primary lymphocyte culture. Tissue Cult Methods 11:201–205
Cocco MT, Congiu C, Onnis V (2003) New bis(pyridyl)methane derivatives from 4-hydroxy-2-pyridones: synthesis and antitumoral activity. Eur J Med Chem 38:37–47
Cocco MT, Congiu C, Lilliu V, Onnis V (2004), Synthesis of new N-(2-(trifluoromethyl)pyridin-4-yl) anthranilic acid derivatives and their evaluation as anticancer agents. Bioorg Med Chem Lett 14:5787–5791
Cocco MT, Congiu C, Lilliu V, Onnis V (2007) Synthesis and in vitro antitumoral activity of new 3,5-dicyanopyridine derivatives. Bioorg Med Chem 5:1859–1867
Esposti MD, Lenaz G, Rotillo G (1984) Effects of dibromothymoquinone on the structure and function of the mitochondrial bc1 complex. Biochem Biophys Acta 767:10–20
Graves DE, Velea LM (2000) Intercalative binding of small molecules to nucleic acids. Curr Org Chem 4:915–929
Grierson DS, Legraverend MJ (2006) The purines: potent and versatile small molecule inhibitors and modulators of key biological targets. Bioorg Med Chem 14:3987–4006
Hurley HL (2002) DNA and its associated processes as targets for cancer therapy. Nat Rev Cancer 2:188–200
Konji VN, Makawity DM, Olowookere JO (1990) Interaction of benzoquinones with mitochondria interferes with oxidative phosphorylation characteristics. FEBS Lett 266:26–28
Korolkovas A (1988) Essentials of medicinal chemistry, 2nd edn. Wiley, New York
Lemke TL, Williams DA (2002) Foye’s principles of medicinal chemistry, 5th edn. Lippincot Williams & Wilkims, Baltimore
O’Brien MD (1991) Molecular mechanism of quinone cytotoxicity. Chem Bio Interact 80:1–41
Powis G (1989) Free radical formation by antitumor quinones. Free Radic Biol Med 6:63–101
Acknowledgements
The authors acknowledge the financial support from the Comisión Nacional de Investigación Científica y Tecnológica, CONICYT (Proyecto FONDECYT 1060447), Dirección de Investigación de la Vicerrectoría de Investigación y Estudios Avanzados de la Pontificia Universidad Católica de Valparaíso, Chile (Projects DI. 125.795-05 and 125.796-06), and Junta de Castilla y León, Spain (Project SA114A06). C. Ojeda acknowledges financial support from the Programa Mecesup, Project UCH 0408 for a research stay at the University of Salamanca, Spain. This research was performed under the auspices of the “Programa Iberoamericano CYTED - Subprograma X”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Molinari, A., Ojeda, C., Oliva, A. et al. Synthesis, characterisation, and antineoplastic cytotoxicity of hybrid naphthohydroquinone–nucleic base mimic derivatives. Med Chem Res 18, 59–69 (2009). https://doi.org/10.1007/s00044-008-9108-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-008-9108-1