Abstract
Objectives
To review the published literature examining the impacts of the Aral Sea disaster on children’s health.
Methods
A systematic review of the English language literature.
Results
The literature search uncovered 26 peer-reviewed articles and four major reports published between 1994 and 2008. Anemia, diarrheal diseases, and high body burdens of toxic contaminants were identified as being among the significant health problems for children. These problems are associated either directly with the environmental disaster or indirectly via the deterioration of the region’s economy and social and health care services. While links between persistent organic pollutant exposures and body burdens are clear, health impacts remain poorly understood. No clear evidence for the link between dust exposure and respiratory function was identified.
Conclusion
While important questions about the nature of the child health and environment relationships remain to be answered, the literature unequivocally illustrates the seriousness of the public health tragedy and provides sufficient evidence to justify immediate action. Regrettably, international awareness of the crisis continues to be poor, and the level of action addressing the situation is wholly inadequate.
Similar content being viewed by others
Introduction
The population living in the Aral Sea area is facing a multitude of environmental problems stemming from what is considered to be one of the worst ecological disasters the world has seen (Micklin 2007; Small and Bunce 2003; Glantz 1999). It was only after the collapse of the Soviet Union in 1991 and the subsequent formation of newly independent Central Asian governments that travel to the Aral Sea area became possible, and the world began to learn the extent of the devastation. Decades of destructive agricultural practices and general environmental mismanagement led to the disappearance of what was once the fourth largest inland body of water in the world, combined with the contamination of the air, soils and water with toxic agricultural chemicals (Micklin 2007). The environmental problems in the region have had significant public health impacts exacerbated by the breakdown of the health care and social security systems and the rise in unemployment and poverty that were associated with the loss of Soviet support (Carpenter et al. 2006; Glantz 1999).
The roots of the demise of the Aral Sea are complex but well understood. At the height of the Cold War in the 1950s the Soviet government was eager to gain self-sufficiency in cotton and imposed ‘modern’ cotton farming methods to achieve this goal. Known by the Soviets as “white gold”, cotton was a highly strategic crop considered necessary to outfit a rapidly expanding military (Small and Bunce 2003). Central Asia was considered the ideal region for cotton agriculture, with its warm climate and large, exploitable population and abundant water source, namely the Amu Darya and the Syr Darya Rivers which feed the Aral Sea. By diverting massive quantities of water away from the Aral Sea into an expanding network of irrigation canals, the region quickly became one of the world’s largest cotton producers; however, the environmental impacts have been enormous (Glantz 1999; Abdullayev 2010). Since the mid 1960s, the sea has been steadily receding to the point that the sea today covers approximately one-fifth of its original area, and Muynak for example, which was once a major fishing port and tourist destination, is now over a hundred kilometers from the coast. The salinity of the sea water has increased by more than ten times in some areas, and the sea itself is dead (Micklin 2007), and the salinization of agricultural land resulting from over-irrigation as well as the wind transport of salts from the exposed sea bed has severely impacted ground and surface water quality and reduced agricultural productivity (Bosch et al. 2007; Glantz 1999; O’Hara et al. 2000). Concomitantly, decades of chemical-dependent agricultural practices have resulted in high levels of toxic pesticides in the local environment and in the population living there (Muntean et al. 2003).
The population living in the Aral Sea area faces numerous health issues, with high rates of anemia, cancers, respiratory illness, and birth defects being among the most commonly reported health problems (Kaneko et al. 2002; Ataniyazova et al. 2001; Giebel et al. 1998; Hashizume et al. 2003; Kunii et al. 2003; Zaridze et al. 1992; Crighton et al. 2003b). These conditions have been linked either directly or indirectly to the areas environmental degradation. While the population as a whole has been affected, physiological, behavioral, and metabolic characteristics, combined with the immaturity of organs and systems make children particularly vulnerable to contaminants and increases their likelihood of exposure (Carpenter et al. 2006).
While local residents and scientists have long claimed that the environmental problems have been significantly affecting their health, limited empirical evidence of the relationships combined with a general disinterest in the disaster internationally have been cited as barriers to action (Small et al. 2001). While the evidence base remains underdeveloped along with the international interest in the disaster, significantly more is now known since studies first began being conducted and published in the early 1990s. No review of extant studies has been undertaken to date. Therefore, the main objective of this review was to assess what is known from the English literature about the relationship between children’s health and the environmental problems in the Aral Sea area in an effort to encourage research where questions remain, and action where none is being taken.
Methods
The geographic region commonly referred to as the Aral Sea area includes Uzbekistan’s Karakalpakstan, a semi-autonomous republic, and Khorezm Oblast (state), the Kazyl Orda region of Kazakhstan, and North-western Turkmenistan (Fig. 1).
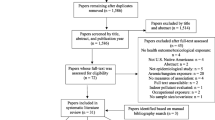
Standard search engines, specifically Medline, SCOPUS, Geobase as well as Google and Google Scholar, were used to conduct a systematic literature search. Searches were limited to English language publications covering the period from March 2010 going back to 1985, 7 years before the earliest previously identified publications, and 6 years before foreign researchers were typically able to gain access to the region. Search terms included geographic locations and health conditions. The following place names were used as search terms: Uzbekistan, Karakalpakstan, Kazakhstan, Turkmenistan, Aral Sea, Kazyl Orda, and Khorezm Oblast. These locations encompass what is commonly referred to as the Aral Sea area (Fig. 1). One problem with using place names as search terms is the range of English spellings that exist. To address this issue, any new spellings that were identified as the search progressed were incorporated into the search criteria, and searches were redone. Health-related search terms ranged from very general terms, such as health, disease, illness, morbidity, and mortality, to specific health conditions. The choice of the latter was an iterative process based initially on discussions with health experts familiar with the situation, with additional terms being added as the search evolved. Experts from international non-governmental organizations (NGOs) working in the region including UNICEF, Médecins Sans Frontières (MSF) and the World Health Organization (WHO), as well as from local NGOs, governments, universities, and hospitals were consulted during a visit in 2008 to Uzbekistan and Karakalpakstan in an effort to identify additional reports and ongoing research.
The review itself was limited to empirical studies of populations living in the Aral Sea area (as defined above) that directly or indirectly address children’s health and examine health conditions that are plausibly linked to the Aral Sea environmental situation, whether identified in the paper or not. The literature included for review did not have to be peer reviewed but was required to have a clear statement of objectives, be explicit about methodology, and include results supported by tabular or other forms of data presentation, and therefore be amenable to critical appraisal. These somewhat loose criteria led only to the exclusion of gray literature (magazine and newspaper articles), NGO promotional material, conference abstracts, and a small number of studies that focused exclusively on adults.
Results
Twenty-six articles or book chapters and four published reports published between 1994 and 2008 were identified that met our inclusion criteria. All but a handful of studies were published between 1997 and 2004 (Fig. 2). The absence of publications in recent years that are specific to children or otherwise suggests that internationally sponsored health research in the area has all but ceased. From the published research, two sets of health problems can be described although they are not mutually exclusive. The first set are those which are indirectly linked to the environmental disaster via economic and social disruption, including high infant mortality, low birthweight, abnormal physical development, diarrheal diseases, and poor self-rated health. The second set are those that are hypothesized, at least by some of the authors, to be directly linked to the environmental disaster, specifically, anemia, respiratory diseases such as asthma and acute respiratory infection (ARI), high body burdens of toxic chemicals, hypercalciuria, and renal tubular dysfunction.
Four published reports were identified for Uzbekistan, the goal of each being to assess the health of populations generally using a broad range of standard epidemiological measures. The first of these was the Uzbekistan Demographic Health Survey (UDHS) (Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997). Using a national representative sample of 4,415 women reporting on 7,271 children, questions on subjects including fertility, maternal care, infant mortality, post-natal growth, and anemia, and data were analyzed regionally. Biodata on a subsample of respondents was also collected. The Uzbekistan Health Examination Survey (UHES) (Ministry of Health Republic of Uzbekistan 2004) was conducted in 2002 using a similar methodology to the UDHS but with a larger sample (n = 7,793 adults). Asthma, anemia, and nutritional status in children were among the health conditions examined. Region-specific data for many conditions were not reported here. Two major studies called the Multiple Indicators Cluster Survey (MICS) (UNICEF 2000, 2006) have been carried out by UNICEF, the Government of Uzbekistan. The first of these (UNICEF 2000) employed a nationally representative, multistage probability sampling strategy to obtain a sample of over 5,300 households distributed across five survey regions, one being Karakalpakstan and Khorezm Oblasts combined. Indicators of children’s health included birthweight, frequency of diarrhea, and ARI. The 2006 MICS (UNICEF 2006), although larger (n = 10,198 households), focused on health care-related indicators. Questions about diarrhea were asked, but not reported regionally.
Infant mortality
Infant and under-five morality rates refer to the likelihood of a child dying before their first and fifth birthdays, respectively. Of the Uzbek national surveys, only the 2006 MICS provides data broken down regionally. In Karakalpakstan and Khorezm, infant mortality and under-five mortality were 56/1,000 and 65/1,000, respectively. Across the Uzbekistan study regions, rates vary between 28/1,000 and 63/1,000 for infant mortality, and between 31/1,000 and 76/1,000 for under-five mortality.
Birthweight and post-natal growth
All of the Uzbek national health surveys mentioned above examined physical development indicators, including birthweight, weight-for-age, and height-for-age. From the UDHS (Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997), no notable difference was identified between the Karakalpakstan/Khorezm study region and other regions in the country for percent of children born weighing less than 2,500 g (defined as low birthweight). With regards to height-for-age, over 26% of children from Karakalpakstan/Khorezm were found to be moderately to severely stunted (defined as −2 standard deviation units from the median WHO international reference population). Although this figure is high, it is somewhat less than what was reported nationally (31%). The UHES (Ministry of Health Republic of Uzbekistan 2004) results suggest that Karakalpakstan/Khorezm children under 5 years on average fared the same or better than other study regions for height-for-age, weight-for-height, and weight-for-age. From the MICS (UNICEF 2000, 2006), we see conflicting findings. In Karakalpakstan/Khorezm in 2000 (UNICEF 2000), 9% of children born within 12 months of the survey were low birthweight defined as being born under 2,500 g as compared to 6.0% for Uzbekistan as a whole. In 2006 (UNICEF 2006), the Karakalpakstan/Khorezm study region had the lowest percentage of children born under 2,500 g.
Diarrheal disease
Diarrheal disease represents a significant cause of morbidity and mortality in the Aral Sea area, particularly among children (Semenza et al. 1998; Herbst et al. 2008; UNICEF 2000). Four studies that examine diarrheal disease were reviewed (Table 1). The first available data on this condition came from the UDHS (Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997), where it was reported that over 8% of children under 3 years in the Karakalpak/Khorezm study region had experienced an episode of diarrhea in the previous 2 weeks as compared to just over 5% nationally. Similar findings were reported in the first MICS study (UNICEF 2000). Herbst et al. (2008) in a study of diarrheal incidence in Khorezm found incidence rates for children under 2 years of age to be above the median estimates for low-income countries globally and that rates were associated with feces-contaminated drinking water and sanitation-related behavior. Semenza et al. (1998) in an intervention study conducted in Nukus, the capital of Karakalpakstan, found that individuals using home chlorination had diarrheal illnesses at rates well below those not using home chlorination regardless of the source and conclude that the existing water treatment and distribution infrastructure does little to reduce the risk.
Persistent organic pollutants (POPs): exposures and body burden
Persistent organic pollutants (POPs) as well as heavy metals found in agricultural chemicals have been reported to be present at high levels in the Aral Sea area environment, local foods, and in the population (Table 2), and are understood from other contexts to pose significant health risks to children, affecting reproductive, endocrine and immune systems, pre- and post-natal growth, and neurological development (Ritter et al. 1996). The earliest identified study on POP body burdens in the Aral Sea area was conducted by Petreas et al. (1996), who, in response to reports of declining breast-feeding rates associated with mother’s concerns about chemical contamination, examined levels of contaminants in breast milk and various foods including fish and dairy products. Results showed that breast milk samples taken from agricultural areas of Southern Kazakhstan had highly elevated levels of highly toxic compounds including 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), a by-product of many common agricultural chemicals. Jensen et al. (1997), in a study comparing Kazakh children living near the Aral Sea and hospitalized for ‘ecological diseases’ and reference populations of comparable age from Europe and Almaty, found that levels of polychlorinated biphenyls (PCBs), dichlorodiphenyldichloroethylene (DDE) and dichlorodiphenyltrichloroethane (DDT) compounds in blood lipids to be significantly higher among the hospitalized children. While these findings are important, the comparison of children with symptoms of ‘ecological diseases’, to those without, is problematic. Hooper et al. (1997) examined levels of PCB’s and organochlorine residues in the breast milk of donors living near Aralsk, Kazakhstan. While levels of DDE, DDT, and PCB were comparable to world averages, levels of beta-hexachlorocyclohexane (β-HCH) were among the highest reported in the published literature. Breast milk samples were also found to contain other dioxin congeners including the most toxic, TCDD, at levels that are among the highest ever documented for populations of reproductive age (Hooper et al. 1998; Lutter et al. 1998). Arsenic, sodium, mercury, bromine, and nickel are among the other compounds identified as being significantly higher in children living near the Aral Sea area (Chiba et al. 2004; Erdinger et al. 2004).
The single body burden study in Karakalpakstan was conducted by Ataniyazova et al. (2001). Here, tests to measure metals, persistent organochlorine pesticides (OCPs), and dioxins were done in cord blood, blood from pregnant women and in breast milk, from subjects living in close proximity to the Aral Sea. Similar to the results reported in Kazakhstan, findings revealed significantly elevated levels of β-HCH, DDT and DDE, and TCDD levels six times higher than those found in Western Europe and among the highest levels in any country in the world.
In an effort to understand the source of the high contaminate body burdens, a number of studies have also examined levels of POPs and agricultural chemicals in the food chain. Muntean et al. (2003) tested samples of foodstuffs commonly produced and consumed in Karakalpakstan. Findings revealed elevated levels DDE and TCDD, with the highest levels of contamination in foods with elevated lipid content such as sheep, eggs, and cottonseed oil. Hooper (1999) had similar findings in a Kazakh context with the highest levels being reported in foodstuffs sourced from cotton-growing areas.
In the single study examining health outcomes associated with contaminate body burdens, Mazhitova et al. (1998) tested the relationship between blood levels of polychlorinated organic compounds to growth and thyroid hormone status in hospitalized Kazakh schoolchildren from the Aral Sea region. Findings revealed an inverse correlation between body mass index (BMI) with total PCB, DDT, and DDE concentrations, but not with β-HCHs. Levels of insulin-like growth factor-1 (IGF-1) were reduced to the same extent as BMI. The authors propose that PCBs and DDTs may affect the body’s ability to absorb nutrients, thereby leading to malnutrition.
Respiratory disease
The retreat of the Aral Sea has exposed the former sea bed to significant winds that have led to airborne dust deposition rates that have been found to be among the highest in the world (O’Hara et al. 2000). Dust (PM10 or smaller) exposure has been hypothesized to be a potential risk factor for ARI, asthma, and other respiratory problems, conditions which have been examined in several studies in the Aral Sea area (Bennion et al. 2007; Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997; Kunii et al. 2003; Ubaydullaev and Uzakova 2002; UNICEF 2000) (Table 3).
The UDHS (Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997) reported that the percentage of children under 3 years with cough and rapid breathing (symptoms compatible with ARI) over a 2-week period was 3.8% compared to 1.2% nationally. The MICS (UNICEF 2000) found negligible rates of ARI symptoms (<1%) although this study was conducted in the summer when respiratory infections are typically at their lowest. Ubaydullaev and Uzakova (2002), in a survey covering three regions in Uzbekistan including Karakalpakstan, reported that nocturnal asthma rates were highest in Karakalpakstan, but lower for all other symptoms. From this latter study, no information on sample selection is provided, and while it is indicated that clinical and laboratory data were collected, none is presented.
Kunii et al. (2003) examined an ‘exposed’ group of children close to the Aral Sea in Kazakhstan and an age- and sex-matched comparison group living far from the sea. Prevalence of current cough and wheeze, and restrictive pulmonary dysfunction were higher in the exposed group, and the percentage of predicted forced vital capacity was lower. No significant differences for other symptoms were identified. Bennion et al. (2007) examined lung function and respiratory symptoms of children across Karakalpakstan and found significant geographical variation in these outcomes, but no significant associations with dust deposition. Overall, the evidence that respiratory health has been affected by environmental exposures in the Aral Sea area remains inconclusive.
Hypercalciuria, urolithiasis, and renal tubular dysfunction
Exposure to the Aral Sea area’s highly salinized environment (Abdullayev 2010) has been reported to be associated with a number of health problems including hypercalciuria—high levels of urinary calcium (Ca) and sodium (Na) excretion (Abdullayev 2010; Kaneko et al. 2002); crystalluria—the formation of crystals in the urine and a precursor to urolithiasis (Arustamov et al. 2001); and urolithiasis—the formation of urinary calculi or ‘stones’ in the urinary tract (Arustamov et al. 2001). Hypothesizing the relationship between crystalluria, urolithiasis and high levels of Ca, Na, and other dissolved solids in drinking water, Arustamov et al. (2001) recruited a random sample of 1,817 residents in a ‘typical settlement’ in Khorezm (Uzbekistan; Fig. 1) for interviews and urological examinations. Results for adults showed rates well below those identified in other local data. Between 50 and 85% of children were identified as having crystalluria, but no comparative rates were provided. Kaneko et al. (2002) undertook a study in August 2000 examining the prevalence of hypercalciuria in a random sample of school-aged children living near the Aral Sea (Karalinsk) and in a Kazakh reference group living far from the sea. Findings revealed hypercalciuria in almost 40% of children in the Aral Sea group compared to 13% in the reference group. While salt intake via water, salty dust, and food is one possible cause of increased urinary Ca and Na excretion, another is renal tubular dysfunction-related toxic chemical (e.g. lead and cadmium) exposures (Kaneko et al. 2003). To test this, Kaneko et al. (2003) examined the same urine samples from their 2002 study for NAG (N-acetyl-β-D-glucosaminidase) and BMG (β2-microglobulin) to identify renal tubular dysfunction. Findings confirmed significantly higher rates of dysfunction among those living near the Aral Sea, but elevated levels of cadmium and lead were not found (Chiba et al. 2004) (Table 2). While the cause of renal tubular dysfunction in this context remains unclear, the authors stress that it should be taken very seriously given its relationship to developmental delay (Kaneko et al. 2003).
Childhood anemia
Childhood anemia is acknowledged to be a significant and growing health problem among children in the region and is hypothesized to be associated with a number of factors, including iron deficiencies and environmental exposures (Hashizume et al. 2004). Six studies examining anemia among children living in the Aral Sea area were conducted (Table 4). Morse (1994), conducted a cross-sectional survey in Muynak (Karakalpakstan) backed by standardized laboratory data of a random sample of children under 5 years of age and found that 70.4% had nutritional anemias. Similar rates were identified among children in Muynak by Giebel et al. (1998) whereby iron-deficiency anemia was again found to be the most common etiology. According to these results, Muynak has among the highest estimated prevalence rates of childhood anemia in the world. The UDHS (Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997) rates among children in Karakalpakstan/Khorezm were found to be well above those for Uzbekistan as a whole.
Hypothesizing a relationship between anemia and environmental contaminate exposures, Hashizume et al. (2003) examined anemia and iron deficiency among schoolchildren in two districts in Kazakhstan and found rates to be significantly higher in the district adjacent to the sea, and iron deficiency anemia constituting only a third of all cases. The authors suggest potential causes of anemia here to be micronutrient status, parasite infestations, hereditary disorders, and exposure to environmental pollutants. By examining dietary and hematological data in a sub-sample of the same population, it was found that iron intake was adequate, but that bioavailable iron intake was well below metabolic requirements when inhibitors of absorption common in the Kazakh diet (e.g. black tea and whole grains) were considered (Hashizume et al. 2004). While there are a number of limitations to this study, including a small sample size and the algorithms for assessing bioavailability used, the authors are correct in suggesting that iron fortification or supplementation programs alone may not solve the anemia problem in the region.
Self-rated and psychosocial health
Three studies were identified that addressed self-rated and psychosocial health issues: Ministry of Health Republic of Uzbekistan 2004; Crighton et al. 2003a, b. In the UHES (Ministry of Health Republic of Uzbekistan 2004), mothers were asked to rate their child’s (under 5 years of age) general health on a scale ranging from excellent to poor. ‘Excellent’ to ‘very good’ health was least commonly reported by mothers in Karakalpakstan/Khorezm compared to the other study regions (5 vs. 15–25%); however, levels of ‘fair to poor’ health were comparable at approximately 10%. Studies by Crighton et al. (2003a, b) examined the psychosocial health and the effects of psychosocial factors and environmental perceptions on self-rated health in Karakalpakstan. Self-rated health was assessed in May 2008 using a questionnaire on 881 randomly selected individuals from three communities. Here 55% reported ‘fair’ or ‘poor’ self-rated health, 41% of respondents reported significant environmental concern, and 48% were identified as emotionally distressed. Environmental concern was found to be significantly related to self-rated health and psychosocial health impacts. While these studies were focused on adults, making comparisons to the UHES difficult, questions about environmental concern and health problems were asked in the context of their families, thereby justifying their inclusion in this review. Overall, the results from these studies suggest that levels of self-rated health in Karakalpakstan and Khorezm are low relative to other parts of Uzbekistan and certainly well below what is found in Western Nations (Crighton et al. 2003a).
Discussion
From this review, the following conditions among children in the Aral Sea area were identified as being significant health issues: anemia, diarrheal diseases, hypercalciuria, renal tubular dysfunction, and high body burdens of persistent organic pollutants. The literature also indicates that infant mortality and under-five mortality rates are high compared to Uzbekistan as a whole, and to other countries at similar levels of development. On the other hand, respiratory function and various child development indicators were not found to be significantly worse in the Aral Sea area.
Before the major research findings are discussed, there are a number of limitations to this review that require some mention. First, while this review can be considered comprehensive for the literature published in English, research on the health of the Aral Sea population published in other languages have not been reviewed here. Another limitation relates to the reliability of the data in the national health reports, including the UDHS (Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997), MICS (UNICEF 2000, 2006), and the UHES (Ministry of Health Republic of Uzbekistan 2004), where the Uzbekistan government played some role. While we cannot provide proof here that results have been ‘adjusted’ to suit government interests, enough concerns have been raised by international and local experts (interviews conducted by Crighton in 2008 with local researchers, NGO, and Ministry of Health representatives in Karakalpakstan) and NGO reports (Médecins Sans Frontières 1999; 2004) that some skepticism of the data is warranted.
Anemia rates have been consistently reported to be very high in the Aral Sea area and can be linked indirectly to the region’s environmental problems. Anemia is caused by poverty and a diminished diet associated with the collapse of the fisheries and agricultural decline. Other dietary practices including the consumption of black tea and whole grains, known iron absorption inhibitors, are also likely to play a role (Hashizume et al. 2004). Direct links that have been hypothesized include parasite infestations and exposures to environmental contaminants although there is little evidence in the literature reviewed here to suggest these are significant factors. While anemia rates are particularly high in these studies, levels are a concern all over Central Asia (Hashizume et al. 2004; Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997). Further, anemia should be considered a particularly important issue in contaminated environments such as this as it can lead to increased susceptibility to poisoning from heavy metals, including lead, and can have an adverse effect on the immune system (Turgut et al. 2007; Wintergerst et al. 2007).
The literature is consistent in its reporting of high childhood diarrheal disease rates in the Aral Sea area. With the MICS (UNICEF 2000) and the UDHS (Institute of Obstetrics and Gynaecology [Uzbekistan] and Macro International Inc. 1997), reporting rates were well above the national average. In the case of the latter study, 8.6% of children under 5 years of age were reported to have had diarrhea in the past 2 weeks as compared to the national rate of 5.3%. High rates are indentified to be associated not only with the quality of the source drinking water but also with the quality of the piped water system, in-house sanitation, and household water-storage practices (Herbst et al. 2008; Semenza et al. 1998). The former relates directly to environmental conditions in the region that require sustainable water management strategies to address agricultural chemical and bacterial contamination run-off. Government actions in recent years have largely focused on the expansion of the piped water system although this is both expensive and prone to contamination due to low-pressure poor system maintenance, and does not benefit children outside major centers. Small-scale water treatment options (e.g. home chlorination, solar distillers, etc.), improved sullage disposal options, and basic hygiene education are among the alternative and more cost-effective solutions proposed in the literature (Herbst 2006; Semenza et al. 1998).
The environmental issue that has received the most attention in the literature relates to POPs. Results are consistent in demonstrating that levels of PCB, DDT, TCDD, and other highly toxic substances are present in children’s blood and breast milk at levels many times higher than any regional or international comparison groups. Studies have also found high levels of many of the same pollutants in various foods produced in cotton agricultural areas near the sea. While exposure and body burdens are relatively well understood, the health impacts are not. One study conducted in the Aral Sea context identified an inverse correlation between body mass index (BMI) with total PCB, DDT and DDE blood levels (Mazhitova et al. 1998). In other contexts, these contaminants have been found to negatively affect brain and motor skills development, pre- and post-natal growth, altered sex ratios, and respiratory health in children (Beard 2006; Ritter et al. 1996).
The literature suggests that respiratory disease and respiratory function as well as measures of development and growth including low birthweight, height-for-age, weight-for-height and weight-for-age are comparable to other areas of the county. In the case of development and growth indicators, local level analysis is required to determine if there are areas within Karakalpakstan where problems exist. As for respiratory diseases, although the evidence was not found to clearly support a relationship between dust exposure and respiratory function among children, it could be expected that lung function is affected by long-term exposures and therefore more likely to impact adults.
Tuberculosis, in particular its multi-drug resistant (MDR) and extreme drug resistant (XDR) strains, represents one of the most significant infectious disease issues among adults in the region today (Cox et al. 2007). TB is also an excellent marker of poverty and therefore as an indirect link to the disaster. While childhood TB has not been studied in the area, there is reason to believe that rates are also very high among children (Brent et al. 2008) and therefore warrants some mention. According to Zager and McNerney (2008), Karakalpakstan ranked third highest internationally for MDR-TB rates. More recently it was reported that 6% of TB patients in one study in Karakalpakstan were XDR cases (Cox et al. 2007). High rates of TB cannot be blamed directly on the environmental problems in the region; however, in contexts of environmental degradation, poverty, and overstretched health care systems, as is the case in much of the Aral Sea area, diseases like TB commonly flourish.
There remain many gaps in our understanding of the relationships between the Aral Sea environmental disaster and child health as this review has illustrated. Potential research questions that need to be addressed include the following: What are the health impacts of long-term ingestion of saline drinking water? What is the relationship between irrigation with sewage-contaminated surface water, helminthic diseases, and child malnutrition? What are the long-term impacts of dust exposure over the life course, and how and to what degree has child health been impacted by toxic agricultural chemicals in the environment? Unfortunately, the work to answer many of these questions has all but ended if the trend in publishing child health research is any indication (Fig. 2). The last identified studies were published 2008, and according to interviews conducted that year by Crighton with local and international researchers as well as NGO and Ministry of Health (Karakalpakstan and Uzbekistan) representatives, there was little evidence of planned or ongoing research. Explanations for why this is the case include the expulsion of many international NGOs from Uzbekistan several years prior, tightened regulations for those that were allowed to remain, and the denial of access to research funds for local NGOs from international donor agencies. Local resources for environmental monitoring and environmental health research were also reported to be increasingly limited.
While national policies are partly to blame for the lack of research going on today in the region, we would remiss to not also point the finger at the international community of health researchers for not advocating for more health research here. If, as the science shows, children in this area have some of the highest recorded body burdens of POPs, then answers to critical questions of global importance could be addressed by taking this population seriously. We must also ask ourselves why the international community has been so negligent for so long about the environmental situation in the region and the health impacts it is having. While some successes in Kazakhstan are now being realized with the help of World Bank funds used to build a dam to rehabilitate the small Aral in the North, few similar efforts have been made elsewhere. The blame for inaction must also be placed on region’s governments—Turkmenistan and Uzbekistan are among the only half a dozen nations that have not signed the Stockholm Convention on Persistent Organic Pollutants (UNEP 2008) or taken steps to limit childhood exposures.
Conclusion
The objective of this review was to assess what is known from the English literature about the relationship between children’s health and the environmental problems in the Aral Sea area. While the body of literature examining this relationship has grown considerably over the past two decades, the research momentum that began in the 1990s has all but disappeared, leaving many questions unanswered. Despite this, we have known enough for many years to warrant immediate action to address many of the major child health problems including anemia, high body burdens of POPs, diarrheal disease, and hypercalciuria. Regrettably this knowledge has not translated into action (Small and Bunce 2003). It will only be through more concerted efforts at the level of the international community and co-operation at the level of the region’s governments that we can hope to see change. Knowledge has its limits.
References
Abdullayev I (2010) Aral Sea crisis: large scale irrigation and its impact on drinking water quality and human health. Asian J Water Environ Pollut 7(1):63–69
Arustamov DL, Fayzieva DK, Nurullaev RB, Klyopov YY (2001) Study on the rate of the urolithiasis in the Aral Sea area and the quality of potable water. In: Fajzieva D, Brebbia CA (eds) Environmental health risk. WIT Press, Southampton, pp 105–111
Ataniyazova OA, Baumann RA, Liem AKD, Mukhopadhyay UA, Vogelaar EF, Boersma ER (2001) Levels of certain metals, organochlorine pesticides and dioxins in cord blood, maternal blood, human milk and some commonly used nutrients in the surroundings of the Aral Sea (Karakalpakstan, Republic of Uzbekistan). Acta Paediatr 90(7):801–808
Beard J (2006) DDT and human health. Sci Total Environ 355(1–3):78–89
Bennion P, Hubbard R, O’Hara S, Wiggs G, Wegerdt J, Lewis S, Small I, van der Meer J, Upshur R (2007) The impact of airborne dust on respiratory health in children living in the Aral Sea region. Int J Epidemiol 36(5):1103–1110
Bosch K, Erdinger L, Ingel F, Khussainova S, Utegenova E, Bresgen N, Eckl PM (2007) Evaluation of the toxicological properties of ground- and surface-water samples from the Aral Sea basin. Sci Total Environ 374(1):43–50
Brent AJ, Anderson ST, Kampmann B (2008) Childhood tuberculosis: out of sight, out of mind? Trans R Soc Trop Med Hyg 102(3):217–278
Carpenter DO, El-Qaderi S, Fayzieva D, Gilani AH, Hambartsumyan A, Herz K, Isobaev M, Kasymov O, Kudyakov R, Majitova Z, Mamadov E, Nemer L, Revich B, Stege P, Suk W, Upshur R, Yilmaz B, Zaineh K (2006) Children’s environmental health in Central Asia and the Middle East. Int J Occup Environ Health 12(4):362–368
Chiba M, Sera K, Hashizume M, Shimoda T, Sasaki S, Kunii O, Inaba Y (2004) Element concentrations in hair of children living in environmentally degraded districts of the East Aral Sea region. J Radioanalytical Nucl Chem 259(1):149–152
Cox HS, Kalon S, Allamuratova S, Sizaire V, Tigay ZN, R.-Gerdes S, Karimovich HA, Kebede Y, Mills C (2007) Multidrug-resistant tuberculosis treatment outcomes in Karakalpakstan, Uzbekistan: treatment complexity and XDR-TB among treatment failures. Plos One 2(11):e1126
Crighton EJ, Elliott SJ, Upshur R, Van Der Meer J, Small I (2003a) The Aral Sea disaster and self-rated health. Health Place 9(2):73–82
Crighton EJ, Elliott SJ, Van der Meer J, Small I, Upshur R (2003b) Impacts of an environmental disaster on psychosocial health and well-being in Karakalpakstan. Soc Sci Med 56(3):551–567
Erdinger L, Eckl P, Ingel F, Khussainova S, Utegenova E, Mann V, Gabrio T (2004) The Aral Sea disaster—human biomonitoring of Hg, as, HCB, DDE, and PCBs in children living in Aralsk and Akchi, Kazakhstan. Int J Hyg Environ Health 207(6):541–547
Giebel HN, Suleymanova D, Evans GW (1998) Anemia in young children of the Muynak District of Karakalpakstan, Uzbekistan: prevalence, type, and correlates. Am J Public Health 88(5):805–807
Glantz MH (1999) Sustainable development and creeping environmental problems in the Aral Sea region. In: Glantz MH (ed) Creeping environmental problems and sustainable development in the Aral Sea Basin. Cambridge University Press, New York, pp 1–25
Hashizume M, Kunii O, Sasaki S, Shimoda T, Wakai S, Mazhitova Z, Dauletbaev D, Caypil W, Aldiyarova M, Farmer A, Yamashiro Y, Chiba M (2003) Anemia and iron deficiency among schoolchildren in the Aral Sea region, Kazakhstan. J Trop Pediatr 49(3):172–177. doi:10.1093/tropej/49.3.172
Hashizume M, Shimoda T, Sasaki S, Kunii O, Caypil W, Dauletbaev D, Chiba M (2004) Anaemia in relation to low bioavailability of dietary iron among school-aged children in the Aral Sea region, Kazakhstan. Int J Food Sci Nutr 55(1):37–43
Herbst S (2006) Water, sanitation, hygiene and diarrheal diseases in the Aral Sea area (Khorezm, Uzbekistan). Ecology and Development Series Center for Development Research (ZEF), University of Bonn, Bonn
Herbst S, Fayzievab D, Kistemann T (2008) Risk factor analysis of diarrhoeal diseases in the Aral Sea area (Khorezm, Uzbekistan). Int J Environ Health Res 18(5):305–321
Hooper K (1999) Breast milk monitoring programs: world-wide early warning system for polyhalogenated POPs and for targeting studies in children’s environmental health. Environ Health Perspect 107(6):429–430
Hooper K, Petreas MX, Chuvakova T, Kazbekova G, Druz N, Semenova G et al (1998) Analysis of breast milk to assess exposure to chlorinated contaminants in Kazakstan: high levels of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) in agricultural villages in southern Kazakstan. Environ Health Perspect 106(12):797–806
Hooper K, Petreas MX, She J, Visita P, Winkler J, McKinney M, Mok M, Sy F, Garcha J, Gill M, Stephens RD, Semenova G, Sharmanov T, Chuvakova T, Hopper K (1997) Analysis of breast milk to assess exposure to chlorinated contaminants in Kazakstan: PCBs and organochlorine pesticides in southern Kazakstan. Environ Health Perspect 105(11):1250–1254
Institute of Obstetrics and Gynaecology [Uzbekistan], Macro International Inc. (1997) Uzbekistan Demographic and Health Survey (UDHS) 1996. Institute of Obstetrics and Gynecology and Macro International Inc., Calverton
Jensen S, Mazhitova Z, Zetterstrom R (1997) Environmental pollution and child health in the Aral Sea region in Kazakhstan. Sci Total Environ 206(2–3):187–193
Kaneko K, Chiba M, Hashizume M, Kunii O, Sasaki S, Shimoda T, Yamashiro Y, Dauletbaev D, Caypil W, Mazhitova Z (2002) Extremely high prevalence of hypercalciuria in children living in the Aral Sea region. Acta Paediatr 91(10):1116–1120
Kaneko K, Chiba M, Hashizume M, Kunii O, Sasaki S, Shimoda T, Yamashiro Y, Caypil W, Dauletbaev D (2003) Renal tubular dysfunction in children living in the Aral Sea region. Arch Dis Child 88(11):966–968
Kunii O, Hashizume M, Chiba M, Sasaki S, Shimoda T, Caypil W, Dauletbaev D (2003) Respiratory symptoms and pulmonary function among school-age children in the Aral Sea region. Arch Environ Health 58(11):676–682
Lutter C, Iyengar V, Barnes R, Chuvakova T, Kazbekova G, Sharmanov T (1998) Breast milk contamination in Kazakhstan: implications for infant feeding. Chemosphere 37(9–12):1761–1772
Mazhitova Z, Jensen S, Ritzen M, Zetterstrom R (1998) Chlorinated contaminants, growth and thyroid function in schoolchildren from the Aral Sea region in Kazakhstan. Acta Paediatr 87(9):991–995
Médecins Sans Frontières (1999) Review of health data. Médecins Sans Frontières, Tashkent
Médecins Sans Frontières (2004) Health data in Uzbekistan. Médecins Sans Frontières, Tashkent
Micklin P (2007) The Aral Sea disaster. Annu Rev Earth Planet Sci 35(1):47–72. doi:10.1146/annurev.earth.35.031306.140120
Ministry of Health Republic of Uzbekistan (2004) Uzbekistan Health Examination Survey (UHES) 2002
Morse C (1994) The prevalence and causes of anemia in Muynak District, Karakalpakstan, the Republic of Uzbekistan. A Chart Book (Results of a study). Crosslink International, Tashkent
Muntean N, Jermini M, Small I, Falzon D, Furst P, Migliorati G, Scortichini G, Forti AF, Anklam E, von Holst C, Niyazmatov B, Bahkridinov S, Aertgeerts R, Bertollini R, Tirado C, Kolb A (2003) Assessment of dietary exposure to some persistent organic pollutants in the Republic of Karakalpakstan of Uzbekistan. Environ Health Perspect 111(10):1306–1311
O’Hara SL, Wiggs GFS, Mamedov B, Davidson G, Hubbard RB (2000) Exposure to airborne dust contaminated with pesticide in the Aral Sea region. Lancet 355(9204):627–628
Petreas MX, Hooper K, She J, Visita P, Winkler J, McKinney M, Mok M, Sy F, Garcha J, Chuvakova T, Paltusheva T, Sharmanov T, Semenova G (1996) Analysis of human breast milk to assess exposure to chlorinated contaminants in Kazakhstan. Organohalogen Compd 30:20–23
Ritter L, Solomon K, Forget J, Stemeroff M, O’Leary C (1996) Persistent organic pollutants. United Nations Environment Programme. http://www.chem.unep.ch/pops/ritter/en/ritteren.pdf. Accessed 15 Mar 2010
Semenza JC, Roberts L, Henderson A, Bogan J, Rubin CH (1998) Water distribution system and diarrheal disease transmission: a case study in Uzbekistan. Am J Trop Med Hyg 59(6):941–946
Small I, Bunce N (2003) The Aral Sea disaster and the disaster of international assistance. J Int Aff 56(2):59
Small I, van der Meer J, Upshur REG (2001) Acting on an environmental health disaster: the case of the Aral Sea. Environ Health Perspect 109(6):547–549
Turgut S, Polat A, Inan M, Turgut G, Emmungil G, Bican M, Karakus TY, Genc O (2007) Interaction between anemia and blood levels of iron, zinc, copper, cadmium and lead in children. Indian J Pediatr 74(9):827–830
Ubaydullaev AM, Uzakova GT (2002) Prevalence of bronchial asthma in Uzbekistan. Problemy Tuberkuleza
UNEP (2008) Stockholm convention on persistent organic pollutants. http://chm.pops.int/default.aspx. Accessed 15 Apr 2010
UNICEF (2000) MICS, Multiple Indicator Cluster Survey. State Department of Statistics (Uzbekistan) and UNICEF–Uzbekistan, Tashkent
UNICEF (2006) MICS, Multiple Indicator Cluster Survey. State Department of Statistics (Uzbekistan) and UNICEF–Uzbekistan, Tashkent
Wintergerst ES, Maggini S, Hornig DH (2007) Contribution of selected vitamins and trace elements to immune function. Ann Nutr Metab 51(4):301–323
Zager EM, McNerney R (2008) Multidrug-resistant tuberculosis. BMC Infect Dis 8:10
Zaridze DG, Basieva T, Kabulov M, Day NE, Duffy SW (1992) Oesophageal cancer in the Republic of Karakalpakstan. Int J Epidemiol 21(4):643–648. doi:10.1093/ije/21.4.643
Acknowledgments
Thanks to Troy Hannah for his efforts in designing the Aral Sea area map. Dr. Upshur is supported by the Canada Research Chair in Primary Care Research.
Conflict of interest
The authors have no conflicts of interest to declare.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Crighton, E.J., Barwin, L., Small, I. et al. What have we learned? A review of the literature on children’s health and the environment in the Aral Sea area. Int J Public Health 56, 125–138 (2011). https://doi.org/10.1007/s00038-010-0201-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00038-010-0201-0