Abstract
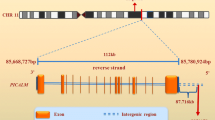
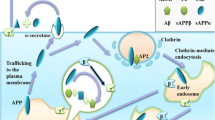
The increased production of the 42 aminoacids long beta-amyloid (Aβ42) peptide has been established as a causal mechanism of the familial early onset Alzheimer’s disease (AD). In contrast, the causal mechanisms of the late-onset AD (LOAD), that affects most AD patients, remain to be established. Indeed, Aβ42 accumulation has been detected more than 30 years before diagnosis. Thus, the mechanisms that control Aβ accumulation in LOAD likely go awry long before pathogenesis becomes detectable. Early on, APOE4 was identified as the biggest genetic risk factor for LOAD. However, since APOE4 is not present in all LOAD patients, genome-wide association studies of thousands of LOAD patients were undertaken to identify other genetic variants that could explain the development of LOAD. PICALM, BIN1, CD2AP, SORL1, and PLD3 are now with APOE4 among the identified genes at highest risk in LOAD that have been implicated in Aβ42 production. Recent evidence indicates that the regulation of the endocytic trafficking of the amyloid precursor protein (APP) and/or its secretases to and from sorting endosomes is determinant for Aβ42 production. Thus, here, we will review the described mechanisms, whereby these genetic risk factors can contribute to the enhanced endocytic production of Aβ42. Dissecting causal LOAD mechanisms of Aβ42 accumulation, underlying the contribution of each genetic risk factor, will be required to identify therapeutic targets for novel personalized preventive strategies.


Similar content being viewed by others
Bibliography
Tampellini D, Gouras GK (2010) Synapses, synaptic activity and intraneuronal abeta in Alzheimer’s disease. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2010.00013
Benilova I, Karran E, De Strooper B (2012) The toxic Aβ oligomer and Alzheimer’s disease: an emperor in need of clothes. Nat Neurosci 15:349–357. https://doi.org/10.1038/nn.3028
Almeida CG, Tampellini D, Takahashi RH et al (2005) Beta-amyloid accumulation in APP mutant neurons reduces PSD-95 and GluR1 in synapses. Neurobiol Dis 20:187–198. https://doi.org/10.1016/j.nbd.2005.02.008
Snyder EM, Nong Y, Almeida CG et al (2005) Regulation of NMDA receptor trafficking by amyloid-beta. Nat Neurosci 8:1051–1058. https://doi.org/10.1038/nn1503
Mucke L, Selkoe DJ (2012) Neurotoxicity of amyloid β-protein: synaptic and network dysfunction. Cold Spring Harb Perspect Med 2:a006338. https://doi.org/10.1101/cshperspect.a006338
Takahashi RH, Nagao T, Gouras GK (2017) Plaque formation and the intraneuronal accumulation of β-amyloid in Alzheimer’s disease. Pathol Int 67:185–193. https://doi.org/10.1111/pin.12520
Takahashi RH, Milner TA, Li F et al (2002) Intraneuronal Alzheimer abeta42 accumulates in multivesicular bodies and is associated with synaptic pathology. Am J Pathol 161:1869–1879
Pensalfini A, Albay R, Rasool S et al (2014) Intracellular amyloid and the neuronal origin of Alzheimer neuritic plaques. Neurobiol Dis 71:53–61. https://doi.org/10.1016/j.nbd.2014.07.011
Gouras GK, Almeida CG, Takahashi RH (2005) Intraneuronal Abeta accumulation and origin of plaques in Alzheimer’s disease. Neurobiol Aging 26:1235–1244. https://doi.org/10.1016/j.neurobiolaging.2005.05.022
Almeida CG, Takahashi RH, Gouras GK (2006) Beta-amyloid accumulation impairs multivesicular body sorting by inhibiting the ubiquitin-proteasome system. J Neurosci 26:4277–4288. https://doi.org/10.1523/JNEUROSCI.5078-05.2006
Sahlin C, Lord A, Magnusson K et al (2007) The Arctic Alzheimer mutation favors intracellular amyloid-beta production by making amyloid precursor protein less available to alpha-secretase. J Neurochem 101:854–862. https://doi.org/10.1111/j.1471-4159.2006.04443.x
Norvin D, Kim G, Baker-Nigh A, Geula C (2015) Accumulation and age-related elevation of amyloid-β within basal forebrain cholinergic neurons in the rhesus monkey. Neuroscience 298:102–111. https://doi.org/10.1016/j.neuroscience.2015.04.011
LaFerla FM, Green KN, Oddo S (2007) Intracellular amyloid-beta in Alzheimer’s disease. Nat Rev Neurosci 8:499–509. https://doi.org/10.1038/nrn2168
Mecozzi VJ, Berman DE, Simoes S et al (2014) Pharmacological chaperones stabilize retromer to limit APP processing. Nat Chem Biol 10:443–449. https://doi.org/10.1038/nchembio.1508
Matsuda S, Matsuda Y, Snapp EL, D’Adamio L (2011) Maturation of BRI2 generates a specific inhibitor that reduces APP processing at the plasma membrane and in endocytic vesicles. Neurobiol Aging 32:1400–1408. https://doi.org/10.1016/j.neurobiolaging.2009.08.005
Sun M, Asghar SZ, Zhang H (2016) The polarity protein Par3 regulates APP trafficking and processing through the endocytic adaptor protein Numb. Neurobiol Dis 93:1–11. https://doi.org/10.1016/j.nbd.2016.03.022
Zhang X, Song W (2013) The role of APP and BACE1 trafficking in APP processing and amyloid-β generation. Alzheimers Res Ther 5:46. https://doi.org/10.1186/alzrt211
Takahashi K, Niidome T, Akaike A et al (2008) Phosphorylation of amyloid precursor protein (APP) at Tyr687 regulates APP processing by alpha- and gamma-secretase. Biochem Biophys Res Commun 377:544–549. https://doi.org/10.1016/j.bbrc.2008.10.013
Tammineni P, Jeong YY, Feng T et al (2017) Impaired axonal retrograde trafficking of the retromer complex augments lysosomal deficits in Alzheimer’s disease neurons. Hum Mol Genet 26:4352–4366. https://doi.org/10.1093/hmg/ddx321
Saunders AM, Strittmatter WJ, Schmechel D et al (1993) Association of apolipoprotein E allele epsilon 4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 43:1467–1472
Corder EH, Saunders AM, Strittmatter WJ et al (1993) Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 261:921–923. https://doi.org/10.1126/science.8346443
Strittmatter WJ, Saunders AM, Schmechel D et al (1993) Apolipoprotein E: high-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease. Proc Natl Acad Sci USA 90:1977–1981
Rebeck GW, Reiter JS, Strickland DK, Hyman BT (1993) Apolipoprotein E in sporadic Alzheimer’s disease: allelic variation and receptor interactions. Neuron 11:575–580. https://doi.org/10.1016/0896-6273(93)90070-8
Chouraki V, Seshadri S, Theodore Friedmann JCD and SFG (2014) Chapter five—genetics of Alzheimer’s disease. Advances in Genetics. Academic Press, Cambridge, pp 245–294
Harold D, Abraham R, Hollingworth P et al (2009) Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer’s disease. Nat Genet 41:1088–1093. https://doi.org/10.1038/ng.440
Hollingworth P, Harold D, Sims R et al (2011) Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer’s disease. Nat Genet 43:429–435. https://doi.org/10.1038/ng.803
Naj AC, Jun G, Beecham GW et al (2011) Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer’s disease. Nat Genet 43:436–441. https://doi.org/10.1038/ng.801
Lambert JC, Ibrahim-Verbaas CA, Harold D et al (2013) Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat Genet 45:1452–1458. https://doi.org/10.1038/ng.2802
Cruchaga C, Karch CM, Jin SC et al (2014) Rare coding variants in the phospholipase D3 gene confer risk for Alzheimer’s disease. Nature 505:550–554. https://doi.org/10.1038/nature12825
Carmona S, Hardy J, Guerreiro R (2018) The genetic landscape of Alzheimer disease. Handb Clin Neurol 148:395–408. https://doi.org/10.1016/B978-0-444-64076-5.00026-0
Das U, Scott DA, Ganguly A et al (2013) Activity-induced convergence of APP and BACE-1 in acidic microdomains via an endocytosis-dependent pathway. Neuron 79:447–460. https://doi.org/10.1016/j.neuron.2013.05.035
Ehehalt R, Keller P, Haass C et al (2003) Amyloidogenic processing of the Alzheimer beta-amyloid precursor protein depends on lipid rafts. J Cell Biol 160:113–123. https://doi.org/10.1083/jcb.200207113
Kalvodova L, Kahya N, Schwille P et al (2005) Lipids as modulators of proteolytic activity of BACE: involvement of cholesterol, glycosphingolipids, and anionic phospholipids in vitro. J Biol Chem 280:36815–36823. https://doi.org/10.1074/jbc.M504484200
Koo EH, Squazzo SL (1994) Evidence that production and release of amyloid beta-protein involves the endocytic pathway. J Biol Chem 269:17386–17389
Lai A, Sisodia SS, Trowbridge IS (1995) Characterization of sorting signals in the beta-amyloid precursor protein cytoplasmic domain. J Biol Chem 270:3565–3573
Van der Kant R, Goldstein LSB (2015) Cellular functions of the amyloid precursor protein from development to dementia. Dev Cell 32:502–515. https://doi.org/10.1016/j.devcel.2015.01.022
Schneider A, Rajendran L, Honsho M et al (2008) Flotillin-dependent clustering of the amyloid precursor protein regulates its endocytosis and amyloidogenic processing in neurons. J Neurosci 28:2874–2882. https://doi.org/10.1523/JNEUROSCI.5345-07.2008
Marquer C, Devauges V, Cossec J-C et al (2011) Local cholesterol increase triggers amyloid precursor protein-Bace1 clustering in lipid rafts and rapid endocytosis. FASEB J 25:1295–1305. https://doi.org/10.1096/fj.10-168633
Kang MJ, Chung YH, Hwang CI et al (2006) Caveolin-1 upregulation in senescent neurons alters amyloid precursor protein processing. Exp Mol Med 38:126–133. https://doi.org/10.1038/emm.2006.16
Zerbinatti CV, Bu G (2005) LRP and Alzheimer’s disease. Rev Neurosci 16:123–135
Pietrzik CU, Busse T, Merriam DE et al (2002) The cytoplasmic domain of the LDL receptor-related protein regulates multiple steps in APP processing. EMBO J 21:5691–5700
Ye S, Huang Y, Müllendorff K et al (2005) Apolipoprotein (apo) E4 enhances amyloid beta peptide production in cultured neuronal cells: apoE structure as a potential therapeutic target. Proc Natl Acad Sci USA 102:18700–18705. https://doi.org/10.1073/pnas.0508693102
Sannerud R, Declerck I, Peric A et al (2011) ADP ribosylation factor 6 (ARF6) controls amyloid precursor protein (APP) processing by mediating the endosomal sorting of BACE1. Proc Natl Acad Sci USA 108:E559–E568. https://doi.org/10.1073/pnas.1100745108
Prabhu Y, Burgos PV, Schindler C et al (2012) Adaptor protein 2-mediated endocytosis of the β-secretase BACE1 is dispensable for amyloid precursor protein processing. Mol Biol Cell 23:2339–2351. https://doi.org/10.1091/mbc.E11-11-0944
Chia PZC, Toh WH, Sharples R et al (2013) Intracellular itinerary of internalised β-secretase, BACE1, and its potential impact on β-amyloid peptide biogenesis. Traffic 14:997–1013. https://doi.org/10.1111/tra.12088
Pastorino L, Ikin AF, Nairn AC et al (2002) The carboxyl-terminus of BACE contains a sorting signal that regulates BACE trafficking but not the formation of total A(beta). Mol Cell Neurosci 19:175–185. https://doi.org/10.1006/mcne.2001.1065
He X, Zhu G, Koelsch G et al (2003) Biochemical and structural characterization of the interaction of memapsin 2 (beta-secretase) cytosolic domain with the VHS domain of GGA proteins. Biochemistry 42:12174–12180. https://doi.org/10.1021/bi035199h
Cirrito JR, Kang J-E, Lee J et al (2008) Endocytosis is required for synaptic activity-dependent release of amyloid-beta in vivo. Neuron 58:42–51. https://doi.org/10.1016/j.neuron.2008.02.003
Zou L, Wang Z, Shen L et al (2007) Receptor tyrosine kinases positively regulate BACE activity and Amyloid-beta production through enhancing BACE internalization. Cell Res 17:389–401. https://doi.org/10.1038/cr.2007.5
Yan R, Vassar R (2014) Targeting the β secretase BACE1 for Alzheimer’s disease therapy. Lancet Neurol 13:319–329. https://doi.org/10.1016/S1474-4422(13)70276-X
Rajendran L, Schneider A, Schlechtingen G et al (2008) Efficient inhibition of the Alzheimer’s disease beta-secretase by membrane targeting. Science 320:520–523. https://doi.org/10.1126/science.1156609
Choy RW-Y, Cheng Z, Schekman R (2012) Amyloid precursor protein (APP) traffics from the cell surface via endosomes for amyloid β (Aβ) production in the trans-Golgi network. Proc Natl Acad Sci USA 109:E2077–E2082. https://doi.org/10.1073/pnas.1208635109
Buggia-Prévot V, Fernandez CG, Udayar V et al (2013) A function for EHD family proteins in unidirectional retrograde dendritic transport of BACE1 and Alzheimer’s disease Aβ production. Cell Rep 5:1552–1563. https://doi.org/10.1016/j.celrep.2013.12.006
Morel E, Chamoun Z, Lasiecka ZM et al (2013) Phosphatidylinositol-3-phosphate regulates sorting and processing of amyloid precursor protein through the endosomal system. Nat Commun 4:2250. https://doi.org/10.1038/ncomms3250
Sannerud R, Esselens C, Ejsmont P et al (2016) Restricted Location of PSEN2/γ-Secretase Determines Substrate Specificity and Generates an Intracellular Aβ Pool. Cell 166:193–208. https://doi.org/10.1016/j.cell.2016.05.020
Edgar JR, Willén K, Gouras GK, Futter CE (2015) ESCRTs regulate amyloid precursor protein sorting in multivesicular bodies and intracellular amyloid-β accumulation. J Cell Sci 128:2520–2528. https://doi.org/10.1242/jcs.170233
Udayar V, Buggia-Prévot V, Guerreiro RL et al (2013) A paired RNAi and RabGAP overexpression screen identifies Rab11 as a regulator of β-amyloid production. Cell Rep 5:1536–1551. https://doi.org/10.1016/j.celrep.2013.12.005
Takahashi RH, Almeida CG, Kearney PF et al (2004) Oligomerization of Alzheimer’s beta-amyloid within processes and synapses of cultured neurons and brain. J Neurosci 24:3592–3599. https://doi.org/10.1523/JNEUROSCI.5167-03.2004
Oropeza RL, Wekerle H, Werb Z (1987) Expression of apolipoprotein E by mouse brain astrocytes and its modulation by interferon-gamma. Brain Res 410:45–51
Bu G (2009) Apolipoprotein E and its receptors in Alzheimer’s disease: pathways, pathogenesis and therapy. Nat Rev Neurosci 10:333–344. https://doi.org/10.1038/nrn2620
Conejero-Goldberg C, Gomar JJ, Bobes-Bascaran T et al (2014) APOE2 enhances neuroprotection against Alzheimer’s disease through multiple molecular mechanisms. Mol Psychiatry 19:1243–1250. https://doi.org/10.1038/mp.2013.194
He X, Cooley K, Chung CHY et al (2007) Apolipoprotein receptor 2 and X11 alpha/beta mediate apolipoprotein E-induced endocytosis of amyloid-beta precursor protein and beta-secretase, leading to amyloid-beta production. J Neurosci 27:4052–4060. https://doi.org/10.1523/JNEUROSCI.3993-06.2007
Hoe H-S, Pocivavsek A, Dai H et al (2006) Effects of apoE on neuronal signaling and APP processing in rodent brain. Brain Res 1112:70–79. https://doi.org/10.1016/j.brainres.2006.07.035
Irizarry MC, Deng A, Lleo A et al (2004) Apolipoprotein E modulates gamma-secretase cleavage of the amyloid precursor protein. J Neurochem 90:1132–1143. https://doi.org/10.1111/j.1471-4159.2004.02581.x
Hopkins PCR, Sáinz-Fuertes R, Lovestone S (2011) The impact of a novel apolipoprotein E and amyloid-β protein precursor-interacting protein on the production of amyloid-β. J Alzheimers Dis 26:239–253. https://doi.org/10.3233/JAD-2011-102115
Cataldo AM, Peterhoff CM, Troncoso JC et al (2000) Endocytic pathway abnormalities precede amyloid β deposition in sporadic Alzheimer’s disease and Down syndrome: differential effects of APOE genotype and presenilin mutations. Am J Pathol 157:277–286
Zhao N, Liu C-C, Van Ingelgom AJ et al (2017) Apolipoprotein E4 impairs neuronal insulin signaling by trapping insulin receptor in the endosomes. Neuron 96(115–129):e5. https://doi.org/10.1016/j.neuron.2017.09.003
Rapp A, Gmeiner B, Hüttinger M (2006) Implication of apoE isoforms in cholesterol metabolism by primary rat hippocampal neurons and astrocytes. Biochimie 88:473–483. https://doi.org/10.1016/j.biochi.2005.10.007
Huang Y-WA, Zhou B, Wernig M, Südhof TC (2017) Apoe2, apoe3, and apoe4 differentially stimulate APP transcription and aβ secretion. Cell 168(427–441):e21. https://doi.org/10.1016/j.cell.2016.12.044
Castellano JM, Kim J, Stewart FR et al (2011) Human apoE isoforms differentially regulate brain amyloid-β peptide clearance. Sci Transl Med 3:89ra57. https://doi.org/10.1126/scitranslmed.3002156
Fryer JD, Simmons K, Parsadanian M et al (2005) Human apolipoprotein E4 alters the amyloid-beta 40:42 ratio and promotes the formation of cerebral amyloid angiopathy in an amyloid precursor protein transgenic model. J Neurosci 25:2803–2810. https://doi.org/10.1523/JNEUROSCI.5170-04.2005
Wildsmith KR, Holley M, Savage JC et al (2013) Evidence for impaired amyloid β clearance in Alzheimer’s disease. Alzheimers Res Ther 5:33. https://doi.org/10.1186/alzrt187
Verghese PB, Castellano JM, Garai K et al (2013) ApoE influences amyloid-β (Aβ) clearance despite minimal apoE/Aβ association in physiological conditions. Proc Natl Acad Sci USA 110:E1807–E1816. https://doi.org/10.1073/pnas.1220484110
Chen Y, Durakoglugil MS, Xian X, Herz J (2010) ApoE4 reduces glutamate receptor function and synaptic plasticity by selectively impairing ApoE receptor recycling. Proc Natl Acad Sci USA 107:12011–12016. https://doi.org/10.1073/pnas.0914984107
Jun G, Naj AC, Beecham GW et al (2010) Meta-analysis confirms CR1, CLU, and PICALM as alzheimer disease risk loci and reveals interactions with APOE genotypes. Arch Neurol 67:1473–1484. https://doi.org/10.1001/archneurol.2010.201
Carrasquillo MM, Belbin O, Hunter TA et al (2010) Replication of CLU, CR1, and PICALM associations with alzheimer disease. Arch Neurol 67:961–964. https://doi.org/10.1001/archneurol.2010.147
Ferrari R, Moreno JH, Minhajuddin AT et al (2012) Implication of common and disease specific variants in CLU, CR1, and PICALM. Neurobiol Aging 33(1846):e7–e18. https://doi.org/10.1016/j.neurobiolaging.2012.01.110
Moreno DJ, Ruiz S, Ríos Á et al (2017) Association of GWAS top genes with late-onset Alzheimer’s disease in Colombian population. Am J Alzheimers Dis Other Demen 32:27–35. https://doi.org/10.1177/1533317516679303
Naj AC, Jun G, Reitz C et al (2014) Effects of multiple genetic loci on age at onset in late-onset Alzheimer disease: a genome-wide association study. JAMA Neurol 71:1394–1404. https://doi.org/10.1001/jamaneurol.2014.1491
Lee JH, Cheng R, Barral S et al (2011) Identification of novel loci for Alzheimer disease and replication of CLU, PICALM, and BIN1 in Caribbean Hispanic individuals. Arch Neurol 68:320–328. https://doi.org/10.1001/archneurol.2010.292
Wang Z, Lei H, Zheng M et al (2016) Meta-analysis of the Association between Alzheimer Disease and Variants in GAB2, PICALM, and SORL1. Mol Neurobiol 53:6501–6510. https://doi.org/10.1007/s12035-015-9546-y
Xu W, Wang H-F, Tan L et al (2016) The impact of PICALM genetic variations on reserve capacity of posterior cingulate in AD continuum. Sci Rep 6:24480. https://doi.org/10.1038/srep24480
Mengel-From J, Christensen K, McGue M, Christiansen L (2011) Genetic variations in the CLU and PICALM genes are associated with cognitive function in the oldest old. Neurobiol Aging 32(554):e7–e11. https://doi.org/10.1016/j.neurobiolaging.2010.07.016
Biffi A, Anderson CD, Desikan RS et al (2010) Genetic variation and neuroimaging measures in Alzheimer disease. Arch Neurol 67:677–685. https://doi.org/10.1001/archneurol.2010.108
Dreyling MH, Martinez-Climent JA, Zheng M et al (1996) The t(10;11)(p13;q14) in the U937 cell line results in the fusion of the AF10 gene and CALM, encoding a new member of the AP-3 clathrin assembly protein family. Proc Natl Acad Sci USA 93:4804–4809
Miller SE, Sahlender DA, Graham SC et al (2011) The molecular basis for the endocytosis of small R-SNAREs by the clathrin adaptor CALM. Cell 147:1118–1131. https://doi.org/10.1016/j.cell.2011.10.038
Yao PJ, Petralia RS, Bushlin I et al (2005) Synaptic distribution of the endocytic accessory proteins AP180 and CALM. J Comp Neurol 481:58–69. https://doi.org/10.1002/cne.20362
Vanlandingham PA, Barmchi MP, Royer S et al (2014) AP180 couples protein retrieval to clathrin-mediated endocytosis of synaptic vesicles. Traffic 15:433–450. https://doi.org/10.1111/tra.12153
Meyerholz A, Hinrichsen L, Groos S et al (2005) Effect of clathrin assembly lymphoid myeloid leukemia protein depletion on clathrin coat formation. Traffic 6:1225–1234. https://doi.org/10.1111/j.1600-0854.2005.00355.x
Sahlender DA, Kozik P, Miller SE et al (2013) Uncoupling the functions of CALM in VAMP sorting and clathrin-coated pit formation. PLoS One 8:e64514. https://doi.org/10.1371/journal.pone.0064514
Koo SJ, Markovic S, Puchkov D et al (2011) SNARE motif-mediated sorting of synaptobrevin by the endocytic adaptors clathrin assembly lymphoid myeloid leukemia (CALM) and AP180 at synapses. Proc Natl Acad Sci USA 108:13540–13545. https://doi.org/10.1073/pnas.1107067108
Ando K, Brion J-P, Stygelbout V et al (2013) Clathrin adaptor CALM/PICALM is associated with neurofibrillary tangles and is cleaved in Alzheimer’s brains. Acta Neuropathol 125:861–878. https://doi.org/10.1007/s00401-013-1111-z
Thomas RS, Lelos MJ, Good MA, Kidd EJ (2011) Clathrin-mediated endocytic proteins are upregulated in the cortex of the Tg2576 mouse model of Alzheimer’s disease-like amyloid pathology. Biochem Biophys Res Commun 415:656–661. https://doi.org/10.1016/j.bbrc.2011.10.131
Parikh I, Fardo DW, Estus S (2014) Genetics of PICALM expression and Alzheimer’s disease. PLoS One 9:e91242. https://doi.org/10.1371/journal.pone.0091242
Xiao Q, Gil S-C, Yan P et al (2012) Role of phosphatidylinositol clathrin assembly lymphoid-myeloid leukemia (PICALM) in intracellular amyloid precursor protein (APP) processing and amyloid plaque pathogenesis. J Biol Chem 287:21279–21289. https://doi.org/10.1074/jbc.M111.338376
Boehm C, Kaden D, St. George-Hyslop P (2012) Picalm but not bin1 alters the secretion of beta-amyloid peptide. Alzheimers Dement 8:P652. https://doi.org/10.1016/j.jalz.2012.05.2175
Thomas RS, Henson A, Gerrish A et al (2016) Decreasing the expression of PICALM reduces endocytosis and the activity of β-secretase: implications for Alzheimer’s disease. BMC Neurosci 17:50. https://doi.org/10.1186/s12868-016-0288-1
Kanatsu K, Morohashi Y, Suzuki M et al (2014) Decreased CALM expression reduces Aβ42 to total Aβ ratio through clathrin-mediated endocytosis of γ-secretase. Nat Commun 5:3386. https://doi.org/10.1038/ncomms4386
Kanatsu K, Hori Y, Takatori S et al (2016) Partial loss of CALM function reduces Aβ42 production and amyloid deposition in vivo. Hum Mol Genet 25:3988–3997. https://doi.org/10.1093/hmg/ddw239
Tian Y, Chang JC, Fan EY et al (2013) Adaptor complex AP2/PICALM, through interaction with LC3, targets Alzheimer’s APP-CTF for terminal degradation via autophagy. Proc Natl Acad Sci USA 110:17071–17076. https://doi.org/10.1073/pnas.1315110110
Zhao Z, Sagare AP, Ma Q et al (2015) Central role for PICALM in amyloid-β blood-brain barrier transcytosis and clearance. Nat Neurosci 18:978–987. https://doi.org/10.1038/nn.4025
Gimber N, Tadeus G, Maritzen T et al (2015) Diffusional spread and confinement of newly exocytosed synaptic vesicle proteins. Nat Commun 6:8392. https://doi.org/10.1038/ncomms9392
Mercer JL, Argus JP, Crabtree DM et al (2015) Modulation of PICALM levels perturbs cellular cholesterol homeostasis. PLoS One 10:e0129776. https://doi.org/10.1371/journal.pone.0129776
Seshadri S, Fitzpatrick AL, Ikram MA et al (2010) Genome-wide analysis of genetic loci associated with Alzheimer disease. JAMA 303:1832–1840. https://doi.org/10.1001/jama.2010.574
Wijsman EM, Pankratz ND, Choi Y et al (2011) Genome-wide association of familial late-onset Alzheimer’s disease replicates BIN1 and CLU and nominates CUGBP2 in interaction with APOE. PLoS Genet 7:e1001308. https://doi.org/10.1371/journal.pgen.1001308
Hu X, Pickering E, Liu YC et al (2011) Meta-analysis for genome-wide association study identifies multiple variants at the BIN1 locus associated with late-onset Alzheimer’s disease. PLoS One 6:e16616. https://doi.org/10.1371/journal.pone.0016616
Kamboh MI, Demirci FY, Wang X et al (2012) Genome-wide association study of Alzheimer’s disease. Transl Psychiatry 2:e117. https://doi.org/10.1038/tp.2012.45
Barral S, Bird T, Goate A et al (2012) Genotype patterns at PICALM, CR1, BIN1, CLU, and APOE genes are associated with episodic memory. Neurology 78:1464–1471. https://doi.org/10.1212/WNL.0b013e3182553c48
Tan M-S, Yu J-T, Jiang T et al (2014) Genetic variation in BIN1 gene and Alzheimer’s disease risk in Han Chinese individuals. Neurobiol Aging 35(1781):e1–e8. https://doi.org/10.1016/j.neurobiolaging.2014.01.151
Vardarajan BN, Ghani M, Kahn A et al (2015) Rare coding mutations identified by sequencing of Alzheimer disease genome-wide association studies loci. Ann Neurol 78:487–498. https://doi.org/10.1002/ana.24466
Sakamuro D, Elliott KJ, Wechsler-Reya R, Prendergast GC (1996) BIN1 is a novel MYC-interacting protein with features of a tumour suppressor. Nat Genet 14:69–77. https://doi.org/10.1038/ng0996-69
David C, McPherson PS, Mundigl O, de Camilli P (1996) A role of amphiphysin in synaptic vesicle endocytosis suggested by its binding to dynamin in nerve terminals. Proc Natl Acad Sci USA 93:331–335
Leprince C, Romero F, Cussac D et al (1997) A new member of the amphiphysin family connecting endocytosis and signal transduction pathways. J Biol Chem 272:15101–15105. https://doi.org/10.1074/jbc.272.24.15101
Micheva KD, Kay BK, McPherson PS (1997) Synaptojanin forms two separate complexes in the nerve terminal. Interactions with endophilin and amphiphysin. J Biol Chem 272:27239–27245
Prokic I, Cowling BS, Laporte J (2014) Amphiphysin 2 (BIN1) in physiology and diseases. J Mol Med 92:453–463. https://doi.org/10.1007/s00109-014-1138-1
Ramjaun AR, Micheva KD, Bouchelet I, McPherson PS (1997) Identification and characterization of a nerve terminal-enriched amphiphysin isoform. J Biol Chem 272:16700–16706. https://doi.org/10.1074/jbc.272.26.16700
Butler MH, David C, Ochoa GC et al (1997) Amphiphysin II (SH3P9; BIN1), a member of the amphiphysin/Rvs family, is concentrated in the cortical cytomatrix of axon initial segments and nodes of ranvier in brain and around T tubules in skeletal muscle. J Cell Biol 137:1355–1367
Wigge P, Köhler K, Vallis Y et al (1997) Amphiphysin heterodimers: potential role in clathrin-mediated endocytosis. Mol Biol Cell 8:2003–2015
Ramjaun AR, McPherson PS (1998) Multiple amphiphysin II splice variants display differential clathrin binding: identification of two distinct clathrin-binding sites. J Neurochem 70:2369–2376
Di Paolo G, Sankaranarayanan S, Wenk MR et al (2002) Decreased synaptic vesicle recycling efficiency and cognitive deficits in amphiphysin 1 knockout mice. Neuron 33:789–804
Muller AJ, Baker JF, DuHadaway JB et al (2003) Targeted disruption of the murine Bin1/Amphiphysin II gene does not disable endocytosis but results in embryonic cardiomyopathy with aberrant myofibril formation. Mol Cell Biol 23:4295–4306
Pant S, Sharma M, Patel K et al (2009) AMPH-1/Amphiphysin/Bin1 functions with RME-1/Ehd1 in endocytic recycling. Nat Cell Biol 11:1399–1410. https://doi.org/10.1038/ncb1986
Chapuis J, Hansmannel F, Gistelinck M et al (2013) Increased expression of BIN1 mediates Alzheimer genetic risk by modulating tau pathology. Mol Psychiatry 18:1225–1234. https://doi.org/10.1038/mp.2013.1
Karch CM, Jeng AT, Nowotny P et al (2012) Expression of novel Alzheimer’s disease risk genes in control and Alzheimer’s disease brains. PLoS One 7:e50976. https://doi.org/10.1371/journal.pone.0050976
Glennon EBC, Whitehouse IJ, Miners JS et al (2013) BIN1 is decreased in sporadic but not familial Alzheimer’s disease or in aging. PLoS One 8:e78806. https://doi.org/10.1371/journal.pone.0078806
Holler CJ, Davis PR, Beckett TL et al (2014) Bridging integrator 1 (BIN1) protein expression increases in the Alzheimer’s disease brain and correlates with neurofibrillary tangle pathology. J Alzheimers Dis 42:1221–1227. https://doi.org/10.3233/JAD-132450
De Rossi P, Buggia-Prévot V, Clayton BLL et al (2016) Predominant expression of Alzheimer’s disease-associated BIN1 in mature oligodendrocytes and localization to white matter tracts. Mol Neurodegener 11:59. https://doi.org/10.1186/s13024-016-0124-1
Miyagawa T, Ebinuma I, Morohashi Y et al (2016) BIN1 regulates BACE1 intracellular trafficking and amyloid-β production. Hum Mol Genet 25:2948–2958. https://doi.org/10.1093/hmg/ddw146
Ubelmann F, Burrinha T, Salavessa L et al (2017) Bin1 and CD2AP polarise the endocytic generation of beta-amyloid. EMBO Rep 18:102–122. https://doi.org/10.15252/embr.201642738
Calafate S, Flavin W, Verstreken P, Moechars D (2016) Loss of bin1 promotes the propagation of tau pathology. Cell Rep 17:931–940. https://doi.org/10.1016/j.celrep.2016.09.063
Shulman JM, Chen K, Keenan BT et al (2013) Genetic susceptibility for Alzheimer disease neuritic plaque pathology. JAMA Neurol 70:1150–1157. https://doi.org/10.1001/jamaneurol.2013.2815
Chen H, Wu G, Jiang Y et al (2015) Analyzing 54,936 samples supports the association between CD2AP rs9349407 polymorphism and Alzheimer’s disease susceptibility. Mol Neurobiol 52:1–7. https://doi.org/10.1007/s12035-014-8834-2
Dustin ML, Olszowy MW, Holdorf AD et al (1998) A novel adaptor protein orchestrates receptor patterning and cytoskeletal polarity in T-cell contacts. Cell 94:667–677. https://doi.org/10.1016/S0092-8674(00)81608-6
Cormont M, Metón I, Mari M et al (2003) CD2AP/CMS regulates endosome morphology and traffic to the degradative pathway through its interaction with Rab4 and c-Cbl. Traffic 4:97–112
Gauthier NC, Monzo P, Gonzalez T et al (2007) Early endosomes associated with dynamic F-actin structures are required for late trafficking of H. pylori VacA toxin. J Cell Biol 177:343–354. https://doi.org/10.1083/jcb.200609061
Monzo P, Mari M, Kaddai V, et al. (2005) CD2AP, Rabip4, and Rabip4′: Analysis of Interaction with Rab4a and Regulation of Endosomes Morphology. Meth Enzymol. Academic Press, Cambridge, pp 107–118
Kobayashi S, Sawano A, Nojima Y et al (2004) The c-Cbl/CD2AP complex regulates VEGF-induced endocytosis and degradation of Flt-1 (VEGFR-1). FASEB J 18:929–931. https://doi.org/10.1096/fj.03-0767fje
Lynch DK, Winata SC, Lyons RJ et al (2003) A Cortactin-CD2-associated protein (CD2AP) complex provides a novel link between epidermal growth factor receptor endocytosis and the actin cytoskeleton. J Biol Chem 278:21805–21813. https://doi.org/10.1074/jbc.M211407200
Tang VW, Brieher WM (2013) FSGS3/CD2AP is a barbed-end capping protein that stabilizes actin and strengthens adherens junctions. J Cell Biol 203:815–833. https://doi.org/10.1083/jcb.201304143
Zhao J, Bruck S, Cemerski S et al (2013) CD2AP links cortactin and capping protein at the cell periphery to facilitate formation of lamellipodia. Mol Cell Biol 33:38–47. https://doi.org/10.1128/MCB.00734-12
Li C, Ruotsalainen V, Tryggvason K et al (2000) CD2AP is expressed with nephrin in developing podocytes and is found widely in mature kidney and elsewhere. Am J Physiol Renal Physiol 279:F785–F792. https://doi.org/10.1152/ajprenal.2000.279.4.F785
Wolf G, Stahl RAK (2003) CD2-associated protein and glomerular disease. The Lancet 362:1746–1748. https://doi.org/10.1016/S0140-6736(03)14856-8
Peitsch WK, Hofmann I, Endlich N et al (2003) Cell biological and biochemical characterization of drebrin complexes in mesangial cells and podocytes of renal glomeruli. J Am Soc Nephrol 14:1452–1463
Kobayashi N (2002) Mechanism of the process formation; podocytes vs. neurons. Microsc Res Tech 57:217–223. https://doi.org/10.1002/jemt.10077
Tao Q-Q, Liu Z-J, Sun Y-M et al (2017) Decreased gene expression of CD2AP in Chinese patients with sporadic Alzheimer’s disease. Neurobiol Aging. https://doi.org/10.1016/j.neurobiolaging.2017.03.013
Liao F, Jiang H, Srivatsan S et al (2015) Effects of CD2-associated protein deficiency on amyloid-β in neuroblastoma cells and in an APP transgenic mouse model. Mol Neurodegener 10:12. https://doi.org/10.1186/s13024-015-0006-y
Cochran JN, Rush T, Buckingham SC, Roberson ED (2015) The Alzheimer’s disease risk factor CD2AP maintains blood-brain barrier integrity. Hum Mol Genet 24:6667–6674. https://doi.org/10.1093/hmg/ddv371
Rogaeva E, Meng Y, Lee JH et al (2007) The neuronal sortilin-related receptor SORL1 is genetically associated with Alzheimer disease. Nat Genet 39:168–177. https://doi.org/10.1038/ng1943
Lee JH, Cheng R, Schupf N et al (2007) The association between genetic variants in SORL1 and Alzheimer disease in an urban, multiethnic, community-based cohort. Arch Neurol 64:501–506. https://doi.org/10.1001/archneur.64.4.501
Feng X, Hou D, Deng Y et al (2015) SORL1 gene polymorphism association with late-onset Alzheimer’s disease. Neurosci Lett 584:382–389. https://doi.org/10.1016/j.neulet.2014.10.055
Piscopo P, Tosto G, Belli C et al (2015) SORL1 gene is associated with the conversion from mild cognitive impairment to Alzheimer’s disease. J Alzheimers Dis 46:771–776. https://doi.org/10.3233/JAD-141551
Verheijen J, Van den Bossche T, van der Zee J et al (2016) A comprehensive study of the genetic impact of rare variants in SORL1 in European early-onset Alzheimer’s disease. Acta Neuropathol 132:213–224. https://doi.org/10.1007/s00401-016-1566-9
Miyashita A, Koike A, Jun G et al (2013) SORL1 is genetically associated with late-onset Alzheimer’s disease in Japanese, Koreans and Caucasians. PLoS One 8:e58618. https://doi.org/10.1371/journal.pone.0058618
Young JE, Boulanger-Weill J, Williams DA et al (2015) Elucidating molecular phenotypes caused by the SORL1 Alzheimer’s disease genetic risk factor using human induced pluripotent stem cells. Cell Stem Cell 16:373–385. https://doi.org/10.1016/j.stem.2015.02.004
Reitz C, Tokuhiro S, Clark LN et al (2011) SORCS1 alters amyloid precursor protein processing and variants may increase Alzheimer’s disease risk. Ann Neurol 69:47–64. https://doi.org/10.1002/ana.22308
Schmidt V, Subkhangulova A, Willnow TE (2017) Sorting receptor SORLA: cellular mechanisms and implications for disease. Cell Mol Life Sci 74:1475–1483. https://doi.org/10.1007/s00018-016-2410-z
Klinger SC, Højland A, Jain S et al (2016) Polarized trafficking of the sorting receptor SorLA in neurons and MDCK cells. FEBS J 283:2476–2493. https://doi.org/10.1111/febs.13758
Caglayan S, Takagi-Niidome S, Liao F et al (2014) Lysosomal sorting of amyloid-β by the SORLA receptor is impaired by a familial Alzheimer’s disease mutation. Sci Transl Med 6:223ra20. https://doi.org/10.1126/scitranslmed.3007747
Andersen OM, Rudolph I-M, Willnow TE (2016) Risk factor SORL1: from genetic association to functional validation in Alzheimer’s disease. Acta Neuropathol 132:653–665. https://doi.org/10.1007/s00401-016-1615-4
Yu L, Chibnik LB, Srivastava GP et al (2015) Association of Brain DNA methylation in SORL1, ABCA7, HLA-DRB5, SLC24A4, and BIN1 with pathological diagnosis of Alzheimer disease. JAMA Neurol 72:15–24. https://doi.org/10.1001/jamaneurol.2014.3049
Grear KE, Ling I-F, Simpson JF et al (2009) Expression of SORL1 and a novel SORL1 splice variant in normal and Alzheimers disease brain. Mol Neurodegener 4:46. https://doi.org/10.1186/1750-1326-4-46
Fjorback AW, Seaman M, Gustafsen C et al (2012) Retromer binds the FANSHY sorting motif in SorLA to regulate amyloid precursor protein sorting and processing. J Neurosci 32:1467–1480. https://doi.org/10.1523/JNEUROSCI.2272-11.2012
Dodson SE, Andersen OM, Karmali V et al (2008) Loss of LR11/SORLA enhances early pathology in a mouse model of amyloidosis: evidence for a proximal role in Alzheimer’s disease. J Neurosci 28:12877–12886. https://doi.org/10.1523/JNEUROSCI.4582-08.2008
Bhalla A, Vetanovetz CP, Morel E et al (2012) The location and trafficking routes of the neuronal retromer and its role in amyloid precursor protein transport. Neurobiol Dis 47:126–134. https://doi.org/10.1016/j.nbd.2012.03.030
Bonifacino JS, Hurley JH (2008) Retromer. Curr Opin Cell Biol 20:427–436. https://doi.org/10.1016/j.ceb.2008.03.009
Eggert S, Gonzalez AC, Thomas C et al (2017) Dimerization leads to changes in APP (amyloid precursor protein) trafficking mediated by LRP1 and SorLA. Cell Mol Life Sci 75:1–22. https://doi.org/10.1007/s00018-017-2625-7
Vieira SI, Rebelo S, Esselmann H et al (2010) Retrieval of the Alzheimer’s amyloid precursor protein from the endosome to the TGN is S655 phosphorylation state-dependent and retromer-mediated. Mol Neurodegener 5:40. https://doi.org/10.1186/1750-1326-5-40
Yajima R, Tokutake T, Koyama A et al (2015) ApoE-isoform-dependent cellular uptake of amyloid-β is mediated by lipoprotein receptor LR11/SorLA. Biochem Biophys Res Commun 456:482–488. https://doi.org/10.1016/j.bbrc.2014.11.111
Na J-Y, Song K, Lee J-W et al (2017) Sortilin-related receptor 1 interacts with amyloid precursor protein and is activated by 6-shogaol, leading to inhibition of the amyloidogenic pathway. Biochem Biophys Res Commun 484:890–895. https://doi.org/10.1016/j.bbrc.2017.02.029
Schulte EC, Kurz A, Alexopoulos P et al (2015) Excess of rare coding variants in PLD3 in late- but not early-onset Alzheimer’s disease. Hum Genome Var 2:14028. https://doi.org/10.1038/hgv.2014.28
Van der Lee SJ, Holstege H, Wong TH et al (2015) PLD3 variants in population studies. Nature 520:E2–E3. https://doi.org/10.1038/nature14038
Cacace R, Van den Bossche T, Engelborghs S et al (2015) Rare variants in PLD3 do not affect risk for early-onset Alzheimer disease in a European consortium cohort. Hum Mutat 36:1226–1235. https://doi.org/10.1002/humu.22908
Wang C, Wang H-F, Tan M-S et al (2016) Impact of common variations in PLD3 on neuroimaging phenotypes in non-demented elders. Mol Neurobiol 53:4343–4351. https://doi.org/10.1007/s12035-015-9370-4
Lin E, Tsai S-J, Kuo P-H et al (2017) Association and interaction effects of Alzheimer’s disease-associated genes and lifestyle on cognitive aging in older adults in a Taiwanese population. Oncotarget 8:24077–24087. https://doi.org/10.18632/oncotarget.15269
Donaldson JG (2009) Phospholipase D in endocytosis and endosomal recycling pathways. Biochim Biophys Acta 1791:845–849. https://doi.org/10.1016/j.bbalip.2009.05.011
Jenkins GM, Frohman MA (2005) Phospholipase D: a lipid centric review. Cell Mol Life Sci 62:2305–2316. https://doi.org/10.1007/s00018-005-5195-z
Osisami M, Ali W, Frohman MA (2012) A role for phospholipase D3 in myotube formation. PLoS One 7:e33341. https://doi.org/10.1371/journal.pone.0033341
Pedersen KM, Finsen B, Celis JE, Jensen NA (1998) Expression of a novel murine phospholipase D homolog coincides with late neuronal development in the forebrain. J Biol Chem 273:31494–31504
Satoh J-I, Kino Y, Yamamoto Y et al (2014) PLD3 is accumulated on neuritic plaques in Alzheimer’s disease brains. Alzheimers Res Ther 6:70. https://doi.org/10.1186/s13195-014-0070-5
Nagaoka-Yasuda R, Matsuo N, Perkins B et al (2007) An RNAi-based genetic screen for oxidative stress resistance reveals retinol saturase as a mediator of stress resistance. Free Radic Biol Med 43:781–788. https://doi.org/10.1016/j.freeradbiomed.2007.05.008
Fazzari P, Horre K, Arranz AM et al (2017) PLD3 gene and processing of APP. Nature 541:E1–E2. https://doi.org/10.1038/nature21030
Acknowledgements
We thank Inês Figueira for revising the manuscript. The Almeida lab has been supported by a Marie Curie Integration Grant (334366 TrafficInAD FP7-PEOPLE-2012-CIG; Marie Curie Actions, EC). iNOVA4Health—UID/Multi/04462/2013, a program financially supported by Fundação para a Ciência e Tecnologia (FCT)/Ministério da Educação e Ciência, through national funds and co-funded by FEDER under the PT2020 Partnership Agreement is acknowledged. CGA is funded by Investigator FCT (IF/00998/2012, FCT). FM has been the recipient of an FCT doctoral fellowship (PD/BD/128344/2017), CP has been the recipient of an FCT doctoral fellowship (SFRH/BD/128374/2017) and TB has been the recipient of an FCT doctoral fellowship (SFRH/BD/131513/2017).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guimas Almeida, C., Sadat Mirfakhar, F., Perdigão, C. et al. Impact of late-onset Alzheimer’s genetic risk factors on beta-amyloid endocytic production. Cell. Mol. Life Sci. 75, 2577–2589 (2018). https://doi.org/10.1007/s00018-018-2825-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-018-2825-9