Abstract
Objective
The CagA (cytotoxin-related gene A, CagA) protein is an important factor for the pathogenicity of Helicobacter pylori (H. pylori). Although H. pylori has previously been shown to activate the NLRP3 inflammasome, it remains unclear what role CagA plays in this process. In the current study, we aimed to investigate the effect of CagA on NLRP3 activation and how it is linked to gastric cancer cell migration and invasion.
Methods
CagA positive H. pylori strain (Hp/CagA+) and CagA gene knockout mutant (Hp/ΔCagA) infected and the pcDNA3.1/CagA plasmid transfected gastric epithelial cell lines, respectively. The morphological alterations of cells under a microscope; the NLRP3 inflammasome-related markers: NLRP3, caspase-1, and ASC protein levels were detected by Western blot, IL-1β and IL-18 levels were determined by ELISA; cell migration and invasion were determined by transwell assay; and the pyroptosis levels and intracellular ROS were determined by flow cytometry analysis. Then, pretreated with 5 mM NAC for 2 h and subsequently transfected with the pcDNA3.1/CagA plasmid for 48 h, the effects of NAC pretreatment on CagA-induced NLRP3 inflammasome-related markers expression and cell pyroptosis were examined, finally assessed the effect of CagA on migration and invasion in NLRP3-silenced cells.
Results
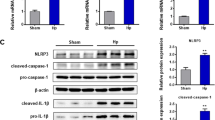
We found that Hp/CagA+ strain infection and pcDNA3.1/CagA vector transfection result in NLRP3 inflammasome activation, generation of intracellular ROS, and increased invasion and migration of gastric cancer cells. Moreover, we found that ROS inhibition via NAC effectively blocks NLRP3 activation and pyroptosis. Silencing of NLRP3 reduces the effects of CagA on gastric cancer cell migration and invasion.
Conclusion
Our study shows that CagA can promote the invasion and migration of gastric cancer cells by activating NLRP3 inflammasome pathway. These findings provide novel insights into the mechanism of gastric cancer induction by H. pylori.










Similar content being viewed by others
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA A Cancer J Clin. 2021;71(1):1542–4863.
Wang F, Meng W, Wang B, et al. Helicobacter pylori-induced gastric inflammation and gastric cancer[J]. Cancer Lett. 2014;345(2):196–202.
Plummer M, De Martel C, Vignat J, et al. Global burden of cancers attributable to infections in 2012: a synthetic analysis[J]. Lancet Glob Health. 2016;4(9):e609–16.
Ahn HJ, Dong SL. Helicobacter pylori in gastric carcinogenesis[J]. World J Gastrointest Oncol. 2015;7(12):455–65.
Rojasrengifo DF, Alvarezsilva MC, Ulloaguerrero CP, et al. Intramolecular energies of the cytotoxic protein CagA of Helicobacter pylori as a possible descriptor of strains’ pathogenicity level[J]. Comput Biol Chem. 2018;76:17–22.
Westmeier D, Posselt G, Hahlbrock A, et al. Nanoparticle binding attenuates the pathobiology of gastric cancer-associated Helicobacter pylori[J]. Nanoscale. 2018;10(3):1453–63.
Torres J, Pérez-Pérez GI, Leal-Herrera Y, et al. Infection with CagA+Helicobacter pylori strains as a possible predictor of risk in the development of gastric adenocarcinoma in Mexico[J]. Int J Cancer. 1998;78(3):298–300.
Franco AT, Johnston E, Krishna U, et al. Regulation of gastric cancinogenesis by Helicobacter pylorivirolence factors. Cancer Res. 2008;68:379–87.
Zhang B, Hu L, Zang M, et al. Helicobacter pylori CagA induces tumor suppressor gene hypermethylation by upregulating DNMT1 via AKT-NFκB pathway in gastric cancer development[J]. Oncotarget. 2016;7(9):9788–800.
Wen S, Moss SF. Helicobaxter pylori virulence factors in gastric carcinogenesis. Cancer Lett. 2009;282:1–8.
Balkwill F, Coussens LM. Cancer: an inflammatory link[J]. Nature. 2004;431(7007):405–6.
Martinon F, Burns K, Tschopp J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta[J]. Mol Cell. 2002;10(2):417–26.
Broz P, Dixit VM. Inflammasomes: mechanism of assembly, regulation and signalling. Nat Rev Immunol. 2016;16:407–20.
Jiang D, Chen S, Sun R, Zhang X, Wang D. The NLRP3 inflammasome: role in metabolic disorders and regulation by metabolic pathways. Cancer Lett. 2018;419:8–19.
El-Sharkawy LY, Brough D, Freeman S. Inhibiting the NLRP3 inflammasome. Molecules. 2020;25(23):5533.
Wei-guoLIN HU, Jin-ye SONG, Qi-bin. Progress in the relationship of inflammasome and tumor metastasis[J]. J Chin Oncol. 2016;161(02):23–9.
Wang H, Luo Q, Feng X, et al. NLRP3 promotes tumor growth and metastasis in human oral squamous cell carcinoma[J]. BMC Cancer. 2018;18(1):500–2.
Xiang L, Sheng L, Jingjing L, et al. Helicobacter pylori induces IL-1β and IL-18 production in human monocytic cell line through activation of NLRP3 inflammasome via ROS signaling pathway[J]. Pathog Dis. 2015;73(4):1–8.
Semper RP, Mejías-Luque R, Groß C, et al. Helicobacter pylori–induced IL-1β secretion in innate immune cells is regulated by the NLRP3 inflammasome and requires the cag Pathogenicity Island[J]. J Immunol. 2014;193(7):3566–76.
Pachathundikandi SK, Backert S. Helicobacter pylori controls NLRP3 expression by regulating hsa-miR-223-3p and IL-10 in cultured and primary human immune cells[J]. Innate Immun. 2018;24(1):11–23.
Zhengmei L, Qiong J, Jianjiang Z, et al. Effect of Helicobacter pylori infection on NLRP3 inflammasome activation[J]. J Shandong Univ. 2016;54(3):9–13.
Correa P. Human gastric carcinogenesis: a multistep and multifactorial process—first American Cancer Society award lecture on cancer epidemiology and prevention[J]. Can Res. 1992;52(24):6735–40.
Huang JY, Sweeney EG, Sigal M, et al. Chemodetection and destruction of host urea allows Helicobacter pylori to locate the epithelium[J]. Cell Host Microbe. 2015;18(2):147–56.
Yamaoka Y. Helicobacter pylori typing as a tool for tracking human migration[J]. Clin Microbiol Infect. 2009;15(9):829–34.
Camorlinga-Ponce M, Perez-Perez G, Gonzalez-Valencia G, et al. Helicobacter pylori genotyping from American indigenous groups shows novel Amerindian vacA and CagA alleles and Asian, African and European admixture[J]. PLoS ONE. 2011;6(11):e27212.,50-e27253.
Higashi H, Tsutsumi R, Muto S, et al. SHP-2 tyrosine phosphatase as an intracellular target of Helicobacter pylori CagA protein[J]. Science. 2002;295(5555):683–6.
Amieva M, Peek RM. Pathobiology of helicobacter pyliori–induced gastric cancer[J]. Gastroenterology. 2016;150(1):64–78.
Lind J, Backert S, Hoffmann R, et al. Systematic analysis of phosphotyrosine antibodies recognizing single phosphorylated EPIYA-motifs in CagA of East Asian-type Helicobacter pylori strains[J]. BMC Microbiol. 2016;16(1):201–17.
Hatakeyama M. Helicobacter pylori CagA and gastric cancer: a paradigm for hit-and-run carcinogenesis[J]. Cell Host Microbe. 2014;15(3):306–16.
Roujeinikova A. Phospholipid binding residues of eukaryotic membrane-remodelling F-BAR domain proteins are conserved in Helicobacter pylori CagA[J]. BMC Res Notes. 2014;7(1):525.
Hoseini Z, Sepahvand F, Rashidi B, et al. NLRP3 inflammasome: Its regulation and involvement in atherosclerosis[J]. J Cell Physiol. 2018;233(3):2116.
Abderrazak A, Syrovets T, Couchie D, et al. NLRP3 inflammasome: from a danger signal sensor to a regulatory node of oxidative stress and inflammatory diseases[J]. Redox Biol. 2015;4:296–307.
Viala J, Chaput C, Boneca IG, et al. Nod1 responds to peptidoglycan delivered by the Helicobacter pylori cag pathogenicity island[J]. Nat Immunol. 2004;5(11):1166.
Kim DJ, Park JH, Franchi L, Backert S, Núñez G. The Cag pathogenicity island and interaction between TLR2/NOD2 and NLRP3 regulate IL-1β production in Helicobacter pylori infected dendritic cells. Eur J Immunol. 2013;43(10):2650–8. https://doi.org/10.1002/eji.201243281.
Paik S, Kim JK, Silwal P, Sasakawa C, Jo EK. An update on the regulatory mechanisms of NLRP3 inflammasome activation. Cell Mol Immunol. 2021;18(5):1141–60.
Man SM, Karki R, Kanneganti TD. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases[J]. Immunol Rev. 2017;277(1):61–75.
Abais JM, Xia M, Zhang Y, et al. Redox regulation of NLRP3 inflammasomes: ROS as trigger or effector? [J]. Antioxid Redox Signal. 2015;22(13):1111–29.
Xiang LI, Yue-Ping HE, Sheng LIU, et al. Helicobacter pylori induces cytokines IL-1β and IL-18 production in human monocytic cell line through activation of NLRP3 inflammasome via ROS signaling pathway[J]. Chin J Immunol. 2015;3:308–13.
Lee YS, Lee DY, Yu DY, Kim S, Lee C. Helicobacter pylori induces cell migration and invasion through casein kinase 2 in gastric epithelial cells. Helicobacter. 2014;19:465–75.
Acknowledgements
The authors are grateful to the editor, the associate editor, and the reviewer.
Funding
The work was supported by National Natural Science Foundation of China (31660031, 31760328, 31960028), Project of Science and Technology of Guiyang (ZhuKeHe[2017]30-4), The Key Project of Science and Technology of Guizhou Province(QianKeHe JC [2020]1Z010), and the Central Government Guides Local Science and Technology Development Projects of Guizhou (Grant no. [2019] 4008).
Author information
Authors and Affiliations
Contributions
YX conceived and designed the experiments, XYZ, YZ, YL, LC and DC performed the experiments and analyzed the data. QW, LYB and JJZ gave intellectual advice and revised the manuscript. YX and XYZ wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The project was approved by the Ethic Committee of The Guizhou Medical University.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, X., Li, C., Chen, D. et al. H. pylori CagA activates the NLRP3 inflammasome to promote gastric cancer cell migration and invasion. Inflamm. Res. 71, 141–155 (2022). https://doi.org/10.1007/s00011-021-01522-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-021-01522-6