Abstract
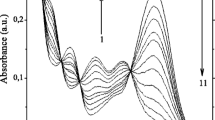
Two newly synthesized cryptands act as sensitive Na+- and K+-selective indicators for cation concentrations above 20 μM. The fluorescence properties change markedly upon cation binding. In addition, the free ligands exhibit a pronounced sensitivity to pH, which is considerably lower for the cation complexes. Time resolved fluorescence is characterized by a decay time of about 5 ns that is attributed to the diprotonated protolytic state of the uncomplexed ligands. Semiempirical calculations show the systematic influence of the nitrogen lone pairs or the N−H bond on the stability of the system. The cause of the strong fluorescence intensity increase observed upon protonation of the fluorescent cryptands may be attributed to an increase in the S1−T x energy gap as a consequence of bridgehead nitrogen protonation.
Similar content being viewed by others
References
F. Kastenholz (1993) Dissertation, Max-Planck-Institute of Biophysics and University of Cologne, Cologne.
J. M. Lehn (1973) in J. D. Dunitz et al. (Eds.),Structure and Bonding, Vol. 16, Verlag Chemie, Weinheim, pp. 1–69.
R. H. Abu-Eittah and B. A. H. El-Tawil (1985)Can. J. Chem. 63, 1173–1179.
A. P. de Silva, H. Q. N. Gunaratne, and C. P. McCoy (1993)Nature 364, 42–44.
D. M. Ferguson and D. J. Raber (1989)J. Am. Chem. Soc. 111, 4371–4378.
M. J. S. Dewar, E. G. Zoebisch, E. F. Healy, and J. J. P. Stewart (1985)J. Am. Chem. Soc. 107, 3902–3909.
Author information
Authors and Affiliations
Additional information
This is a peer-reviewed conference proceeding article from the Third Conference on Methods and Applications of Fluorescence Spectroscopy, Prague, Czech Republic, October 18–21, 1993.
Rights and permissions
About this article
Cite this article
Kastenholz, F., Grell, E., Bats, J.W. et al. Fluorescence studies and semiempirical calculations on alkali ion indicators. J Fluoresc 4, 243–246 (1994). https://doi.org/10.1007/BF01878457
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01878457