Abstract
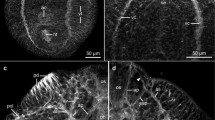
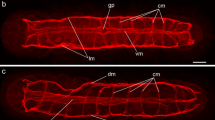
Immunofluorescent labeling and neural tracing techniques were employed in conjunction with confocal scanning laser microscopy to study the intact neuroanatomy of the central nervous system of the trypanorhynch tapewormGrillotia erinaceus Immunocytochemical labeling for the general nerve fibre marker PGP 9.5 showed a pattern of extensive labeling that paralleled findings obtained with the neural tracer DiI. In constrast, immunocytochemical labeling for 5-hydroxytryptamine (5-HT) was localised to cell bodies lying on the periphery of the ganglion, with fine immunoreactive fibres radiating out towards the bothridia. Following the retrograde transport of the fluorescent molecule DiI through axotomised nerve cords, it successfully labeled both the cerebral ganglion and associated nerve fibres within the scolex. The cerebral ganglion was shown to give rise to posterior nerve cords, an array of radial fibres that pass out to the bothridia, and to contain a centrally disposed group of cell bodies thought to be involved in efferent functions.
Similar content being viewed by others
References
Aschoff A, Hollander H (1982) Fluorescent compounds as retrograde tracers compared with horseradish peroxidase (HRP). Visual system of the albino rat. J Neurosci Methods 6:179–197
Coons AH, Leduc EH, Connolly JM (1955) Studies on antibody production. I. A method for the histochemical demonstration of specific antibody and its application to a study of the hyperimmune rabbit. J Exp Med 102:49–60
Fairweather I, Macartney GA, Johnston CF, Halton DW, Buchanan KD (1988) Immunocytochemical demonstration of 5-hydroxytryptamine (serotonin) and vertebrate neuropeptides in the nervous system of excysted cysticerci larvae of the rat tapewormHymenolepis diminuta (Cestoda: Cyclophyllidea). Parasitol Res 74:371–379
Godement P, Vanselow J, Thanos S, Bonhoeffer F (1987) A study in developing visual systems with a new method of staining neurones and their processes in fixed tissue. Development 101:697–713
Gustafsson MKS, Wikgren MC, Karhi TJ, Schot LPC (1985) Immunocytochemical demonstration of neuropeptides and serotonin in the tapewormDiphyllobothrium dendriticum. Cell Tissue Res 240:255–260
Honig MG, Hume RI (1986) Fluorescent carbocyanine dyes allow living neurones of identified origin to be studied in long-term cultures. J Cell Biol 103:171–187
Honig MG, Hume RI (1989) Dil and DiO: versatile fluorescent dyes for neuronal labeling and pathway tracing. TINS 12:333–341
Hrckova G, Halton DW, Maule AG, Shaw C, Johnston CF (1994) 5-Hydroxytryptamine (serotonin)-immunoreactivity in the nervous system ofMesocestoides corti tetrathyridia (Cestoda, Cyclophyllidea). J Parasitol 80: 144–148
Jackson P, Thomson VM, Thompson RJ (1985) A comparison of the evolutionary distribution of the two neuroendocrine markers, neurone-specific enolase and protein gene product 9.5. J Neurochem 45:185–190
Johnston CF, Shaw C, Halton DW, Fairweather I (1990) Confocal scanning laser microscopy and helminth neuroanatomy. Parasitol Today 6:305–308
Marder E, Calabrese RL, Nusbaum B, Trimmer B (1987) Distribution and partial characterisation of FMRFamide-like peptide in the somatogastric nervous system of the rockcrab,Cancer borealis, and the spiny lobster,Panulirus interruptus. J Comp Neurol 259:150–163
McKerr G (1985) The fine structure and physiology of a trypanorhynch tapeworm. PhD Thesis, Queen's University of Belfast, North Ireland
Sukhdeo MVK, Hsu SC, Thompson CS, Mettrick DF (1984)Hymenolepis diminuta: behavioural effects of 5-hydroxytryptamine, acetylcholine, histamine and somatostatin. J Parasitol 70:682–688
Swanson LW (1983) The use of retrogradely transported fluorescent markers in neuroanatomy. Curr Methods Cell Neurobiol 1:219–240
Taylor DCM, Pierau FR-K, Schmid H (1983) The use of fluorescent tracers in the peripheral sensory nervous system. J Neurosci Methods 8:211–224
Ward SM (1986) Observations on the structure, ultrastructure and physiology of the trypanorhynch tapewormGrillotia erinaceus. Ph D Thesis, University of Ulster, North Ireland
Ward SM, Allen JM, McKerr G (1986) Neuromuscular physiology ofGrillotia erinaceus metacestodes (Cestoda: Trypanorhyncha) in vitro. Parasitology 93:121–132
Webb RA, Mizukawa K (1985) Serotonin-like immunoreactivity in the cestodeHymenolepis diminuta. J Comp Neurol 234:431–440
Wikgren M, Reuter M, Gustafsson MKS (1986) Neuropeptides in free-living and parasitic flatworms (Platyhelminthes). An immunocytochemical study. Hydrobiology 132:93–99
Yoshikami D, Okun LM (1984) Staining of living presynaptic nerve terminals with selective fluorescent dyes. Nature 310:53–56
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crangle, K.D., McKerr, G., Allen, J.M. et al. The central nervous system ofGrillotia erinaceus (Cestoda: Trypanorhyncha) as revealed by immunocytochemistry and neural tracing. Parasitol Res 81, 152–157 (1995). https://doi.org/10.1007/BF00931622
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00931622