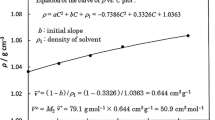
Abstract
In general, the vapor-pressure change due to the addition of a solute to a solvent mixture does not follow Raoult's law. We have demonstrated thermodynamically that if one adds to a binary solvent mixture solute and solvents in such a way that the vapor-phase composition remains constant, then the decrease of total pressure follows Raoult's law; a supplementary term which vanishes for very dilute solute concentration is introduced as a consequence of the nonideality of the ternary solution. Precise vapor-pressure measurements of dilute solutions of electrolytes and nonelectrolytes in a 40.000 wt. % water-tetrahydrofuran mixture are used in order to illustrate the applicability of Raoult's law under the above conditions. These may be regarded as a particular case of what has been called endostatic conditions, i.e., addition of a solute under constant solvent activity ratio.
Similar content being viewed by others
References
W. L. Miller,J. Phys. Chem. 1, 633 (1897); A. I. Johnson and W. F. Furter,Can. J. Chem. Eng., 78 (1960).
R. Wright,J. Chem. Soc., 124, 2068 (1924).
E. Grünwald and A. L. Barcarella,J. Am. Chem. Soc. 80, 3840 (1958); E. Grünwald, G. Baugham, and G. Kohnstam,J. Am. Chem. Soc. 82, 5801 (1960).
E. Grünwald and A. Effio,J. Am. Chem. Soc. 96, 423 (1974).
C. Treiner and P. Finas,J. Chim. Phys. 71, 76 (1974).
C. Treiner,J. Chim. Phys. 70, 1183 (1973).
C. Treiner and P. Tzias,J. Solution Chem. 4, 471 (1975).
J. L. Hawes and R. L. Kay,J. Phys. Chem. 69, 2420 (1965).
C. Treiner, J. F. Bocquet, and M. Chemla,J. Chim. Phys. 70, 68 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tzias, P., Treiner, C. & Chemla, M. Applicability of Raoult's law in nonideal mixed solvents. J Solution Chem 6, 393–402 (1977). https://doi.org/10.1007/BF00645512
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00645512