Abstract
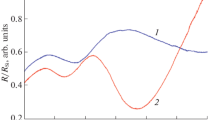
Vanadium dioxide thin films were prepared by an atmospheric-pressure chemical vapour deposition method. The raw material was vanadium(III) acetylacetonate. Polycrystalline thin films were obtained at a reaction temperature of 500°C. Slow post-deposition cooling of the deposits on a substrate of fused quartz or sapphire single crystal yields vanadium dioxide films which are not mixed with other phases, i.e. V3O7 or V4O9. Optical and electrical switching behaviours strongly depend on film thickness. At a film thickness of about 300 nm the transition temperature showed a minimum value of 44 °C.
Similar content being viewed by others
References
C. B. Greenberg, Thin Solid Films 110 (1983) 73.
Y. Takahashi, M. Kanamori, H. Hashimoto, Y. Moritani and Y. Masuda, J. Mater. Sci. 24 (1989) 192.
D. P. Partlow, S. R. Gurkovich, K. C. Radford and L. J. Denes, J. Appl. Phys. 70 (1991) 443.
E. E. Chain, J. Vac. Sci. Technol. A4 (1986) 432.
J. F. De Natale, P. J. Hood and A. B. Haker, J. Appl. Phys. 66 (1989) 5844.
F. Cardillo Case, J. Vac. Sci. Technol. A2 (1984) 1509.
J. B. MacChesney, J. F. Potter and H. J. Guggenheim, J. Electrochem. Soc. 115 (1968) 52.
L. A. Ryabova, I. A. Serbinov and A. S. Darevsky, ibid. 119 (1972) 427.
R. L. Remke, R. M. Walser and R. W. Bené, Thin Solid Films 61 (1979) 73.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maruyama, T., Ikuta, Y. Vanadium dioxide thin films prepared by chemical vapour deposition from vanadium(III) acetylacetonate. JOURNAL OF MATERIALS SCIENCE 28, 5073–5078 (1993). https://doi.org/10.1007/BF00361182
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00361182