Summary
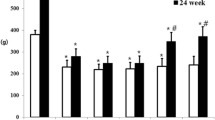
The effects of orally-administered myo-inositol have been compared with those of an aldose reductase inhibitor on acute neurological defects in experimentally diabetic rats. Three groups of streptozotocin-treated diabetic rats (50mg/kg, IP) together with three groups of age-matched controls (saline, IP) were compared. One pair of groups (control and diabetic) were untreated for 3 weeks, another pair of groups received daily oral myo-inositol (667 mg/kg) and the third pair received an aldose reductase inhibitor (ICI 105552; 50 mg·kg-1·day-1, orally). The untreated diabetic group showed statistically significant deficits in accumulation, proximal to 24 h sciatic nerve constrictions, of choline acetyltransferase activity by comparison with untreated controls (2.8±0.4 versus 5.1±0.4nmol acetylcholine ·h-1·nerve-1; p<0.001). The untreated diabetic rats also showed a fall in motor nerve conduction velocity of 6.2±0.7 m/s which was statistically significant (p<0.001). Treatment of the diabetic group with myo-inositol prevented the development of both defects of axonal transport and conduction velocity; both measurements were similar to those of the myo-inositol treated control rats. Likewise the diabetic rats which received aldose reductase inhibitor showed prevention of both defects. Nerves from untreated diabetic rats showed marked sorbitol accumulation and a statistically significant reduction in myo inositol content by comparison with the untreated controls (sorbitol, 1.56 ±0.22 versus 0.8±0.01 and myo-inositol, 1.47±0.10 versus 2.3±0.10 nmol/mg; p<0.001). Treatment of the diabetic rats with myo-inositol elevated the myo-inositol levels in nerve (3.05±0.22 nmol/mg) without affecting nerve sorbitol levels. Treatment of diabetic rats with aldose reductase inhibitor also normalised nerve myo-inositol (2.75±0.20 nmol/mg) and reduced nerve sorbitol (0.08±0.01 nmol/mg). These findings are consistent with the suggestion that both treatments prevent the development of the two functional defects by normalising the nerve myo-inositol content.
Article PDF
Similar content being viewed by others
References
Brockhuyse RM (1968) Changes in myo-inositol permeability in the lens due to caractous condition. Biochim Biophys Acta 163: 269–272
Caspary WF, Crane RK (1970) Active transport of myo-inositol and its relation to the sugar transport system in hamster small intestine. Biochim Biophys Acta 203: 308–316
Clements RS Jr, Stockard CR (1980) Abnormal sciatic nerve myo-inositol metabolism in the streptozotocin-diabetic rat. Diabetes 29: 227–235
Fagius J, Jameson S (1981) Effects of aldose reductase inhibitor treatment in diabetic polyneuropathy — a clinical and neurophysiological study. J Neurol Neurosurg Psychiatr 44: 991–1001
Fonnum F (1973) A rapid radiochemical method for the determination of choline acetyltransferase. J Neurochem 24: 407–409
Gabbay KH (1973) Role of sorbitol pathway in neuropathy. In: Camerini-Davalos RA, Cole HS (eds) Vascular and neurological changes in early diabetes. Academic Press, New York London, p417–424
Gillon KRW, Hawthorne JN (1983) Transport of myo-inositol into endoneurial preparations of sciatic nerve from normal and streptozotocin-diabetic rats. Biochem J 210: 775–781
Greene DA, de Jesus PV Jr, Winegrad AI (1975) Effects of insulin and dietary myo-inositol on impaired peripheral motor nerve conduction velocity in acute streptozotocin-diabetes. J Clin Invest 55: 1326–1336
Greene DA, Lewis RA, Lattimer SA, Brown MJ (1982) Selective effects of myo-inositol administration on sciatic and tibial motor nerve conduction parameters in the streptozotocin diabetic rat. Diabetes 31: 573–578
Hauser G (1965) Energy- and sodium-dependent uptake of inositol by kidney cortex slices. Biochem Biophys Res Commun 19: 696–701
Judzewitsch RG, Jaspan JB, Polonsky KS, Weinberg CR, Halter JB, Halar E, Pfeifer MA, Vukadinovic C, Bernstein L, Schneider M, Liang K-Y, Gabbay KH, Rubenstein AH, Porte D Jr (1983) Aldose reductase inhibition improves nerve conduction velocity in diabetic patients. New Engl J Med 308: 119–125
Mayer JH, Moriarty RH, Tomlinson DR (1983) Prevention and amelioration of neurological defects in acute experimental diabetes by Sorbinil, an aldose reductase inhibitor. Br J Pharmacol (in press)
Palmano KP, Whiting PH, Hawthorne JN (1977) Free and lipid myo-inositol in tissues from rats with acute and less severe streptozotocin-induced diabetes. Biochem J 167: 229–235
Perísíc M, Cuénod M (1972) Synaptic transmission depressed by colchicine blockade of axoplasmic flow. Science 175: 1140–1142
Schmidt RE, Matschinskly FM, Godfrey DA, Williams AD, McDougal DB (1975) Fast and slow axoplasmic flow in sciatic nerve of diabetic rats. Diabetes 24: 1081–1085
Sharma AK, Thomas PK (1974) Peripheral nerve structure and function in experimental diabetes. J Neurol Sci 23: 1–15
Sidenius P (1982) The axonopathy of diabetic neuropathy. Diabetes 31: 356–363
Simmons DA, Winegrad AI, Martin DB (1982) Significance of tissue myo-inositol concentrations in metabolic regulation in nerve. Science 217: 848–851
Sweeley CC, Bentley R, Makita M, Wells WW (1963) Gas-liquid chromatography of trimethylsilyl derivatives of sugars and related substances. J Am Chem Soc 85: 2497–2507
Tomlinson DR, Holmes PR, Mayer JH (1982) Reversal, by treatment with an aldose reductase inhibitor, of impaired axonal transport and motor nerve conduction velocity in experimental diabetes. Neurosci Lett 31: 189–193
Varma SD, Chakraponi B, Reddy VN (1970) Intraocular transport of myo-inositol. II. Accumulation in the rabbit lens in vitro. Invest Ophthalmol 9: 794–800
Ward JD, Baker RWR, Davis BH (1972) Effect of blood sugar control on the accumulation of sorbitol and fructose in nervous tissue. Diabetes 21: 1173–1178
Winegrad AI, Greene DA (1977) Diabetic polyneuropathy: the importance of insulin deficiency, hyperglycaemia and alterations in myo-inositol metabolism in its pathogenesis. New Engl J Med 295: 1416–1421
Winegrad AI, Simmons DA, Martin DB (1983) Has one diabetic complication been explained? New Engl J Med 308: 152–154
Yue DK, Hanwell MA, Satchell PM, Turtle JR (1982) The effect of aldose reductase inhibition on motor nerve conduction velocity in diabetic rats. Diabetes 31: 789–794
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mayer, J.H., Tomlinson, D.R. Prevention of defects of axonal transport and nerve conduction velocity by oral administration of myo-inositol or an aldose reductase inhibitor in streptozotocin-diabetic rats. Diabetologia 25, 433–438 (1983). https://doi.org/10.1007/BF00282524
Issue Date:
DOI: https://doi.org/10.1007/BF00282524