Abstract
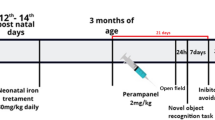
Excessive manganese (Mn) in the brain promotes a variety of abnormal behaviors, including memory deficits, decreased motor skills and psychotic behavior resembling Parkinson’s disease. Hereditary hemochromatosis (HH) is a prevalent genetic iron overload disorder worldwide. Dysfunction in HFE gene is the major cause of HH. Our previous study has demonstrated that olfactory Mn uptake is altered by HFE deficiency, suggesting that loss of HFE function could alter manganese-associated neurotoxicity. To test this hypothesis, Hfe-knockout (Hfe−/−) and wild-type (Hfe+/+) mice were intranasally-instilled with manganese chloride (MnCl2 5 mg/kg) or water daily for 3 weeks and examined for memory function. Olfactory Mn diminished both short-term recognition and spatial memory in Hfe+/+ mice, as examined by novel object recognition task and Barnes maze test, respectively. Interestingly, Hfe−/− mice did not show impaired recognition memory caused by Mn exposure, suggesting a potential protective effect of Hfe deficiency against Mn-induced memory deficits. Since many of the neurotoxic effects of manganese are thought to result from increased oxidative stress, we quantified activities of anti-oxidant enzymes in the prefrontal cortex (PFC). Mn instillation decreased superoxide dismutase 1 (SOD1) activity in Hfe+/+ mice, but not in Hfe−/− mice. In addition, Hfe deficiency up-regulated SOD1 and glutathione peroxidase activities. These results suggest a beneficial role of Hfe deficiency in attenuating Mn-induced oxidative stress in the PFC. Furthermore, Mn exposure reduced nicotinic acetylcholine receptor levels in the PFC, indicating that blunted acetylcholine signaling could contribute to impaired memory associated with intranasal manganese. Together, our model suggests that disrupted cholinergic system in the brain is involved in airborne Mn-induced memory deficits and loss of HFE function could in part prevent memory loss via a potential up-regulation of anti-oxidant enzymes in the PFC.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- GPx:
-
glutathione peroxidase
- Mn :
-
manganese
- nAChR:
-
α7 nicotinic acetylcholine receptor
- PFC:
-
prefrontal cortex
- SOD:
-
superoxide dismutase
References
Cotzias, G.C. (1958) Manganese in health and disease. Physiol. Rev., 38, 503–532.
Zielhuis, R.L., del Castilho, P., Herber, R.F. and Wibowo, A.A. (1978) Levels of lead and other metals in human blood: suggestive relationships, determining factors. Environ. Health Perspect., 25, 103–109.
Bertinchamps, A.J., Miller, S.T. and Cotzias, G.C. (1966) Interdependence of routes excreting manganese. Am. J. Physiol., 211, 217–224.
Brenneman, K.A., Wong, B.A., Buccellato, M.A., Costa, E.R., Gross, E.A. and Dorman, D.C. (2000) Direct olfactory transport of inhaled manganese ((54)MnCl(2)) to the rat brain: toxicokinetic investigations in a unilateral nasal occlusion model. Toxicol. Appl. Pharmacol., 169, 238–248.
Merryweather-Clarke, A.T., Pointon, J.J., Jouanolle, A.M., Rochette, J. and Robson, K.J. (2000) Geography of HFE C282Y and H63D mutations. Genet. Test., 4, 183–198.
Hanson, E.H., Imperatore, G. and Burke, W. (2001) HFE gene and hereditary hemochromatosis: a HuGE review. Human Genome Epidemiology. Am. J. Epidemiol., 154, 193–206.
Kim, J., Li, Y., Buckett, P.D., Böhlke, M., Thompson, K.J., Takahashi, M., Maher, T.J. and Wessling-Resnick, M. (2012) Iron-responsive olfactory uptake of manganese improves motor function deficits associated with iron deficiency. PLoS One, 7, e33533.
Kim, J., Buckett, P.D. and Wessling-Resnick, M. (2013) Absorption of manganese and iron in a mouse model of hemochromatosis. PLoS One, 8, e64944.
Garcia, S.J., Gellein, K., Syversen, T. and Aschner, M. (2007) Iron deficient and manganese supplemented diets alter metals and transporters in the developing rat brain. Toxicol. Sci., 95, 205–214.
Phattanarudee, S., Han, M. and Kim, J. (2014) Effect of olfactory manganese dose on motor coordination in iron-deficient rats. MO. J. Toxicol., 1, 1–7.
Claus Henn, B., Kim, J., Wessling-Resnick, M., Téllez-Rojo, M.M., Jayawardene, I., Ettinger, A.S., Hernández-Avila, M., Schwartz, J., Christiani, D.C., Hu, H. and Wright, R.O. (2011) Associations of iron metabolism genes with blood manganese levels: a population-based study with validation data from animal models. Environ. Health, 10, 97.
Levy, J.E., Montross, L.K., Cohen, D.E., Fleming, M.D. and Andrews, N.C. (1999) The C282Y mutation causing hereditary hemochromatosis does not produce a null allele. Blood, 94, 9–11.
Hou, Y., Zhang, S., Wang, L., Li, J., Qu, G., He, J., Rong, H., Ji, H. and Liu, S. (2012) Estrogen regulates iron homeostasis through governing hepatic hepcidin expression via an estrogen response element. Gene, 511, 398–403.
Kim, J. and Wessling-Resnick, M. (2014) Iron and mechanisms of emotional behavior. J. Nutr. Biochem., 25, 1101–1107.
Dobarro, M., Orejana, L., Aguirre, N. and Ramírez, M.J. (2013) Propranolol restores cognitive deficits and improves amyloid and Tau pathologies in a senescence-accelerated mouse model. Neuropharmacology, 64, 137–144.
Chang, J., Kueon, C. and Kim, J. (2014) Influence of lead on repetitive behavior and dopamine metabolism in a mouse model of iron overload. Toxicol. Res., 30, 267–276.
Han, M. and Kim, J. (2015) Effect of dietary iron loading on recognition memory in growing rats. PLoS One, 10, e0120609.
HaMai, D. and Bondy, S.C. (2004) Oxidative basis of manganese neurotoxicity. Ann. N. Y. Acad. Sci., 1012, 129–141.
Donaldson, J., LaBella, F.S. and Gesser, D. (1981) Enhanced autoxidation of dopamine as a possible basis of manganese neurotoxicity. Neurotoxicology, 2, 53–64.
Guilarte, T.R. and Chen, M.K. (2007) Manganese inhibits NMDA receptor channel function: implications to psychiatric and cognitive effects. Neurotoxicology, 28, 1147–1152.
Blecharz-Klin, K., Piechal, A., Joniec-Maciejak, I., Pyrzanowska, J. and Widy-Tyszkiewicz, E. (2012) Effect of intranasal manganese administration on neurotransmission and spatial learning in rats. Toxicol. Appl. Pharmacol., 265, 1–9.
Zecca, L., Youdim, M.B., Riederer, P., Connor, J.R. and Crichton, R.R. (2004) Iron, brain ageing and neurodegenerative disorders. Nat. Rev. Neurosci., 5, 863–873.
Ali-Rahmani, F., Grigson, P.S., Lee, S., Neely, E., Connor, J.R. and Schengrund, C.L. (2014) H63D mutation in hemochromatosis alters cholesterol metabolism and induces memory impairment. Neurobiol. Aging, 35, e1–12.
Nandar, W. and Connor, J.R. (2011) HFE gene variants affect iron in the brain. J. Nutr., 141, 729S#x2013;739S.
Finkelstein, Y., Milatovic, D. and Aschner, M. (2007) Modulation of cholinergic systems by manganese. Neurotoxicology, 28, 1003–1014.
Chtourou, Y., Fetoui, H., Garoui el, M., Boudawara, T. and Zeghal, N. (2012) Improvement of cerebellum redox states and cholinergic functions contribute to the beneficial effects of silymarin against manganese-induced neurotoxicity. Neurochem. Res., 37, 469–479.
Sarter, M., Gehring, W.J. and Kozak, R. (2006) More attention must be paid: the neurobiology of attentional effort. Brain Res. Rev., 51, 145#x2013;160.
Lorković, H. and Feyrer, A. (1984) Manganese ions inhibit acetylcholine receptor synthesis in cultured mouse soleus muscles. Neurosci. Lett., 51, 331–335.
Kwik-Uribe, C. and Smith, D.R. (2006) Temporal responses in the disruption of iron regulation by manganese. J. Neurosci. Res., 83, 1601–1610.
Feder, J.N., Penny, D.M., Irrinki, A., Lee, V.K., Lebrón, J.A., Watson, N., Tsuchihashi, Z., Sigal, E., Bjorkman, P.J. and Schatzman, R.C. (1998) The hemochromatosis gene product complexes with the transferrin receptor and lowers its affinity for ligand binding. Proc. Natl. Acad. Sci. U.S.A., 95, 1472–1477.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Ye, Q., Kim, J. Loss of Hfe Function Reverses Impaired Recognition Memory Caused by Olfactory Manganese Exposure in Mice. Toxicol Res. 31, 17–23 (2015). https://doi.org/10.5487/TR.2015.31.1.017
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2015.31.1.017