En
Abstract
Aim
The aim of the study was to investigate the role of intratympanic (IT) dexamethasone in the treatment of medically refractory Meniere’s disease (MD) using two different concentrations.
Patients and methods
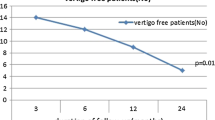
Twenty-four adults with unilateral MD received a single IT injection of dexamethasone at 4 or 10 mg/ml concentration. Partial or no improvement over the next 1 month following injection necessitated a second injection. Before and after injection, all patients underwent detailed history taking, were evaluated with the Dizziness Handicap Inventory scale, and underwent basic audiological evaluation and assessment of cervical-vestibular evoked myogenic potential. The presence or absence of spontaneous, post-head-shaking, and positional nystagmus was assessed using a video-nystagmography system. Twelve patients served as the control group and were followed up subjectively for 1 month.
Results
Both 4 and 10 mg/ml IT dexamethasone improved all subjective symptoms and pure-tone audiometry thresholds. The cervical-vestibular evoked myogenic potential asymmetry ratio dropped after injection. However, the 10 mg/ml concentration was superior to the 5 mg/ml concentration in improving the signs of disease activity, Dizziness Handicap Inventory scores, and the duration of vertigo attacks. No change was reported by the controls.
Conclusion
IT dexamethasone injection might be helpful in controlling MD in some patients. The 10 mg/ml dexamethasone concentration controls vertigo at both the subjective and objective level. Especially if used early in the course of the disease
Article PDF
Similar content being viewed by others
References
Nin F, Hibino H, Doi K, Suzuki T, Hisa Y, Kurachi Y. The endocochlear potential depends on two K+ diffusion potentials and an electrical barrier in the stria vascularis of the inner ear. Proc Natl Acad Sci USA 2008; 105: 1751–1756.
Phillips JS, Westerberg B. Intratympanic steroids for Ménière’s disease or syndrome. Cochrane Database Syst Rev 2011; 7: CD008514.
Santos PM, Hall RA, Snyder JM, Hughes LF, Dobie RA. Diuretic and diet effect on Meniere’s disease evaluated by the 1985 Committee on Hearing and Equilibrium guidelines. Otolaryngol Head Neck Surg 1993; 109(4): 680–689.
Claes J, Van de Heyning PH. Medical treatment of Meniere’s disease: a review of literature. Acta Otolaryngol 1997 pp. 37–42.
Suzuki M, Krug MS, Cheng KC, Yazawa Y, Bernstein J, Yoo TJ. Antibodies against inner-ear proteins in the sera of patients with inner-ear diseases. ORL J Otorhinolaryngol Relat Spec 1997; 59: 10–17.
Itoh A, Sakata E. Treatment of vestibular disorders. Acta Otolaryngol Suppl 1991; 481: 617–623.
Hamid M. Recent advances in medical otology-neurotology. Mediterr J Otol. 2008; 3: 145–153.
Hamid M, Trune D. Issues, indications, and controversies regarding intratympanic steroid perfusion. Curr Opin Otolaryngol Head Neck Surg 2008; 16: 434–440.
Hain T. Pharmacological treatment of vertigo. CNS Drugs 2011; 17: 85–100.
Committee on Hearing and Equilibrium guidelines for the diagnosis and evaluation of therapy in Menière’s disease. American Academy of Otolaryngology-Head and Neck Foundation, Inc. Otolaryngol Head Neck Surg 1995;113:181–185.
El-Gohary M, Kamal N, El-Kahky A, Taha H. Vestibulo-cochlear response to glycerol in Meniere’s patients. Meniere’s disease-update. Proceedings of the 4th International Symposium on Meniere’s disease. 11–14 April; Paris; 2000. pp. 265–274.
Jacobson G, Newman C. Assessing dizziness-related quality of life. Jacobson GP, Shepard NT, editors. Balance assessment and management. Plural Publishing. San Diego, CA.; 2007. 109.
Shepard N, Telian S. Practical management of the balance disorder patient. San Diego: Singular Publishing Group Inc.; 1996.
Colebatch JG. Vestibular evoked potentials. Curr Opin Neurol 2001; 14: 21–26.
Kingma CM, Wit Hp. The effect of changes in perilymphatic K+ on the vestibular evoked potential in the guinea pig. Arch Otorhinolaryngol 2011; 267: 1679–1684.
Boleas-Aguirre MS, Lin FR, Della Santina CC, Minor LB, Carey JP. Longitudinal results with intratympanic dexamethasone in the treatment of Meniere’s disease. Otol Neurotol 2008; 29: 33–38.
Dallan I, Bruschini L, Nacci A, Bignami M, Casani AP. Drop attacks and vertical vertigo after transtympanic gentamicin: diagnosis and management. Acta Otorhinolaryngol Ital 2005; 25: 370–373.
Vassiliou A, Vlastarakos A, Maragoudakis P, Candiloros D, Nikolopoulos T. Meniere’s disease: still a mystery disease with difficult differential diagnosis. Ann Indian Acad Neurol 2011; 14: 12–18.
Garduño-Anaya MA, Couthino De Toledo H, Hinojosa-González R, Pane-Pianese C, Ríos-Castañeda LC. Dexamethasone inner ear perfusion by intratympanic injection in unilateral Ménière’s disease: a two-year prospective, placebo-controlled, double-blind, randomized trial. Otolaryngol Head Neck Surg 2005; 133: 285–294.
Casani A, Piaggi P, Cerchiai N, Seccia V, Sellari Franceschini S, Dallan I. Intratympanic treatment of intractable unilateral Meniere disease: gentamicin or dexamethasone? A randomized controlled trial. Otolaryngol Head Neck Surg 2011; 18: 29–66.
ElBeltagya Y, Gouda A, Mahmouda A, Hazaa N. Intratympanic injection in Meniere’s disease; symptomatic and audiovestibular; comparative, prospective randomized 1-year control study. Egypt J Otolaryngol 2012; 28: 171–183.
Silverstein H, Isaacson JE, Olds MJ, Rowan PT, Rosenberg S. Dexamethasone inner ear perfusion for the treatment of Meniere’s disease: a prospective, randomized, double-blind, crossover trial. Am J Otol 1998; 19: 196–201.
Lu Y, Ren J, Wu W, Yin T, Yang X, Xie D. Intratympanic dexamethasone injections for intractable Meniere’s disease. Lin Chuang Er Bi Yan Hou Ke Za Zhi 2004; 18: 385–387.
Huang Y, She Y, Li W, Cheng C. Intratympanic dexamethasone injections for intractable Meniere’s disease. Lin Chuang Er Bi Yan Hou Ke Za Zhi 2005; 19: 748–749.
Silverstein H, Farrugia M, Van Ess M. Dexamethasone inner ear perfusion for subclinical endolymphatic hydrops. Ear Nose Throat J 2009; 88: 778–785.
Hillman TM, Arriaga MA, Chen DA. Intratympanic steroids: do they acutely improve hearing in cases of cochlear hydrops?. Laryngoscope 2003; 113: 1903–1907.
Hamid M. Clinical advances and issues in auditory and vestibular medicine. Egypt J Ear Nose Throat Allied Sci 2010; 11: 1–6.
Kotimaki J. Meniere’s disease in Finland. An epidemiological and clinical study on occurrence, clinical picture and policy [Dissertation]. Finland: ORL Dept., University of Oulu; 2003.
Salt AN, Sirjani DB, Hartsock JJ, Gill RM, Plontke SK. Marker retention in the cochlea following injections through the round window membrane. Hear Res 2007; 232: 78–86.
Murofushi T, Shimizu K, Takegoshi H, Cheng PW. Diagnostic value of prolonged latencies in the vestibular evoked myogenic potential. Arch Otolaryngol Head Neck Surg 2001; 127: 1069–1072.
Dodson KM, Woodson E, Sismanis A. Intratympanic steroid perfusion for the treatment of Meniere’s disease: a retrospective study. Ear Nose Throat J 2004; 83: 394–398.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflicts of interest
None declared.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maksoud, A.A., Hassan, D.M., Nafie, Y. et al. Intratympanic dexamethasone injection in Meniere’s disease. Egypt J Otolaryngol 31, 128–134 (2015). https://doi.org/10.4103/1012-5574.156098
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.4103/1012-5574.156098