Abstract
Early parenting relies on emotion regulation capabilities, as mothers are responsible for regulating both their own emotional state and that of their infant during a time of new parenting-related neural plasticity and potentially increased stress. Previous research highlights the importance of frontal cortical regions in facilitating effective emotion regulation, but few studies have investigated the neural regulation of emotion among postpartum women. The current study employed a functional neuroimaging (fMRI) approach to explore the association between perceived stress, depressive symptoms, and the neural regulation of emotion in first-time mothers. Among 59 postpartum mothers, higher perceived stress during the postpartum period was associated with less self-reported use of cognitive reappraisal in everyday life, and greater use of emotion suppression. While viewing standardized aversive images during the Emotion Regulation Task (ERT), mothers were instructed to experience their natural emotional state (Maintain) or to decrease the intensity of their negative emotion by using cognitive reappraisal (Reappraise). Whole-brain analysis revealed a two-way interaction of perceived stress x condition in the right dorsolateral prefrontal cortex (DLPFC) at p < .05 cluster-wise corrected, controlling for postpartum months and scanner type. Higher levels of perceived stress were associated with heightened right DLPFC activity while engaging in cognitive reappraisal versus naturally responding to negative stimuli. Higher right DLPFC activity during Reappraise versus Maintain was further associated with elevated parenting stress. Findings suggest that stress and everyday reappraisal use is reflected in mothers’ neural regulation of emotion and may have important implications for their adaptation to parenthood.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For first-time mothers, the postpartum period is a time of rapid transformation. New changes can bring about joy and excitement while also leading to significant levels of stress and heightened risk for postpartum depression. Research demonstrates that stressors during the postpartum period are numerous, including not only stressors pertaining to infant care, but also physical (i.e., fatigue, pain, sexual concerns), intrapersonal (i.e., adjustment to maternal identity, concerns about weight, self-care), and interpersonal stressors (i.e., caring for other family members, work, relationship with partner) (Groer et al., 2002). Although increases in levels of stress to some degree are normative during the postpartum period (Abidin, 1995; Kristensen et al., 2018), excessive stress constitutes a serious threat to maternal, infant, and family wellbeing (Booth et al., 2018; Crnic et al., 1983; Leppert et al., 2018; Razurel et al., 2013; Rodgers, 1998). This is likely by challenging the allostatic load as shown in other work on populations with higher levels of stress (Juster et al., 2010). Researchers have called for greater investigation into the psychological experience of stress as a distinct construct, as maternal stress has important implications for but differs from the experience of depression and anxiety (Rallis et al., 2014). Many researchers also have become interested in how stress reduction can curtail the development of mood disorders and impaired maternal-infant bonding, particularly among disadvantaged communities (Groer et al., 2002; Kudo et al., 2014). Understanding how mothers’ emotion regulatory capacities might buffer against stress is one way to address this question. The current study examined how postpartum perceived stress, depressive symptoms, and emotion regulation strategies are related to mothers’ neural regulation of negative emotion and parenting adaptation.
A putative mechanism underlying the development of maternal stress and depression during the postpartum period is individual differences in emotion regulation. Emotion regulation is the process of monitoring, evaluating, and modulating one’s emotional reactions to accomplish one’s goals (Eisenberg & Spinrad, 2004; Thompson, 1994). Different emotion regulation strategies can be employed to modulate one’s emotion, such as cognitive reappraisal, expressive suppression, and distraction (Gross, 1998; McRae & Gross, 2020). Cognitive reappraisal involves reinterpreting the meaning of an emotional stimulus as less negative; by contrast, expressive suppression involves inhibiting one’s own emotional responses. Cognitive reappraisal and distraction are considered antecedent-focused strategies (i.e., changing one’s emotional response or attention before or as the emotion develops), whereas suppression is a response-focused strategy (i.e., changing one’s emotional response only after the emotion has developed) (Ortner et al., 2016). Cognitive reappraisal often is considered an adaptive emotion regulation strategy, although its effectiveness can depend on the individual’s skill and the context of the stressor (Ford & Troy, 2019). For example, cognitive reappraisal is more effective than distraction for decreasing negative emotions over the long-term (Hermann et al., 2017); however, distraction is very effective at decreasing emotion rapidly in the moment (Denson et al., 2012). Cognitive reappraisal is not without cognitive costs and requires the use of self-control resources (Ortner et al., 2016; Sheppes et al., 2009; Sheppes & Meiran, 2008). Suppression has been shown to deplete physiological and cognitive resources (Friese et al., 2013; Wang et al., 2014), whereas this is less seen with distraction (Ochsner & Gross, 2005; Sheppes et al., 2009). Overall, a large body of research suggests that the habitual use of cognitive reappraisal is associated with healthier outcomes compared with expressive suppression, such as greater experience and expression of positive emotions, fewer depressive symptoms, resilience to stressors, and better interpersonal relationships (Carlson et al., 2012; Cutuli, 2014). These capacities are likely important for caregiving as well.
The capacity of mothers to regulate their emotions facilitates more sensitive caregiving and may have important implications for both maternal and child emotional health (Morris et al., 2017). Early parenting relies heavily on emotion regulation capabilities, as the parent is tasked with regulating both their emotional state and that of their infant (Morelen et al., 2016; Rutherford et al., 2015). Mothers of toddlers who report less everyday use of cognitive reappraisal describe experiencing greater negative emotion when their child misbehaves; this is further associated with mothers’ use of overreactive and harsh disciplinary strategies (Lorber, 2012). Although it can be difficult to determine the causality of findings, research suggests that postpartum distress and mothers’ everyday use of emotion regulation strategies are highly linked. Mothers with higher postpartum depressive symptoms are more likely to use maladaptive emotion regulation strategies, such as self-blame and rumination (Haga et al., 2009). Further, the use of more effective regulation strategies, such as cognitive reappraisal, are associated with lower postpartum depressive symptoms (Haga et al., 2009). Among samples of first-time mothers, higher perceived stress is associated with less use of cognitive reappraisal, higher depressive symptoms, and decreased life satisfaction (Watcharakitippong et al., 2017). Maternal distress and emotion regulation strategy likely influence one another and may be reflected in mothers’ neural regulation of emotion.
We are interested in how postpartum perceived stress and depressive symptoms are reflected in emotion regulation processes and associated brain circuitry, particularly in the prefrontal cortex. Previous work highlights the importance of frontal cortical regions in modulating amygdala activity and facilitating effective emotion regulation (Banks et al., 2007; Ochsner et al., 2002). Cognitive reappraisal is typically associated with increased activity in the ventromedial, dorsolateral, and medial prefrontal cortex (PFC) (Wager et al., 2008). This increased activity often is associated with decreased amygdala reactivity and reduced perceived negative emotion (Wager et al., 2008). Some studies have found modulation of other emotion-related regions, such as the ventral striatum and insula (Ochsner et al., 2012). Individuals who use cognitive reappraisal more frequently exhibit increased dorsolateral PFC and dorsal anterior cingulate cortex (ACC) activation, along with reduced activation in limbic regions, such as the amygdala and hippocampus, while viewing aversive images (Cutuli, 2014; Drabant et al., 2009; Vanderhasselt et al., 2013). Cognitive reappraisal is a multistep and effortful process, and brain regions involved are thought to support different functions. Individuals must maintain their reappraisal goal in working memory and direct their attention to relevant stimulus features (e.g., dorsolateral and posterior PFC, inferior parietal regions), select appropriate information from semantic memory to support reappraisal (e.g., ventrolateral PFC), and engage in performance monitoring to assess success at modifying their emotional response (e.g., dorsal region of ACC) (Ochsner et al., 2012). This process is likely disrupted in the context of stress and adversity.
Individuals with mood disorders, as well as those living under stressful conditions, such as poverty, generally show reduced prefrontal cortex recruitment while attempting to regulate negative emotion in an effortful way (Erk et al., 2010; Kim et al., 2013; Liberzon et al., 2015; Picó-Pérez et al., 2017; Zilverstand et al., 2016), suggesting difficulties in top-down control of negative emotion. However, contradictory findings suggest that distressed individuals exhibit hyperactivation in some areas, including greater, but counterproductive or compensatory recruitment of cortical regions (Johnstone et al., 2007), as well as greater activation in regions important for inhibitory control, such as the superior parietal lobule and supramarginal gyri (Picó-Pérez et al., 2017).
Some studies have investigated how distressed mothers’ brains respond when simply viewing negative noninfant-related stimuli, although not within the context of instructed reappraisal. Studies suggest that postpartum women respond differently than nonpregnant controls when viewing negative noninfant-related stimuli, such as enhanced reactivity in the insula and inferior frontal gyrus (IFG) (Gingnell et al., 2015). These differences may place women at increased risk for postpartum mood problems. Increased reactivity in the insula and IFG when viewing angry and fearful adult expressions was associated with subclinical elevations in postpartum anxiety and depressive symptoms (Gingnell et al., 2015). In the amygdala, women who experience postpartum depression and anxiety exhibit reduced activation to negative noninfant cues, such as International Affective Picture System (IAPS) pictures or adult face images (Moses-Kolko et al., 2010; Silverman et al., 2007, 2011). However, to our knowledge, no prior study has examined how postpartum stress and depressive symptoms are reflected in the neural correlates of effortful emotional regulation capacities, such as cognitive reappraisal.
The current study employed a functional neuroimaging (fMRI) approach to test the association between perceived stress and depressive symptoms on the neural regulation of emotion in first-time mothers. We utilized a validated and widely used Emotion Regulation Task (ERT), involving cognitive reappraisal of distressing images (i.e., noninfant stimuli from IAPS) to assess effortful attempts to regulate emotion (Banks et al., 2007; Phan et al., 2005). We were interested in the association between maternal perceived stress and depressive symptoms on neural regions, including the PFC and amygdala activation during emotion regulation. We hypothesized that elevated maternal perceived stress and depressive symptoms would be associated with differing activation levels in prefrontal control regions (e.g., dorsolateral, dorsomedial, and ventrolateral PFC) and in the amygdala during reappraisal compared with maintain conditions. However, based on the literature that suggests both directionalities, we did not have a hypothesis that includes specific directions of the associations in both the PFC and amygdala regions. Furthermore, we predicted that the differences in neural activation associated with perceived stress and depressive symptoms would be associated with lower reappraisal success in the scanner, reduced everyday use of reappraisal strategies, and higher levels of parenting stress.
Materials and methods
Participants
Participants were English-speaking, first-time mothers and their biological infants, at an average of 4.4 months postpartum. Ninety-one percent of participants were recruited within the first 6 months postpartum. Mothers were recruited through flyers and brochures in Denver metro areas, such as midwifery clinics, Women, Infants, and Children (WIC) centers, and Colorado state Prenatal Plus programs to ensure a socioeconomically diverse sample. Exclusion criteria included: a current/historical self-reported psychiatric/neurological illness other than depression or anxiety (to achieve a controlled, but ecologically valid community sample approach), psychoactive drug use (except antidepressants), birthing complications or infant medical illnesses involving more than a one-night stay in the neonatal intensive care unit (NICU), and maternal magnetic metal in the body. The original study focused on recruiting low or middle-income mothers; thus, participants had an income-to-needs ratio (INR) below 8.0 (Kim et al., 2017).
Sixty-five participants completed MRI scanning. Five participants were excluded due to excessive motion (>20% TRs removed; motion cutoff was framewise displacement in any direction exceeding 0.5 mm) and one was excluded because she completed only one run of the emotion regulation task (ERT), resulting in 59 participants in the final analyses. Demographic characteristics are described in Table 1. Of 59 participants who were included in the analysis, 35 participants overlap with Kim et al. (2017), 26 overlap with Kim et al. (2016), 49 overlap with Olsavsky et al. (2019), and 46 overlap with Dufford et al. (2019), all of which used different tasks from the ERT or performed a resting state functional connectivity analysis. Therefore, there is no direct overlap of neuroimaging data among these manuscripts and the current manuscript, although the cohort is similar and has some overlap in terms of participants.
Procedures
The Infant Development, Environment, and Attachment (IDEA) Project study protocol was approved by the university institutional review board. Researchers contacted mothers by phone and assessed eligibility for the study. If eligible, mothers completed two sessions. First, mothers completed a home visit protocol, with interviews and questionnaires. Subsequently, mothers visited the Intermountain Neuroimaging Center at the University of Colorado – Boulder to complete the fMRI portion of the study (median of 23 days following home visit). Mothers received financial compensation for all visits, and childcare and transportation assistance were provided if needed.
Measures
Perceived Stress Scale
Generalized, nonspecific perceptions of stress were assessed by using the 4-item version of Cohen’s Perceived Stress Scale (PSS-4; Cohen & Williamson, 1988; Cohen et al., 1983). This measure evaluated participants’ perceived ability to handle their personal problems and control the important things in their life, and how often they felt things were going well during the past month. An example item includes, “In the last month, how often have you felt difficulties were piling up so high that you could not overcome them?” The PSS-4 has been validated and used in many studies of pregnant and postpartum populations (Karam et al., 2012; Murphey et al., 2017; Park et al., 2009). The PSS had acceptable internal consistency in the current study with a Cronbach’s α coefficient of 0.72, consistent with prior research (Warttig et al., 2013). Mean and standard deviation of total perceived stress scores were similar to those found in a community sample of postpartum women (Murphey et al., 2017).
Beck Depression Inventory
Depressive symptoms were assessed using the Beck Depression Inventory (BDI; Beck et al., 1961). This measure consists of 21 items scored on a 4-point scale. Items assess depressive symptoms in the past week, including low mood, anhedonia, feelings of guilt, irritability, fatigue, and poor appetite. The BDI has been used in other studies of postpartum women (O’Hara et al., 2019; Poyatos-León et al., 2017; Roomruangwong et al., 2017). In the current study, the BDI had good internal consistency with a Cronbach’s α coefficient of 0.81. BDI scores of 10-18 are described as mild to moderate depression, scores of 19-29 are classified as moderate to severe depression, and scores of 30-63 are considered severe depression. Eighteen participants within our sample (30.5%) met criteria for mild to moderate depression, and two participants (3.4%) met criteria for moderate to severe depression.
Parenting Stress Index
Maternal parenting stress was assessed using the Parenting Stress Index – Short Form (PSI-SF; Abidin, 1995). This self-report measure consists of 36 items scored on a 5-point scale. The Total Stress Index is a sum of Parental Distress, Parent-Child Dysfunctional Interaction, and Difficult Child subscales. The Parental Distress subscale assesses stress related to personal factors, such as depression, conflict with partner, and life restrictions due to caregiving. The Parent-Child Dysfunctional Interaction subscale assesses the extent to which the child is meeting the parent’s expectations and the parent-child relationship is stable. The Difficult Child subscale measures parents’ perceptions of the child’s self-regulatory abilities and whether they view the child as disruptive. The PSI-SF demonstrates good internal consistency, retest reliability, and correlates strongly with the full PSI (Abidin, 1995; Reitman et al., 2002; Roggman et al., 1994). The PSI-SF has acceptable to good internal consistency in the current study with a Cronbach’s α coefficient of 0.89 for the Total Stress Index.
Emotion Regulation Questionnaire
Mothers completed a questionnaire assessing emotion regulation style, the Emotion Regulation Questionnaire (ERQ; Gross & John, 2003). This is a 10-item scale assessing the tendency to regulate emotions via cognitive reappraisal and expressive suppression. Mothers responded on a 7-point Likert-type scale ranging from 1 (strongly disagree) to 7 (strongly agree). Six items were averaged to assess reappraisal (e.g., “When I want to feel less negative emotion (such as sadness or anger), I change what I'm thinking about”), and four items were averaged to assess suppression (e.g. “When I am feeling negative emotions, I make sure not to express them”). The ERQ has been used previously in postpartum samples of women (Martini et al., 2017; Rutherford et al., 2015). Consistent with prior validation studies (Gross & John, 2003), the ERQ had good internal consistency with a Cronbach’s α coefficient of 0.88 for Cognitive Reappraisal and 0.78 for Expressive Suppression. Means and standard deviation of reappraisal (M = 5.40, SD = 1.12; range: 2.83–7.00) and expressive suppression (M = 3.08, SD = 1.27; range: 1.00–6.00) were similar to those found in larger samples (Gross & John, 2003; Haga et al., 2009).
fMRI paradigm
We utilized a validated and widely used Emotion Regulation Task (ERT), involving cognitive reappraisal of negative images, to assess effortful attempts to regulate emotion (Banks et al., 2007; Ochsner et al., 2002; Phan et al., 2005). This task has been used in many studies with community and clinical samples (McRae et al., 2008; Perlman et al., 2012; Wager et al., 2008). Negative and neutral images were selected from the International Affective Picture System (IAPS; Lang et al., 1997) based on normative ratings of valence and arousal. Participants were instructed to view neutral images (Look; “Simply look at the images that appear”), experience their natural emotional state while viewing aversive images (Maintain; “Look at the images and be aware of and experience how the image makes you feel”), or to decrease the intensity of their negative affect by using cognitive reappraisal while viewing aversive images (Reappraise; “Use the techniques we’ve told you to try to reevaluate the image in a less negative context”).
Before entering the scanner, participants received instruction in cognitive reappraisal and practiced with a research assistant. The research assistant gave explicit instructions and a number of examples of how to transform a depicted scenario into a less negative or more positive situation (e.g., the people are crying tears of joy because it is a wedding) or to rationalize or objectify the content of the picture (e.g., the people are actors in a movie). To confirm that participants fully understood the reappraisal process, participants were presented with negative images and were instructed to narrate their reappraisal out loud to the research assistant. Feedback and training continued until it was clear the participant understood the process. The fMRI sessions were conducted only when participants fully understood each condition.
The fMRI task utilized a block design, with 20 second blocks, with instructions before each block to “look,” “maintain,” or “reappraise” displayed for 5 seconds. Then, participants viewed 20 seconds of distressing or neutral pictures. Each picture appeared for 5 seconds before being replaced by another picture, without an interstimulus interval. After each block, a rating scale immediately appeared for 5 seconds. Participants were asked to rate the intensity of their negative emotion via a button box (“How negative do you feel?”), with responses ranging from 1 (not at all) to 4 (very much). Interspersed between look, maintain, and reappraise blocks were baseline blocks consisting of a fixation cross and lasting 20 seconds. During this time, participants were instructed to simply relax and wait for the next group of pictures. Total task length was 15.5 minutes and consisted of three runs of 5 min 15 seconds each. Each run contained six blocks, including two look, two maintain, and two reappraise blocks which were pseudo-randomized. The behavioral indicator of the success of regulating negative emotion was calculated by subtracting average ratings following reappraise blocks from average ratings following maintain blocks.
Data acquisition and analysis
fMRI acquisition
Scanning was conducted using two different scanners due to a scanner update; scanners were Siemens Trio and Siemens Prisma. 36 participants were scanned on the Siemens Trio and 23 on the Siemens Prisma. Both were 3.0 T Siemens magnet scanners using a standard 32-channel head coil, acquiring 540 T2*-weighted echo-planar-imaging (EPI) volumes. The parameters of T2* functional sequences were matched across the scanners (TR = 2,300 ms; TE = 27 ms; flip angle = 73; field of view = 192 mm; matrix size, 64 × 64; 36 axial slices; voxels = 3 mm3). In addition to functional data, high-resolution anatomical T1-weighted images were acquired using 3D magnetisation-prepared rapid gradient-echo (MPRAGE) protocol. For the Siemens Trio, high resolution T1-weighted magnetization prepared rapid gradient-echo (MPRAGE) images were acquired with the following parameters: 192 sagittal slices, TR = 2,530 ms, TE = 1.64 ms, flip angle = 7°, FOV = 256 mm2 and voxel size 1 x 1 x 1 mm. For the Siemens Prisma, T1 sequence parameters were 224 sagittal slices, TR = 2,400 ms, TE = 2.07 ms, flip angle = 8°, FOV = 256 mm2, and voxel size 0.8 x 0.8 x 0.8 mm.
Analyses were conducted to examine potential systematic effects of scanner type. First, the noise variance across scanners was examined. The mean average temporal signal-to-noise ratio (TSNR) did not significantly differ by scanner type, t(57) = 1.35, p = .182 (Siemens Trio: M = 224.47, SD = 33.07; Siemens Prisma: M = 211.78, SD = 38.92). Second, the main variable of interest, perceived stress, did not significantly differ by scanner type t(57) = 0.78, p = .938 (Siemens Trio: M = 5.06, SD = 2.89; Siemens Prisma: M = 5.00, SD = 2.59). Third, when only scanner type was included in the whole-brain model, there were no main effects of scanner type and no significant two-way interactions of scanner type x condition. While it is not possible to completely rule out, the results of these analyses do not provide strong evidence for systematic scanner effect. However, to be on the conservative side, scanner type was included as a covariate in the whole-brain analysis.
Covariate selection
Scanner type and postpartum months were included in all fMRI models. Postpartum months was included as a covariate, because the current sample had considerable variability in postpartum age (M = 4.58 months, SD = 1.98, range: 0–10). The following sociodemographic variables were tested and included in post-hoc analyses if they were associated p < .10 with the independent variable (perceived stress, depressive symptoms): maternal age, education, race and ethnicity, handedness, breastfeeding, income-to-need ratio in the past year, maternal psychiatric medication use, maternal self-reported history of depression, anxiety, or other psychiatric disorder, and infant sex. Post-hoc perceived stress models included history of self-reported psychiatric disorder and current psychiatric medication use as covariates as they were associated p < .10 with perceived stress.
Behavioral data
Participants’ ratings of negative affective state were compared in Look, Maintain, and Reappraise conditions using a repeated measures ANOVA. Additionally, associations between perceived stress and depressive symptoms with reappraisal success in the scanner were examined.
Image preprocessing
Preprocessing and statistical analysis were conducted in Analysis of Functional Neuroimages software (AFNI) (Cox, 1996). The first four pre-steady-state volumes (2 dummy TRs and 2 additional TRs) for each run were discarded. Preprocessing steps included slice timing correction, motion correction, affine alignment, normalization, smoothing, and scaling. Slice timing correction temporally interpolated slices such that the resulting data were as close as possible to if the brain image was acquired at a single time point. Participants’ sequential functional volumes were realigned to the last volume of the last run to correct for head movement. Images with motion greater than 0.5 mm in any direction were censored. Participants with excessive motion (>20% of TRs removed) were excluded from the analysis (see Participants section). Within the data included in the analysis, the range of number of volumes censored was 0–80 (M = 13.56 ± 20.14; median = 6; ≤20% of the total volumes). Thus, at least 90 volumes for each condition were included in the present analysis. After motion correction, the functional, echo-planar image (EPI) was aligned with the structural image via coregistration. Each participant’s brain was then normalized to map onto the Talairach template (Talairach & Tournoux, 1988). Lastly, spatial smoothing was applied using 6-mm full width at half maximum blur estimates and intensity scaling was applied to compare data on a standardized unit.
fMRI Single-Subject Models
At the individual level, a general linear model (GLM) was used to analyze BOLD signal changes in response to three task conditions. Linear regression modeling was performed per voxel with the following regressors: three condition regressors, six motion parameter regressors (3 translational, 3 rotational), and a third-order polynomial modeling baseline drift.
Whole-brain models
Group level analyses examined single-subject images for Look, Maintain, and Reappraise conditions, utilizing a 3dLME model in AFNI (Cox, 1996), controlling for relevant covariates and scanner type. A whole brain mask was created based on 90% EPI coverage. Perceived stress and depressive symptoms were modeled separately to avoid collinearity as the continuous between-subject factor, because these two scores were correlated with one another (r = 0.640, p < .001). Within subject factors were task condition: Look vs. Maintain and Maintain vs. Reappraise. We ran two separate models, Look vs. Maintain and Maintain vs. Reappraise, as Look vs. Reappraise was not the contrast of interest. We examined main effects and interaction effects of perceived stress and depressive symptoms in the whole-brain analysis by entering each of these covariates separately within the 3dLME models. Correction was performed for multiple comparisons within the whole brain using the cluster extent threshold of k ≥ 33 with a height threshold of p < .001, which was determined via 3dClustSim with spatial autocorrelation function (ACF) option. To decompose significant interactions, post-hoc analyses were performed using average percent signal change values that were extracted from significant clusters and analyzed further using SPSS (Statistical Package for the Social Sciences version 22, Chicago, IL).
Exploratory regression analyses—emotional coping and parenting stress
We examined exploratory associations between neural activation extracted from the results of the 3dLME whole-brain analysis in suprathreshold clusters (subtraction data of Reappraise minus Maintain), reappraisal success in the scanner, everyday reappraisal and suppression, and parenting stress indices using bivariate Pearson correlations in SPSS.
Results
Sample characteristics
Mothers were 26 years old on average (M = 25.98, SD = 5.52), primarily Hispanic (44.1%) and non-Hispanic White (42.4%), and almost half of the participants had 13 or fewer years of education. The sample was socioeconomically diverse; almost half of families were low-income (47.5% with an income-to-needs ratio ≤2). See Table 1 for detailed participant demographic information. Elevated perceived stress was significantly associated with elevated depressive symptoms (r = 0.640, p < .001). Some data were not approximately normally distributed, so Mann-Whitney U tests were conducted when appropriate. Perceived stress scores (t(56) = −3.462, p = .001) and depressive symptoms (Mann-Whitney U = 217.00, p = .002) were higher among women with a self-reported history of depression, anxiety, posttraumatic stress disorder (PTSD), or obsessive compulsive disorder (OCD). Perceived stress also was higher among mothers currently taking psychiatric medications (Mann-Whitney U = 58.50, p = .039). However, perceived stress and depressive symptoms were not associated with any other demographic variables, including maternal age, education, income-to-needs ratio (INR) in the past year, race, ethnicity, breastfeeding, and postpartum months (ps > .10). Parenting stress was associated with history of self-reported psychiatric disorder (Mann-Whitney U = 256.50, p = .010) but was not significantly associated with psychotropic use (Mann-Whitney U = 132.50, p = .506).
Behavioral data
Participant ratings of negative emotional state differed between the Look, Maintain, and Reappraise conditions (F(2) = 52.91, p < .001). Post-hoc testing using the Bonferroni correction revealed that ratings increased from the Look (M = 1.09 ± 0.15) to Maintain condition (M = 2.94 ± 0.58), increased from the Look (M = 1.09 ± 0.15) to Reappraise condition (M = 2.38 ± 0.55), and decreased from the Maintain (M = 2.94 ± 0.58) to Reappraise condition (M = 2.38 ± 0.55) (all ps < 0.001). Perceived stress and depressive symptoms were not significantly correlated with reappraisal success in the scanner (ps > .10).
Whole-brain analysis
Perceived Stress
Look vs. Maintain
There were no main effects of perceived stress symptoms and no significant two-way interactions of perceived stress x condition (Look, Maintain).
Maintain vs. Reappraise
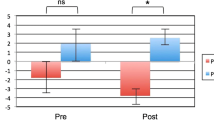
A significant two-way interaction of perceived stress x condition (Maintain, Reappraise) was identified in the right dorsolateral prefrontal cortex (DLPFC; Brodmann Area 9 & 10) at p < .05 corrected (41 voxels, x,y,z = 26,56,26; Fig. 1a). Surface visualization was conducted using Caret version 6.65 (Van Essen et al., 2001). Post-hoc correlation analysis revealed that elevated perceived stress was associated with increased neural activity in this cluster during Reappraise compared to Maintain (Fig. 2a). Perceived stress was positively associated with neural activation during Reappraise activation (r = .287, p = .027). Perceived stress was not associated with Maintain activation (r = −.132, p = .319).
Main effects of perceived stress in the whole-brain analysis revealed a significant cluster in the left caudate nucleus at p < .05, corrected (52 voxels, x,y,z = −13,−4,20; Fig. 1b). Post-hoc correlation analysis revealed that elevated perceived stress was associated with less neural activation in the caudate nucleus across Maintain and Reappraise conditions (Fig. 2b).
Given the significant correlations between variables of interest and maternal self-reported history of psychiatric disorder and current psychiatric medication use, we examined the effect of those potential confounding factors. When self-reported psychiatric history and current medication use were included in the whole-brain model, the association between perceived stress and right DLPFC during Reappraise compared to Maintain remained, p < .05 corrected. The main effect of perceived stress on left caudate activity also remained, p < .05 corrected.
Depression
Look vs. Maintain
There were no main effects of depressive symptoms and no significant two-way interactions of depressive symptoms x condition (Look, Maintain).
Maintain vs. Reappraise
There were no main effects of depressive symptoms and no significant two-way interactions of depressive symptoms x condition (Maintain, Reappraise).
Associations with emotional coping and parenting stress
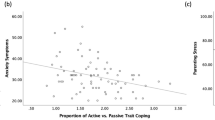
Elevated perceived stress was associated with greater reported use of suppressive expression (r = .283, p = .031), less use of cognitive reappraisal (r = −.290, p = .027), and increased parenting stress (r = .564, p < .001) (Fig. 3). Everyday use of cognitive reappraisal was further associated with reduced parenting stress (r = −.281, p = .032); however, suppression was not significantly associated with parenting stress.
Depressive symptoms were not associated with everyday suppressive expression or cognitive reappraisal (ps > .10) but were associated with greater parenting stress (r = .528, p < .001).
Exploratory analyses revealed that right DLPFC activation extracted from the main analysis (subtraction data of Reappraise minus Maintain) was associated with increased parenting stress (r = .564, p < .001) (Fig. 4). However, caudate activation was not significantly associated with parenting stress (ps > .10). Right DLPFC activation during Reappraise compared with Maintain and left caudate activation during Maintain and Reappraise were not significantly associated with reappraisal success in the scanner or everyday reported use of reappraisal or suppression (all ps > .10).
Discussion
The present study examined whether maternal distress levels were associated with behavioral and neural activation during emotion regulation among a socioeconomically diverse group of first-time mothers. Mothers experiencing higher levels of perceived stress during the postpartum period reported being less likely to use reappraisal strategies and more likely to use suppression. We found that women reporting higher levels of perceived stress also showed enhanced right dorsolateral prefrontal cortex (DLPFC) activity when engaging in cognitive reappraisal (Reappraise condition), compared to when naturally responding to negative stimuli (Maintain condition). No significant differences were found among mothers reporting higher levels of depressive symptoms. Additionally, higher levels of perceived stress were associated with reduced left caudate nucleus activity across Maintain and Reappraise conditions. Exploratory analyses revealed that enhanced right DLPFC activity during Reappraise compared to Maintain conditions was associated with elevated parenting stress. Findings suggest that stress and reappraisal use are reflected in mothers’ neural regulation of emotion and may have implications for their transition to parenthood.
Maternal perceived stress was associated both with self-reported emotion regulation strategy use, as well as brain response during volitional emotion regulation. Mothers with higher perceived stress reported using reappraisal strategies less during their everyday life. This may be because they find these strategies more effortful and must expend greater energy to effectively reappraise negative emotional stimuli. This explanation is consistent with research demonstrating that higher perceived stress is associated with increased avoidant coping and reduced approach coping (Soderstrom et al., 2000; Watcharakitippong et al., 2017). Higher perceived stress also was associated with increased right dorsolateral prefrontal cortex (DLPFC) activation during Reappraise compared with Maintain conditions. The DLPFC is involved in the processing of emotion-related meanings and is activated following presentation of both negative and positive emotional stimuli (Keightley et al., 2003; Lane et al., 1997; León-Carrión et al., 2007; Teasdale et al., 1999). Furthermore, the DLPFC plays a central role in the prefrontal emotion regulation network and supports the cognitive modulation of emotion and strategies, such as reappraisal (Buhle et al., 2014; Morawetz et al., 2016).
Despite significant findings for Reappraise compared to Maintain conditions, perceived stress was not associated with differential brain activation during Maintain versus Look conditions. Maintain (i.e., naturally responding to a negative picture) and Look (i.e., naturally responding to a neutral picture) conditions can be interpreted as tapping into emotional reactivity. Perhaps perceived stress did not have a significant impact on brain activation due to overall dampened stress reactivity during the perinatal period (de Weerth & Buitelaar, 2005).
Surprisingly, depressive symptoms were not significantly associated with use of emotion regulation strategies or brain activation during the emotion regulation task. Nonsignificant findings may be due to the recruitment of a community sample and limited range in depression scores. Although perceived stress and depressive symptoms were highly correlated in our sample, only 3.4% of participants met criteria for moderate to severe depression based on the clinical cutoff for the Beck Depression Inventory. The majority of the sample exhibited minimal depressive symptoms (84.7%, BDI ≤ 13); study findings should be interpreted within the context of this largely healthy population. Future research should investigate whether emotion regulation strategies and brain activation differ among women diagnosed with postpartum depression.
The association between perceived stress and heightened DLPFC activation during reappraisal may indicate greater effort required to down-regulate emotional responses. Although the majority of prior studies describe decreased DLPFC activity during reappraisal among distressed populations (Zilverstand et al., 2016), there are some studies that find enhanced activations. Johnstone et al. (2007) found that depressed individuals showed greater recruitment of the right lateral and ventrolateral PFC during efforts to reappraise negative emotional stimuli. In a right lateral PFC region (BA10), overlapping with the right dorsolateral cluster in the current study, depressed individuals showed increased activation during Reappraise compared with Maintain. By contrast, healthy controls showed less activation during Reappraise compared with Maintain conditions. Reappraisal while viewing violent scenes was also associated with greater right DLPFC activation in individuals with social anxiety compared with healthy controls (Goldin et al., 2009). Hyperactivation of the DLPFC may be a signal of inefficient regulation. The DLPFC also shows higher activation among depressed individuals on cognitive tasks, which helps to facilitate cognitive performance (Fitzgerald et al., 2017; Matsuo et al., 2007).
Greater right DLPFC activation among stressed mothers also may be related to heightened anxiety during reappraisal. Individuals experiencing psychological distress often show enhanced right DLPFC activation when viewing negative stimuli (Anand et al., 2005; Keedwell et al., 2005; Ritchey et al., 2011). In fact, there is a pattern of relative hyperactivity in the right DLPFC and hypoactivity in the left DLPFC among depressed and phobic individuals during emotional activation (Grimm et al., 2008; Liotti et al., 2002; Schienle et al., 2013). Distressed individuals can benefit from treatments, such as repetitive transcranial magnetic stimulation (rTMS) that decrease right DLPFC activation (Bermpohl et al., 2006; Fitzgerald et al., 2003; Gershon et al., 2003; Mantovani et al., 2013); reported outcomes include improvements in emotion regulation (Diefenbach et al., 2016). Among community populations, decreasing right DLPFC activity is associated with improved inhibitory control of emotion (Cho et al., 2010; Ironside et al., 2016), whereas increasing it is associated with negative emotional responding (Kelley et al., 2013). In the current study, mothers experiencing elevated perceived stress may exhibit salience biases and differences in their interpretation of stimuli during reappraisal.
Exploratory analyses showed that neural activation during cognitive reappraisal was linked to concerns surrounding parenting. Specifically, elevated right DLPFC activation was associated with elevated total parenting stress, including worries about child misbehavior, maternal-child bonding, and maternal depressive symptoms and role changes. It is important to note that parenting-specific stress and general perceived stress are distinct, but overlapping constructs (Creasey & Reese, 1996). There is likely some spillover from parenting stress to general stress, and vice versa (Barnett et al., 1992). However, research suggests that parenting-specific stress is a unique and sometimes stronger predictor of maternal psychological distress and child outcomes compared with general life stress (Creasey & Reese, 1996; Quittner et al., 1990). Maternal stress surrounding her new parenting role may have important implications for her health and wellbeing during the postpartum period, as well as the health and wellbeing of her child. Overall, maternal neural response during emotion regulation is associated with indicators of her adaptation to her new parenting role.
Perceived stress also was associated with reduced caudate nucleus activation, both when viewing and reappraising aversive stimuli, which may reflect altered processing of negative emotion. The caudate, part of the striatum, is highly activated by emotionally arousing content (Carretié et al., 2009; Gerdes et al., 2010). It is involved in implicit and reward-based learning (Haruno et al., 2004; Seger & Cincotta, 2005) and may be particularly activated during automatic emotional processing (Bogert et al., 2016). Caudate activation is reduced among distressed individuals; patients with depression and PTSD show decreased activations of the caudate when viewing negative and positive images (Elman et al., 2009; Felmingham et al., 2014; Lee et al., 2008; Ritchey et al., 2011). After receiving cognitive behavioral therapy, depressed patients demonstrated an increase in caudate activation to viewing emotional images (Ritchey et al., 2011). Individuals exposed to a stressor (e.g., cold pressor, negative performance feedback), also show dampened caudate activation (Kumar et al., 2014; Porcelli et al., 2012). Dampened activation of subcortical structures, such as the caudate may reflect altered emotional arousal and dysfunctional prefrontal-subcortical circuitry following stress. Research suggests structural and functional connectivity between the caudate nucleus and DLPFC (Draganski et al., 2008; Leh et al., 2007; Robinson et al., 2012). In the present study, reduced caudate activation among stressed mothers may indicate less implicit emotion processing and regulation. This could contribute to greater effortful and conscious attempts at cognitive reappraisal and heightened right DLPFC activation (Rive et al., 2013).
Study Limitations & Future Directions
This study should be considered in light of several limitations. First, all measures were assessed concurrently, such that it is impossible to infer causality of findings or to examine mediation. General perceived stress and parenting stress were highly correlated in the present sample, and both were related to reduced use of cognitive reappraisal during everyday life. High levels of stress may contribute to the use of more suppression and fewer reappraisal strategies; conversely, more frequent use of suppression and lower utilization of reappraisal strategies may be associated with higher stress levels in mothers. Longitudinal studies can help to disentangle the impact of emotion regulation strategy selection and stress on parenting success and challenges. For example, longitudinal training in reappraisal techniques such as distancing has been shown to reduce participants’ level of perceived stress in their daily lives (Denny & Ochsner, 2014). Parenting interventions also are associated with reductions in parenting stress and altered neural responses to infant cry, namely increases in child versus self-focused activity in social brain areas (Swain et al., 2017).
Second, brain activation, stress, and everyday reappraisal were not associated with reports of reappraisal success in the scanner. This result is consistent with findings that only 16% of studies find significant group differences in self-reported reappraisal success between clinical and control populations while in the scanner (Dillon & Pizzagalli, 2013; Zilverstand et al., 2016). Highly stressed mothers may be able to regulate their emotions effectively when given appropriate instructions, but this process may be more effortful and not as well integrated into their everyday life.
It is important to note that the relationship of right DLPFC during reappraisal and parenting stress was an exploratory analysis. Future studies are needed to test the relationship between parenting stress and the neural regulation of emotion, particularly identifying how parenting stress is associated with observed caregiving behaviors and neural response during parenting-relevant tasks. Although the use of a well-validated task for emotion regulation was a strength of the current study, it is important to acknowledge that we assessed mothers’ neural regulation of emotion in the context of noninfant-related stimuli. Assessing emotion regulation processes more directly related to parenting may yield important findings. Future research should examine mothers’ neural regulation of emotion in response to negative infant stimuli, such as infant cry sounds, sad baby pictures, or challenging parenting situations. Importantly, studies also should assess how maternal emotion regulation capabilities influence observed parenting behaviors, such as sensitivity and hostility (Crandall et al., 2015; Firk et al., 2018; Lorber, 2012; Schultheis et al., 2019). Research is needed to examine whether neural activity during emotion regulation is related to parenting perceptions, as well as parenting behaviors and child outcomes (Firk et al., 2018; Riem & Karreman, 2019).
Lastly, future studies should assess mothers’ prior experiences in therapy and mothers’ brain activity and physiological response while engaging in regulation strategies such as suppression and distraction. The current study did not assess if mothers had prior experiences with therapy. Prior training in cognitive reappraisal could have influenced mothers’ brain response and skill in implementing reappraisal strategies (Troy et al., 2013). It also is important to acknowledge that expressive suppression was assessed via maternal self-report in the current study. Future research should examine maternal brain response while using regulation strategies, such as expressive suppression, acceptance, and distraction (Firk et al., 2018). Self-reports of suppression were associated with elevated perceived stress in the current study. Thus, in a future study, suppression may be associated with heightened amygdala activation during the down-regulation of negative emotion (Abler et al., 2010; Vanderhasselt et al., 2013). Pupillometry data, skin conductance, and facial expressivity also could be used to assess emotional reactivity in conjunction with fMRI scanning in future studies (Mckinnon et al., 2020; Riem & Karreman, 2019). It is currently unknown if some subjects may have averted their eyes when viewing negatively valenced images, which requires future investigation. Although the current study assessed for potential systematic effects of scanner type (which are described in the methods section) and included scanner type as a covariate in the analyses, future studies should seek to replicate these findings using a single scanner.
Study Implications
Although cognitive reappraisal is generally considered an adaptive emotion regulation strategy, there may be other more effective coping strategies for parenting, particularly parenting under stress. Distraction has been shown to be an effective regulation strategy for decreasing mothers’ emotional intensity and bilateral amygdala activation while listening to infant cry sounds (Firk et al., 2018). Acceptance and commitment therapy (ACT) approaches, such as mindfulness and cognitive defusion may be particularly helpful among postpartum samples (Bonacquisti et al., 2017). Teaching mothers mindfulness skills can promote adaptive coping in stressful contexts (e.g., poverty, relationship conflict) and parenting challenges (e.g., nursing, sleep deprivation) to facilitate sensitive and responsive parenting (Duncan & Bardacke, 2010; Gershy et al., 2017). Additionally, individuals report that acceptance and mindfulness strategies are easier to use than cognitive reappraisal, and participants exhibit less depletion of cognitive resources after engaging in these strategies (Keng et al., 2013; Troy et al., 2018). Future research should evaluate how distraction and mindfulness emotion regulation strategies support maternal mental health during the postpartum period, particularly among mothers experiencing high levels of contextual stressors and psychiatric symptoms.
It also is important to note that expressive suppression may be an appropriate emotion regulation strategy for some mothers and in some cultural contexts. Studies find that individuals with Western-European values experience more negative consequences of suppression (e.g., increased negative emotion). By contrast, suppression of negative emotion was associatied with fewer or no adverse effects among individuals endorsing more Asian values, as well as in Mexican American and Chinese American samples (Butler et al., 2007; Su et al., 2015). Research recruiting diverse postpartum samples, including diversity in culture, race and ethnicity, and socioeconomic status, can help us to identify adaptive emotion regulation strategies and brain response and develop appropriate interventions for new mothers.
The postpartum period is a time of elevated maternal stress and increased vulnerability. Although effective maternal emotion regulation is important for sensitive caregiving and the adaptation to parenthood, few studies have investigated how to best support maternal emotion regulation during this stressful life transition. The current study finds that highly stressed first-time mothers exhibit differences in their habitual emotion regulation strategies, favoring suppression to reappraisal strategies. Stressed mothers also demonstrate altered brain activation in the DLPFC during cognitive reappraisal, which is further associated with elevated concerns about parenting. Future research should assess levels of perceived stress among diverse samples of postpartum mothers, including characterizing longitudinal changes between postpartum stress, brain response during volitional emotion regulation, and parenting behaviors. Elevated perceived stress may interfere with mothers’ ability to engage in cognitive reappraisal, especially when stressors are chronic and outside of mothers’ control (Crandall et al., 2015; Kim et al., 2013). Investigation of contextual risk factors, such as economic and social stressors, and multiple emotion regulation strategies, such as acceptance and distraction, are critical to support the most vulnerable mothers and infants. High perceived stress could be used as a risk factor to identify postpartum mothers who would benefit from additional support, such as enhanced training in cognitive reappraisal or alternative emotion regulation strategies. Research is needed to understand the neural mechanisms underlying adaptive emotion regulation during the postpartum period, due to the unique emotional demands during this time and the long-term implications for maternal and infant wellbeing.
References
Abidin, R. R. (1995). Parenting stress index third edition: Professional manual. Psychological Assessment Resources.
Abler, B., Hofer, C., Walter, H., Erk, S., Hoffmann, H., Traue, H. C., & Kessler, H. (2010). Habitual emotion regulation strategies and depressive symptoms in healthy subjects predict fMRI brain activation patterns related to major depression. Psychiatry Research: Neuroimaging, 183(2), 105–113. https://doi.org/10.1016/j.pscychresns.2010.05.010
Anand, A., Li, Y., Wang, Y., Wu, J., Gao, S., Bukhari, L., Mathews, V. P., Kalnin, A., & Lowe, M. J. (2005). Activity and connectivity of brain mood regulating circuit in depression: A functional magnetic resonance study. Biological Psychiatry, 57(10), 1079–1088. https://doi.org/10.1016/j.biopsych.2005.02.021
Banks, S. J., Eddy, K. T., Angstadt, M., Nathan, P. J., & Phan, K. L. (2007). Amygdala–frontal connectivity during emotion regulation. Social Cognitive and Affective Neuroscience, 2(4), 303–312. https://doi.org/10.1093/scan/nsm029
Barnett, R., Marshall, N. L., & Sayer, A. (1992). Positive-spillover effects from job to home: A closer look. Women & Health, 19(2–3), 13–41. https://doi.org/10.1300/J013v19n02_02
Beck, A. T., Ward, C. H., Mendelson, M., Mock, J., & Erbaugh, J. (1961). An inventory for measuring depression. Archives of General Psychiatry, 4(6), 561–571. https://doi.org/10.1001/archpsyc.1961.01710120031004
Bermpohl, F., Fregni, F., Boggio, P. S., Thut, G., Northoff, G., Otachi, P. T. M., Rigonatti, S. P., Marcolin, M. A., & Pascual-Leone, A. (2006). Effect of low-frequency transcranial magnetic stimulation on an affective go/no-go task in patients with major depression: Role of stimulation site and depression severity. Psychiatry Research, 141(1), 1–13. https://doi.org/10.1016/j.psychres.2005.07.018
Bogert, B., Numminen-Kontti, T., Gold, B., Sams, M., Numminen, J., Burunat, I., Lampinen, J., & Brattico, E. (2016). Hidden sources of joy, fear, and sadness: Explicit versus implicit neural processing of musical emotions. Neuropsychologia, 89, 393–402. https://doi.org/10.1016/j.neuropsychologia.2016.07.005
Bonacquisti, A., Cohen, M. J., & Schiller, C. E. (2017). Acceptance and commitment therapy for perinatal mood and anxiety disorders: Development of an inpatient group intervention. Archives of Women’s Mental Health, 20(5), 645–654. https://doi.org/10.1007/s00737-017-0735-8
Booth, A. T., Macdonald, J. A., & Youssef, G. J. (2018). Contextual stress and maternal sensitivity: A meta-analytic review of stress associations with the Maternal Behavior Q-Sort in observational studies. Developmental Review, 48, 145–177. https://doi.org/10.1016/j.dr.2018.02.002
Buhle, J. T., Silvers, J. A., Wager, T. D., Lopez, R., Onyemekwu, C., Kober, H., Weber, J., & Ochsner, K. N. (2014). Cognitive reappraisal of emotion: A meta-analysis of human neuroimaging studies. Cerebral Cortex, 24(11), 2981–2990. https://doi.org/10.1093/cercor/bht154
Butler, E. A., Lee, T. L., & Gross, J. J. (2007). Emotion regulation and culture: Are the social consequences of emotion suppression culture-specific? Emotion, 7(1), 30–48. https://doi.org/10.1037/1528-3542.7.1.30
Carlson, J. M., Dikecligil, G. N., Greenberg, T., & Mujica-Parodi, L. R. (2012). Trait reappraisal is associated with resilience to acute psychological stress. Journal of Research in Personality, 46(5), 609–613. https://doi.org/10.1016/j.jrp.2012.05.003
Carretié, L., Ríos, M., de la Gándara, B. S., Tapia, M., Albert, J., López-Martín, S., & Álvarez-Linera, J. (2009). The striatum beyond reward: Caudate responds intensely to unpleasant pictures. Neuroscience, 164(4), 1615–1622. https://doi.org/10.1016/j.neuroscience.2009.09.031
Cho, S. S., Ko, J. H., Pellecchia, G., Van Eimeren, T., Cilia, R., & Strafella, A. P. (2010). Continuous theta burst stimulation of right dorsolateral prefrontal cortex induces changes in impulsivity level. Brain Stimulation, 3(3), 170–176. https://doi.org/10.1016/j.brs.2009.10.002
Cohen, S., & Williamson, G. (1988). Perceived stress in a probability sample of the United States. In The Social Psychology of Health (pp. 31–68).
Cohen, Sheldon, Kamarck, T., & Mermelstein, R. (1983). A global measure of perceived stress. Journal of Health and Social Behavior, 24(4), 385–396.
Cox, R. W. (1996). AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Computers and Biomedical Research, 29(3), 162–173. https://doi.org/10.1006/cbmr.1996.0014
Crandall, A., Deater-Deckard, K., & Riley, A. W. (2015). Maternal emotion and cognitive control capacities and parenting: A conceptual framework. Developmental Review, 36, 105–126. https://doi.org/10.1016/j.dr.2015.01.004
Creasey, G., & Reese, M. (1996). Mothers’ and fathers’ perceptions of parenting hassles: Associations with psychological symptoms, nonparenting hassles, and child behavior problems. Journal of Applied Developmental Psychology, 17(3), 393–406. https://doi.org/10.1016/S0193-3973(96)90033-7
Crnic, K. A., Greenberg, M. T., Ragozin, A. S., Robinson, N. M., & Basham, R. B. (1983). Effects of stress and social support on mothers and premature and full-term infants. Child Development, 54(1), 209–217. JSTOR. https://doi.org/10.2307/1129878
Cutuli, D. (2014). Cognitive reappraisal and expressive suppression strategies role in the emotion regulation: An overview on their modulatory effects and neural correlates. Frontiers in Systems Neuroscience, 8. https://doi.org/10.3389/fnsys.2014.00175
de Weerth, C., & Buitelaar, J. K. (2005). Physiological stress reactivity in human pregnancy—A review. Neuroscience and Biobehavioral Reviews, 29(2), 295–312. https://doi.org/10.1016/j.neubiorev.2004.10.005
Denny, B. T., & Ochsner, K. N. (2014). Behavioral effects of longitudinal training in cognitive reappraisal. Emotion, 14(2), 425–433. https://doi.org/10.1037/a0035276.
Denson, T. F., Moulds, M. L., & Grisham, J. R. (2012). The Effects of Analytical Rumination, Reappraisal, and Distraction on Anger Experience. Behavior Therapy, 43(2), 355–364. https://doi.org/10.1016/j.beth.2011.08.001
Diefenbach, G. J., Assaf, M., Goethe, J. W., Gueorguieva, R., & Tolin, D. F. (2016). Improvements in emotion regulation following repetitive transcranial magnetic stimulation for generalized anxiety disorder. Journal of Anxiety Disorders, 43, 1–7. https://doi.org/10.1016/j.janxdis.2016.07.002
Dillon, D. G., & Pizzagalli, D. A. (2013). Evidence of successful modulation of brain activation and subjective experience during reappraisal of negative emotion in unmedicated depression. Psychiatry Research: Neuroimaging, 212(2), 99–107. https://doi.org/10.1016/j.pscychresns.2013.01.001
Drabant, E. M., McRae, K., Manuck, S. B., Hariri, A. R., & Gross, J. J. (2009). Individual differences in typical reappraisal use predict amygdala and prefrontal responses. Biological Psychiatry, 65(5), 367–373. https://doi.org/10.1016/j.biopsych.2008.09.007
Draganski, B., Kherif, F., Klöppel, S., Cook, P. A., Alexander, D. C., Parker, G. J. M., Deichmann, R., Ashburner, J., & Frackowiak, R. S. J. (2008). Evidence for segregated and integrative connectivity patterns in the human basal ganglia. Journal of Neuroscience, 28(28), 7143–7152. https://doi.org/10.1523/JNEUROSCI.1486-08.2008
Dufford, A. J., Erhart, A., & Kim, P. (2019). Maternal brain resting-state connectivity in the postpartum period. Journal of Neuroendocrinology, 31(9). https://doi.org/10.1111/jne.12737
Duncan, L. G., & Bardacke, N. (2010). Mindfulness-based childbirth and parenting education: Promoting family mindfulness during the perinatal period. Journal of Child and Family Studies, 19(2), 190–202. https://doi.org/10.1007/s10826-009-9313-7
Eisenberg, N., & Spinrad, T. L. (2004). Emotion-related regulation: Sharpening the definition. Child Development, 75(2), 334–339. https://doi.org/10.1111/j.1467-8624.2004.00674.x
Elman, I., Lowen, S., Frederick, B. B., Chi, W., Becerra, L., & Pitman, R. K. (2009). Functional neuroimaging of reward circuitry responsivity to monetary gains and losses in posttraumatic stress disorder. Biological Psychiatry, 66(12), 1083–1090. https://doi.org/10.1016/j.biopsych.2009.06.006
Erk, S., Mikschl, A., Stier, S., Ciaramidaro, A., Gapp, V., Weber, B., & Walter, H. (2010). Acute and Sustained Effects of Cognitive Emotion Regulation in Major Depression. Journal of Neuroscience, 30(47), 15726–15734. https://doi.org/10.1523/JNEUROSCI.1856-10.2010
Felmingham, K. L., Falconer, E. M., Williams, L., Kemp, A. H., Allen, A., Peduto, A., & Bryant, R. A. (2014). Reduced amygdala and ventral striatal activity to happy faces in PTSD is associated with emotional numbing. PLoS ONE, 9(9). https://doi.org/10.1371/journal.pone.0103653
Firk, C., Dahmen, B., Lehmann, C., Herpertz-Dahlmann, B., & Konrad, K. (2018). Down-regulation of amygdala response to infant crying: A role for distraction in maternal emotion regulation. Emotion, 18(3), 412–423. https://doi.org/10.1037/emo0000373
Fitzgerald, J. M., Phan, K. L., Kennedy, A. E., Shankman, S. A., Langenecker, S. A., & Klumpp, H. (2017). Prefrontal and amygdala engagement during emotional reactivity and regulation in generalized anxiety disorder. Journal of Affective Disorders, 218, 398–406. https://doi.org/10.1016/j.jad.2017.05.013
Fitzgerald, P. B., Brown, T. L., Marston, N. A. U., Daskalakis, Z. J., Castella, A. de, & Kulkarni, J. (2003). Transcranial magnetic stimulation in the treatment of depression: A double-blind, placebo-controlled trial. Archives of General Psychiatry, 60(10), 1002–1008. https://doi.org/10.1001/archpsyc.60.9.1002
Ford, B. Q., & Troy, A. S. (2019). Reappraisal reconsidered: A closer look at the costs of an acclaimed emotion-regulation strategy. Current Directions in Psychological Science, 28(2), 195–203. https://doi.org/10.1177/0963721419827526
Friese, M., Binder, J., Luechinger, R., Boesiger, P., & Rasch, B. (2013). Suppressing emotions impairs subsequent stroop performance and reduces prefrontal brain activation. PLOS ONE, 8(4), e60385. https://doi.org/10.1371/journal.pone.0060385
Gerdes, A. B. M., Wieser, M. J., Muehlberger, A., Weyers, P., Alpers, G. W., Plichta, M. M., Breuer, F., & Pauli, P. (2010). Brain activations to emotional pictures are differentially associated with valence and arousal ratings. Frontiers in Human Neuroscience, 4. https://doi.org/10.3389/fnhum.2010.00175
Gershon, A. A., Dannon, P. N., & Grunhaus, L. (2003). Transcranial magnetic stimulation in the treatment of depression. American Journal of Psychiatry, 160(5), 835–845. https://doi.org/10.1176/appi.ajp.160.5.835
Gershy, N., Meehan, K. B., Omer, H., Papouchis, N., & Schorr Sapir, I. (2017). Randomized clinical trial of mindfulness skills augmentation in parent training. Child & Youth Care Forum, 46(6), 783–803. https://doi.org/10.1007/s10566-017-9411-4
Gingnell, M., Bannbers, E., Moes, H., Engman, J., Sylvén, S., Skalkidou, A., Kask, K., Wikström, J., & Sundström-Poromaa, I. (2015). Emotion reactivity is increased 4-6 weeks postpartum in healthy women: A longitudinal fMRI study. PLOS ONE, 10(6), e0128964. https://doi.org/10.1371/journal.pone.0128964
Goldin, P. R., Manber-Ball, T., Werner, K., Heimberg, R., & Gross, J. J. (2009). Neural mechanisms of cognitive reappraisal of negative self-beliefs in social anxiety disorder. Biological Psychiatry, 66(12), 1091–1099. https://doi.org/10.1016/j.biopsych.2009.07.014
Grimm, S., Beck, J., Schuepbach, D., Hell, D., Boesiger, P., Bermpohl, F., Niehaus, L., Boeker, H., & Northoff, G. (2008). Imbalance between left and right dorsolateral prefrontal cortex in major depression is linked to negative emotional judgment: An fMRI study in severe major depressive disorder. Biological Psychiatry, 63(4), 369–376. https://doi.org/10.1016/j.biopsych.2007.05.033
Groer, M. W., Davis, M. W., & Hemphill, J. (2002). Postpartum stress: Current concepts and the possible protective role of breastfeeding. Journal of Obstetric, Gynecologic, & Neonatal Nursing, 31(4), 411–417. https://doi.org/10.1111/j.1552-6909.2002.tb00063.x
Gross, J. J. (1998). Antecedent- and response-focused emotion regulation: Divergent consequences for experience, expression, and physiology. Journal of Personality and Social Psychology, 74(1), 224–237. https://doi.org/10.1037/0022-3514.74.1.224
Gross, J. J., & John, O. P. (2003). Individual differences in two emotion regulation processes: Implications for affect, relationships, and well-being. Journal of Personality and Social Psychology, 85(2), 348–362. https://doi.org/10.1037/0022-3514.85.2.348
Haga, S. M., Kraft, P., & Corby, E.-K. (2009). Emotion regulation: Antecedents and well-being outcomes of cognitive reappraisal and expressive suppression in cross-cultural samples. Journal of Happiness Studies, 10(3), 271–291. https://doi.org/10.1007/s10902-007-9080-3
Haruno, M., Kuroda, T., Doya, K., Toyama, K., Kimura, M., Samejima, K., Imamizu, H., & Kawato, M. (2004). A neural correlate of reward-based behavioral learning in caudate nucleus: A functional magnetic resonance imaging study of a stochastic decision task. Journal of Neuroscience, 24(7), 1660–1665. https://doi.org/10.1523/JNEUROSCI.3417-03.2004
Hermann, A., Kress, L., & Stark, R. (2017). Neural correlates of immediate and prolonged effects of cognitive reappraisal and distraction on emotional experience. Brain Imaging and Behavior, 11(5), 1227–1237. https://doi.org/10.1007/s11682-016-9603-9
Ironside, M., O’Shea, J., Cowen, P. J., & Harmer, C. J. (2016). Frontal cortex stimulation reduces vigilance to threat: Implications for the treatment of depression and anxiety. Biological Psychiatry, 79(10), 823–830. https://doi.org/10.1016/j.biopsych.2015.06.012
Johnstone, T., Reekum, C. M. van, Urry, H. L., Kalin, N. H., & Davidson, R. J. (2007). Failure to regulate: Counterproductive recruitment of top-down prefrontal-subcortical circuitry in major depression. Journal of Neuroscience, 27(33), 8877–8884. https://doi.org/10.1523/JNEUROSCI.2063-07.2007
Juster, R.-P., McEwen, B. S., & Lupien, S. J. (2010). Allostatic load biomarkers of chronic stress and impact on health and cognition. Neuroscience & Biobehavioral Reviews, 35(1), 2–16. https://doi.org/10.1016/j.neubiorev.2009.10.002
Karam, F., Bérard, A., Sheehy, O., Huneau, M.-C., Briggs, G., Chambers, C., Einarson, A., Johnson, D., Kao, K., Koren, G., Martin, B., Polifka, J. E., Riordan, S. H., Roth, M., Lavigne, S. V., & Wolfe, L. (2012). Reliability and validity of the 4-item perceived stress scale among pregnant women: Results from the OTIS antidepressants study. Research in Nursing & Health, 35(4), 363–375. https://doi.org/10.1002/nur.21482
Keedwell, P. A., Andrew, C., Williams, S. C. R., Brammer, M. J., & Phillips, M. L. (2005). A double dissociation of ventromedial prefrontal cortical responses to sad and happy stimuli in depressed and healthy individuals. Biological Psychiatry, 58(6), 495–503. https://doi.org/10.1016/j.biopsych.2005.04.035
Keightley, M. L., Winocur, G., Graham, S. J., Mayberg, H. S., Hevenor, S. J., & Grady, C. L. (2003). An fMRI study investigating cognitive modulation of brain regions associated with emotional processing of visual stimuli. Neuropsychologia, 41(5), 585–596. https://doi.org/10.1016/S0028-3932(02)00199-9
Kelley, N. J., Hortensius, R., & Harmon-Jones, E. (2013). When anger leads to rumination: Induction of relative right frontal cortical activity with transcranial direct current stimulation increases anger-related rumination. Psychological Science, 24(4), 475–481. https://doi.org/10.1177/0956797612457384
Keng, S.-L., Robins, C. J., Smoski, M. J., Dagenbach, J., & Leary, M. R. (2013). Reappraisal and mindfulness: A comparison of subjective effects and cognitive costs. Behaviour Research and Therapy, 51(12), 899–904. https://doi.org/10.1016/j.brat.2013.10.006
Kim, P., Capistrano, C., & Congleton, C. (2016). Socioeconomic disadvantages and neural sensitivity to infant cry: Role of maternal distress. Social Cognitive and Affective Neuroscience, 11(10), 1597–1607. https://doi.org/10.1093/scan/nsw063
Kim, P., Capistrano, C. G., Erhart, A., Gray-Schiff, R., & Xu, N. (2017). Socioeconomic disadvantage, neural responses to infant emotions, and emotional availability among first-time new mothers. Behavioural Brain Research, 325, 188–196. https://doi.org/10.1016/j.bbr.2017.02.001
Kim, P., Evans, G. W., Angstadt, M., Ho, S. S., Sripada, C. S., Swain, J. E., Liberzon, I., & Phan, K. L. (2013). Effects of childhood poverty and chronic stress on emotion regulatory brain function in adulthood. Proceedings of the National Academy of Sciences, 110(46), 18442–18447. https://doi.org/10.1073/pnas.1308240110
Kristensen, I. H., Simonsen, M., Trillingsgaard, T., Pontoppidan, M., & Kronborg, H. (2018). First-time mothers’ confidence mood and stress in the first months postpartum. A cohort study. Sexual & Reproductive Healthcare, 17, 43–49. https://doi.org/10.1016/j.srhc.2018.06.003
Kudo, N., Shinohara, H., & Kodama, H. (2014). Heart rate variability biofeedback intervention for reduction of psychological stress during the early postpartum period. Applied Psychophysiology and Biofeedback, 39(3), 203–211. https://doi.org/10.1007/s10484-014-9259-4
Kumar, P., Berghorst, L. H., Nickerson, L. D., Dutra, S. J., Goer, F. K., Greve, D. N., & Pizzagalli, D. A. (2014). Differential effects of acute stress on anticipatory and consummatory phases of reward processing. Neuroscience, 266, 1–12. https://doi.org/10.1016/j.neuroscience.2014.01.058
Lane, R. D., Reiman, E. M., Bradley, M. M., Lang, P. J., Ahern, G. L., Davidson, R. J., & Schwartz, G. E. (1997). Neuroanatomical correlates of pleasant and unpleasant emotion. Neuropsychologia, 35(11), 1437–1444. https://doi.org/10.1016/S0028-3932(97)00070-5
Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (1997). International affective picture system (IAPS): Technical manual and affective ratings. NIMH Center for the Study of Emotion and Attention, 1, 39–58.
Lee, B.-T., Seok, J.-H., Lee, B.-C., Cho, S. W., Yoon, B.-J., Lee, K.-U., Chae, J.-H., Choi, I.-G., & Ham, B.-J. (2008). Neural correlates of affective processing in response to sad and angry facial stimuli in patients with major depressive disorder. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 32(3), 778–785. https://doi.org/10.1016/j.pnpbp.2007.12.009
Leh, S. E., Ptito, A., Chakravarty, M. M., & Strafella, A. P. (2007). Fronto-striatal connections in the human brain: A probabilistic diffusion tractography study. Neuroscience Letters, 419(2), 113–118. https://doi.org/10.1016/j.neulet.2007.04.049
León-Carrión, J., Martín-Rodríguez, J. F., Damas-López, J., Pourrezai, K., Izzetoglu, K., Barroso y Martin, J. M., & Domínguez-Morales, M. R. (2007). A lasting post-stimulus activation on dorsolateral prefrontal cortex is produced when processing valence and arousal in visual affective stimuli. Neuroscience Letters, 422(3), 147–152. https://doi.org/10.1016/j.neulet.2007.04.087
Leppert, B., Junge, K. M., Röder, S., Borte, M., Stangl, G. I., Wright, R. J., Hilbert, A., Lehmann, I., & Trump, S. (2018). Early maternal perceived stress and children’s BMI: Longitudinal impact and influencing factors. BMC Public Health, 18(1), 1211. https://doi.org/10.1186/s12889-018-6110-5
Liberzon, I., Ma, S. T., Okada, G., Shaun Ho, S., Swain, J. E., & Evans, G. W. (2015). Childhood poverty and recruitment of adult emotion regulatory neurocircuitry. Social Cognitive and Affective Neuroscience, 10(11), 1596–1606. https://doi.org/10.1093/scan/nsv045
Liotti, M., Mayberg, H. S., McGinnis, S., Brannan, S. L., & Jerabek, P. (2002). Unmasking disease-specific cerebral blood flow abnormalities: Mood challenge in patients With remitted unipolar depression. American Journal of Psychiatry, 159(11), 1830–1840. https://doi.org/10.1176/appi.ajp.159.11.1830
Lorber, M. F. (2012). The role of maternal emotion regulation in overreactive and lax discipline. Journal of Family Psychology, 26(4), 642–647. https://doi.org/10.1037/a0029109
Mantovani, A., Aly, M., Dagan, Y., Allart, A., & Lisanby, S. H. (2013). Randomized sham controlled trial of repetitive transcranial magnetic stimulation to the dorsolateral prefrontal cortex for the treatment of panic disorder with comorbid major depression. Journal of Affective Disorders, 144(1), 153–159. https://doi.org/10.1016/j.jad.2012.05.038
Martini, J., Petzoldt, J., Knappe, S., Garthus-Niegel, S., Asselmann, E., & Wittchen, H.-U. (2017). Infant, maternal, and familial predictors and correlates of regulatory problems in early infancy: The differential role of infant temperament and maternal anxiety and depression. Early Human Development, 115, 23–31. https://doi.org/10.1016/j.earlhumdev.2017.08.005
Matsuo, K., Glahn, D. C., Peluso, M. A. M., Hatch, J. P., Monkul, E. S., Najt, P., Sanches, M., Zamarripa, F., Li, J., Lancaster, J. L., Fox, P. T., Gao, J.-H., & Soares, J. C. (2007). Prefrontal hyperactivation during working memory task in untreated individuals with major depressive disorder. Molecular Psychiatry, 12(2), 158–166. https://doi.org/10.1038/sj.mp.4001894
Mckinnon, A. I., Gray, N. S., & Snowden, R. J. (2020). Enhanced emotional response to both negative and positive images in post-traumatic stress disorder: Evidence from pupillometry. Biological Psychology, 154, 107922. https://doi.org/10.1016/j.biopsycho.2020.107922
McRae, K., & Gross, J. J. (2020). Emotion regulation. Emotion, 20(1), 1. https://doi.org/10.1037/emo0000703
McRae, K., Ochsner, K. N., Mauss, I. B., Gabrieli, J. J. D., & Gross, J. J. (2008). Gender differences in emotion regulation: An fMRI study of cognitive reappraisal. Group Processes & Intergroup Relations, 11(2), 143–162. https://doi.org/10.1177/1368430207088035
Morawetz, C., Bode, S., Baudewig, J., Kirilina, E., & Heekeren, H. R. (2016). Changes in effective connectivity between dorsal and ventral prefrontal regions moderate emotion regulation. Cerebral Cortex, 26(5), 1923–1937. https://doi.org/10.1093/cercor/bhv005
Morelen, D., Shaffer, A., & Suveg, C. (2016). Maternal emotion regulation: Links to emotion parenting and child emotion regulation. https://doi.org/10.1177/0192513X14546720
Morris, A. S., Criss, M. M., Silk, J. S., & Houltberg, B. J. (2017). The impact of parenting on emotion regulation during childhood and adolescence. Child Development Perspectives, 11(4), 233–238. https://doi.org/10.1111/cdep.12238
Moses-Kolko, E. L., Perlman, S. B., Wisner, K. L., James, J., Saul, A. T., & Phillips, M. L. (2010). Abnormally reduced dorsomedial prefrontal cortical activity and effective connectivity with amygdala in response to negative emotional faces in postpartum depression. American Journal of Psychiatry, 167(11), 1373–1380. https://doi.org/10.1176/appi.ajp.2010.09081235
Murphey, C., Carter, P., Price, L. R., Champion, J. D., & Nichols, F. (2017). Psychological distress in healthy low-risk first-time mothers during the postpartum period: An exploratory study. Nursing Research and Practice, 2017, e8415083. https://doi.org/10.1155/2017/8415083
Ochsner, K. N., Bunge, S. A., Gross, J. J., & Gabrieli, J. D. E. (2002). Rethinking feelings: An fMRI study of the cognitive regulation of emotion. Journal of Cognitive Neuroscience, 14(8), 1215–1229. https://doi.org/10.1162/089892902760807212
Ochsner, K. N., & Gross, J. J. (2005). The cognitive control of emotion. Trends in Cognitive Sciences, 9(5), 242–249. https://doi.org/10.1016/j.tics.2005.03.010
Ochsner, K. N., Silvers, J. A., & Buhle, J. T. (2012). Functional imaging studies of emotion regulation: A synthetic review and evolving model of the cognitive control of emotion. Annals of the New York Academy of Sciences, 1251(1), E1–E24. https://doi.org/10.1111/j.1749-6632.2012.06751.x
O’Hara, M. W., Pearlstein, T., Stuart, S., Long, J. D., Mills, J. A., & Zlotnick, C. (2019). A placebo controlled treatment trial of sertraline and interpersonal psychotherapy for postpartum depression. Journal of Affective Disorders, 245, 524–532. https://doi.org/10.1016/j.jad.2018.10.361
Olsavsky, A. K., Stoddard, J., Erhart, A., Tribble, R. C., & Kim, P. (2019). Neural processing of infant and adult face emotion and maternal exposure to childhood maltreatment. Social Cognitive and Affective Neuroscience.
Ortner, C. N. M., Marie, M. S., & Corno, D. (2016). Cognitive costs of reappraisal depend on both emotional stimulus intensity and individual differences in habitual reappraisal. PLOS ONE, 11(12), e0167253. https://doi.org/10.1371/journal.pone.0167253
Park, E. R., Chang, Y., Quinn, V., Regan, S., Cohen, L., Viguera, A., Psaros, C., Ross, K., & Rigotti, N. (2009). The association of depressive, anxiety, and stress symptoms and postpartum relapse to smoking: A longitudinal study. Nicotine & Tobacco Research: Official Journal of the Society for Research on Nicotine and Tobacco, 11(6), 707–714. https://doi.org/10.1093/ntr/ntp053
Perlman, G., Simmons, A. N., Wu, J., Hahn, K. S., Tapert, S. F., Max, J. E., Paulus, M. P., Brown, G. G., Frank, G. K., Campbell-Sills, L., & Yang, T. T. (2012). Amygdala response and functional connectivity during emotion regulation: A study of 14 depressed adolescents. Journal of Affective Disorders, 139(1), 75–84. https://doi.org/10.1016/j.jad.2012.01.044
Phan, K. L., Fitzgerald, D. A., Nathan, P. J., Moore, G. J., Uhde, T. W., & Tancer, M. E. (2005). Neural substrates for voluntary suppression of negative affect: A functional magnetic resonance imaging study. Biological Psychiatry, 57(3), 210–219. https://doi.org/10.1016/j.biopsych.2004.10.030
Picó-Pérez, M., Radua, J., Steward, T., Menchón, J. M., & Soriano-Mas, C. (2017). Emotion regulation in mood and anxiety disorders: A meta-analysis of fMRI cognitive reappraisal studies. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 79, 96–104. https://doi.org/10.1016/j.pnpbp.2017.06.001
Porcelli, A. J., Lewis, A. H., & Delgado, M. R. (2012). Acute stress influences neural circuits of reward processing. Frontiers in Neuroscience, 6. https://doi.org/10.3389/fnins.2012.00157
Poyatos-León, R., García-Hermoso, A., Sanabria-Martínez, G., Álvarez-Bueno, C., Cavero-Redondo, I., & Martínez-Vizcaíno, V. (2017). Effects of exercise-based interventions on postpartum depression: A meta-analysis of randomized controlled trials. Birth, 44(3), 200–208. https://doi.org/10.1111/birt.12294
Quittner, A. L., Glueckauf, R. L., & Jackson, D. N. (1990). Chronic parenting stress: Moderating versus mediating effects of social support. Journal of Personality and Social Psychology, 59(6), 1266–1278. https://doi.org/10.1037/0022-3514.59.6.1266
Rallis, S., Skouteris, H., McCabe, M., & Milgrom, J. (2014). The transition to motherhood: Towards a broader understanding of perinatal distress. Women and Birth, 27(1), 68–71. https://doi.org/10.1016/j.wombi.2013.12.004
Razurel, C., Kaiser, B., Sellenet, C., & Epiney, M. (2013). Relation between perceived stress, social support, and coping strategies and maternal well-being: A review of the literature. Women & Health, 53(1), 74–99. https://doi.org/10.1080/03630242.2012.732681
Reitman, D., Currier, R. O., & Stickle, T. R. (2002). A critical evaluation of the parenting stress index-short form (PSI-SF) in a head start population. Journal of Clinical Child & Adolescent Psychology, 31(3), 384–392. https://doi.org/10.1207/S15374424JCCP3103_10
Riem, M. M. E., & Karreman, A. (2019). Experimental manipulation of emotion regulation changes mothers’ physiological and facial expressive responses to infant crying. Infant Behavior and Development, 55, 22–31. https://doi.org/10.1016/j.infbeh.2019.02.003
Ritchey, M., Dolcos, F., Eddington, K. M., Strauman, T. J., & Cabeza, R. (2011). Neural correlates of emotional processing in depression: Changes with cognitive behavioral therapy and predictors of treatment response. Journal of Psychiatric Research, 45(5), 577–587. https://doi.org/10.1016/j.jpsychires.2010.09.007
Rive, M. M., van Rooijen, G., Veltman, D. J., Phillips, M. L., Schene, A. H., & Ruhé, H. G. (2013). Neural correlates of dysfunctional emotion regulation in major depressive disorder. A systematic review of neuroimaging studies. Neuroscience and Biobehavioral Reviews, 37(10 Pt 2), 2529–2553. https://doi.org/10.1016/j.neubiorev.2013.07.018
Robinson, J. L., Laird, A. R., Glahn, D. C., Blangero, J., Sanghera, M. K., Pessoa, L., Fox, P. M., Uecker, A., Friehs, G., Young, K. A., Griffin, J. L., Lovallo, W. R., & Fox, P. T. (2012). The functional connectivity of the human caudate: An application of meta-analytic connectivity modeling with behavioral filtering. NeuroImage, 60(1), 117–129. https://doi.org/10.1016/j.neuroimage.2011.12.010
Rodgers, A. Y. (1998). Multiple sources of stress and parenting behavior. Children and Youth Services Review, 20(6), 525–546. https://doi.org/10.1016/S0190-7409(98)00022-X
Roggman, L. A., Moe, S. T., Hart, A. D., & Forthun, L. F. (1994). Family leisure and social support: Relations with parenting stress and psychological well-being in head start parents. Early Childhood Research Quarterly, 9(3–4), 463–480. https://doi.org/10.1016/0885-2006(94)90020-5
Roomruangwong, C., Kanchanatawan, B., Sirivichayakul, S., & Maes, M. (2017). High incidence of body image dissatisfaction in pregnancy and the postnatal period: Associations with depression, anxiety, body mass index and weight gain during pregnancy. Sexual & Reproductive Healthcare, 13, 103–109. https://doi.org/10.1016/j.srhc.2017.08.002
Rutherford, H. J. V., Wallace, N. S., Laurent, H. K., & Mayes, L. C. (2015). Emotion regulation in parenthood. Developmental Review, 36, 1–14. https://doi.org/10.1016/j.dr.2014.12.008
Schienle, A., Scharmüller, W., Leutgeb, V., Schäfer, A., & Stark, R. (2013). Sex differences in the functional and structural neuroanatomy of dental phobia. Brain Structure & Function, 218(3), 779–787. https://doi.org/10.1007/s00429-012-0428-z
Schultheis, A. M., Mayes, L. C., & Rutherford, H. J. V. (2019). Associations between emotion regulation and parental reflective functioning. Journal of Child and Family Studies, 28(4), 1094–1104. https://doi.org/10.1007/s10826-018-01326-z
Seger, C. A., & Cincotta, C. M. (2005). The roles of the caudate nucleus in human classification learning. Journal of Neuroscience, 25(11), 2941–2951. https://doi.org/10.1523/JNEUROSCI.3401-04.2005
Sheppes, G., Catran, E., & Meiran, N. (2009). Reappraisal (but not distraction) is going to make you sweat: Physiological evidence for self-control effort. International Journal of Psychophysiology, 71(2), 91–96. https://doi.org/10.1016/j.ijpsycho.2008.06.006
Sheppes, G., & Meiran, N. (2008). Divergent cognitive costs for online forms of reappraisal and distraction. Emotion, 8(6), 870–874. https://doi.org/10.1037/a0013711
Silverman, M. E., Loudon, H., Liu, X., Mauro, C., Leiter, G., & Goldstein, M. A. (2011). The neural processing of negative emotion postpartum: A preliminary study of amygdala function in postpartum depression. Archives of Women’s Mental Health, 14(4), 355–359. https://doi.org/10.1007/s00737-011-0226-2
Silverman, M. E., Loudon, H., Safier, M., Protopopescu, X., Leiter, G., Liu, X., & Goldstein, M. (2007). Neural dysfunction in postpartum depression: An fMRI pilot study. CNS Spectrums, 12(11), 853–862. https://doi.org/10.1017/S1092852900015595
Soderstrom, M., Dolbier, C., Leiferman, J., & Steinhardt, M. (2000). The relationship of hardiness, coping strategies, and perceived stress to symptoms of illness. Journal of Behavioral Medicine, 23(3), 311–328. https://doi.org/10.1023/A:1005514310142
Su, J. C., Lee, R. M., Park, I. J. K., Soto, J. A., Chang, J., Zamboanga, B. L., Kim, S. Y., Ham, L. S., Dezutter, J., Hurley, E. A., Seol, K. O., & Brown, E. (2015). Differential links between expressive suppression and well-being among Chinese and Mexican American college students. Asian American Journal of Psychology, 6(1), 15–24. https://doi.org/10.1037/a0036116
Swain, J. E., Ho, S. S., Rosenblum, K. L., Morelen, D., Dayton, C. J., & Muzik, M. (2017). Parent–child intervention decreases stress and increases maternal brain activity and connectivity during own baby-cry: An exploratory study. Development and Psychopathology, 29(2), 535–553. https://doi.org/10.1017/S0954579417000165
Talairach, J., & Tournoux, P. (1988). Co-planar stereotaxic atlas of the human brain: 3-D proportional system: An approach to cerebral imaging (1st ed.). Thieme.
Teasdale, J. D., Howard, R. J., Cox, S. G., Ha, Y., Brammer, M. J., Williams, S. C. R., & Checkley, S. A. (1999). Functional MRI study of the cognitive generation of affect. American Journal of Psychiatry, 156(2), 209–215. https://doi.org/10.1176/ajp.156.2.209
Thompson, R. A. (1994). Emotion regulation: A theme in search of definition. Monographs of the Society for Research in Child Development, 59(2–3), 25–52. https://doi.org/10.1111/j.1540-5834.1994.tb01276.x
Troy, A. S., Shallcross, A. J., Brunner, A., Friedman, R., & Jones, M. C. (2018). Cognitive reappraisal and acceptance: Effects on emotion, physiology, and perceived cognitive costs. Emotion, 18(1), 58–74. https://doi.org/10.1037/emo0000371
Troy, A. S., Shallcross, A. J., Davis, T. S., & Mauss, I. B. (2013). History of mindfulness-based cognitive therapy is associated with increased cognitive reappraisal ability. Mindfulness, 4(3), 213–222. https://doi.org/10.1007/s12671-012-0114-5
Van Essen, D. C., Drury, H. A., Dickson, J., Harwell, J., Hanlon, D., & Anderson, C. H. (2001). An integrated software suite for surface-based analyses of cerebral cortex. Journal of the American Medical Informatics Association: JAMIA, 8(5), 443–459. https://doi.org/10.1136/jamia.2001.0080443
Vanderhasselt, M.-A., Baeken, C., Van Schuerbeek, P., Luypaert, R., & De Raedt, R. (2013). Inter-individual differences in the habitual use of cognitive reappraisal and expressive suppression are associated with variations in prefrontal cognitive control for emotional information: An event related fMRI study. Biological Psychology, 92(3), 433–439. https://doi.org/10.1016/j.biopsycho.2012.03.005
Wager, T. D., Davidson, M. L., Hughes, B. L., Lindquist, M. A., & Ochsner, K. N. (2008). Prefrontal-subcortical pathways mediating successful emotion regulation. Neuron, 59(6), 1037–1050. https://doi.org/10.1016/j.neuron.2008.09.006
Wang, Y., Yang, L., & Wang, Y. (2014). Suppression (but Not Reappraisal) Impairs Subsequent Error Detection: An ERP Study of Emotion Regulation’s Resource-Depleting Effect. PLOS ONE, 9(4), e96339. https://doi.org/10.1371/journal.pone.0096339
Warttig, S. L., Forshaw, M. J., South, J., & White, A. K. (2013). New, normative, English-sample data for the Short Form Perceived Stress Scale (PSS-4). Journal of Health Psychology, 18(12), 1617–1628. https://doi.org/10.1177/1359105313508346
Watcharakitippong, J., Blauw, J., & Tuicomepee, A. (2017). Relationships among perceived stress, spousal support, emotion regulation, subjective well-being, and marital satisfaction of Thai first-time parents. Scholar: Human Sciences, 9(2).
Zilverstand, A., Parvaz, M. A., & Goldstein, R. Z. (2016). Neuroimaging cognitive reappraisal in clinical populations to define neural targets for enhancing emotion regulation. A systematic review. NeuroImage. https://doi.org/10.1016/j.neuroimage.2016.06.009
Acknowledgments
This work was supported by the National Institute of Child Health and Human Development [R21HD078797; R01 HD090068], the National Institute of Health [R21DA046556]; the Professional Research Opportunity for Faculty (PROF) and Faculty Research Fund (FRF), University of Denver; NARSAD Independent Investigator Grant, and the Victoria S. Levin Award For Early Career Success in Young Children’s Mental Health Research, Society for Research in Child Development (SRCD). Aviva K. Olsavsky was supported by Developmental Psychobiology Research Group Postdoctoral Fellowship T32MH015442 and the National Institutes of Health Loan Repayment Program. For disclosure, Aviva Olsavsky is the recipient of additional non-governmental funding sources which did not support this research project (American Academy of Child and Adolescent Psychiatry Pilot Award and Developmental Psychobiology Endowment Fund small grant award), and Aviva’s husband works for Thermo Fisher Scientific, a biomedical company unrelated to this line of research. The authors declare that they have no conflicts of interest in the research. The authors thank the families that participated in the study and the individuals that supported recruitment. The authors also wish to acknowledge Amy Anderson, Lindsay Blanton, Christian Capistrano, Christina Congleton, Tanisha Crosby-Attipoe, Victoria Everts, Rachel Gray, Claire Jeske, Laura Jeske, and Nanxi Xu for research assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
none
Additional information
Open Practices Statement
The data that support the findings of this study are available on request from the corresponding author and the experiments were not preregistered. The data are not publicly available due to containing information that could compromise research participant consent and privacy. Due to the sensitivity of the human participant data and the potential for identification of de-identified participant, it would be important to make the data available to other researchers under a data-sharing agreement that provides for a commitment to data security approved by the corresponding author’s institution IRB. The preprocessing and analysis of data were conducted using a publicly available software, Analysis of Functional Neuroimages software.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 18.1 kb)
Rights and permissions
About this article
Cite this article
Grande, L., Olsavsky, A., Erhart, A. et al. Postpartum Stress and Neural Regulation of Emotion among First-Time Mothers. Cogn Affect Behav Neurosci 21, 1066–1082 (2021). https://doi.org/10.3758/s13415-021-00914-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13415-021-00914-9