Abstract—
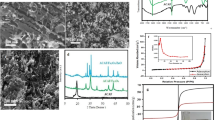
In this study, a novel approach was adopted to determine the drug resistance developed due to the presence of antibiotics in industrial effluents. As a remedy, magnetic carbon nano-composites (MCNs) were prepared from waste biomass and characterized by a surface area analyzer, by SEM, EDX, XRD, FTIR, and TG/DTA. Langmuir, Freundlich and Temkin isotherms were used for the determination of adsorption parameters. The Langmuir adsorption isotherm was found to fit better the data of the equilibrium adsorption of enrofloxacin (ENR) on MCN (R2 = 0.99). The effect of pH on the adsorption process was evaluated and a decline in percentage of adsorption was noted at both highly acidic and alkaline pH. The maximum percentage of adsorption was observed at pH 6–8. The effect of ionic strength and humic acid was also tested for the removal of ENR by MCNs. Various kinetic models were used to analyze the kinetics data. The best fit was obtained with a pseudo 2nd order kinetic equation. The thermodynamic parameters were also determined: ΔS° was positive (80 J mol–1K –1) while ΔH° (23.57 kJ mol–1) and ΔG° were negative with numerical values of –0.27, –1.47, and –3.07 kJ mol–1 corresponding to temperatures 25, 40, and 60°C, respectively. MCNs were regenerated with 3% NaOH solution, methanol, and distilled water.




Similar content being viewed by others
REFERENCES
Golet, E.M., Alder, A.C., and Giger, W., Environ. Sci. Technol., 2002, vol. 36, pp. 3645–3651.
Michael, I., Rizzo, L., McArdell, C., Manaia, C., et al., Water Res., 2013, vol. 47, pp. 957–995.
Nieto, M., Alovero, F., Manzo, R., and Mazzieri, M.R., Eur. J. Med. Chem., 2005, vol. 40, pp. 361–369.
Sayah, R.S., Kaneene, J.B., Johnson, Y., and Miller, R., Appl. Environ. Microbiol., 2005, vol. 71, pp. 1394–1404.
Fent, K., Weston, A.A., and Caminada, D., Aquat. Toxicol., 2006, vol. 76, pp. 122–159.
Martinez, M., McDermott, P., and Walker, R., Vet. J., 2006, vol. 172, pp. 10–28.
Seifrtova, M., Pena, A., Lino, C., and Solich, P., Anal. Bioanal. Chem., 2008, vol. 391, pp. 799–805.
Focazio, M.J., Kolpin, D.W., Barnes, K.K., Furlong, E.T., et al., Sci. Total Environ., 2008, vol. 402, pp. 201–216.
Wang, Q.J., Mo, C.H., Li, Y.W., Gao, P., et al., Environ. Pollut., 2010, vol. 158, pp. 2350–2358.
Yan, W., Hu, S., and Jing, C., J. Colloid Interface Sci., 2012, vol. 372, pp. 141–147.
Zhou, C., Assem, M., Tay, J.C., Watkins, P.B., et al., J. Clin. Invest., 2006, vol. 116, pp. 1703–1712.
Guinea, E., Brillas, E., Centellas, F., Cañizares, P., et al., Water Res., 2009, vol. 43, pp. 2131–2138.
Rivagli, E., Pastorello, A., Sturini, M., Maraschi, F., et al., J. Environ. Chem. Eng., 2014, vol. 2, pp. 738–744.
Tong, D.S., Zhou, C.H.C., Lu, Y., Yu, H., et al., Appl. Clay Sci., 2010, vol. 50, pp. 427–431.
Li, H., Zhang, D., Han, X., and Xing, B., Chemosphere, 2014, vol. 95, pp. 150–155.
Zahoor, M. and Mahramanlioglu, M., Chem. Biochem. Eng. Q., 2011, vol. 25, pp. 55–63.
Zahoor, M., Desalin. Water Treat., 2014, vol. 52, pp. 3104–3114.
Muhammad, M.U.R.K., Zahoor, M., Bakhtiar, M., Farhat, A.K., et al., J. Nanomater., 2017, vol. 2017, pp. 1–10.
Zahoor, M. and Farhat, A.K., Desalin. Water Treat., 2016, vol. 57, pp. 11893–11903.
Zhu, Y., Zhang, L., Schappacher, F.M., Pöttgen, R., et al., J. Phys. Chem. C, 2008, vol. 112, pp. 8623–8628.
Wu, S., Huang, J., Zhuo, C., Zhang, F., et al., J. Inorg. Organomet. Polym. Mater., 2016, vol. 26, pp. 632–639.
Zhu, M. and Diao, G., Nanoscale, 2011, vol. 3, pp. 2748–2767.
Anirudhan, T. and Shainy, F., J. Colloid Interface Sci., 2015, vol. 456, pp. 22–31.
Mao, H., Wang, S., Lin, J.Y., Wang, Z., et al., J. Environ. Sci., 2016, vol. 49, pp. 179–188.
Badi, M.Y., Azari, A., Pasalari, H., Esrafili, A., et al., J. Mol. Liq., 2018, vol. 261, pp. 146–154.
Zhang, Z. and Kong, J., J. Hazard. Mater., 2011, vol. 15, pp. 325–329.
Mahdavi, M., Ahmad, M.B., Haron, M.J., Namvar, F., et al., Molecules, 2013, vol. 18, pp. 7533–7548.
Li, X., Wang, W., Dou, J., Gao, J., et al., J. Water Process. Eng., 2016, vol. 9, pp. e14–e20.
Li, Z., Hong, H., Liao, L., Ackley, C.J., et al., Colloids Surf. B, 2011, vol. 88, pp. 339–344.
Langmuir, I., J. Am. Chem. Soc., 1918, vol. 40, pp. 1361–1403.
Freundlich, H., J. Phys. Chem., 1906, vol. 57, pp. 384–470.
Park, J.C., Joo, J.B., and Yi, J., Korean J. Chem. Eng., 2005, vol. 22, pp. 276–280.
Lin, P., Zhang, Y., Zhang, X., Chen, C., et al., Front. Environ. Sci. Eng., 2015, vol. 9, pp. 138–146.
Fu, H., Li, X., Wang, J., Lin, P., et al., J. Environ. Sci., 2017, vol. 56, pp. 145–152.
Gao, Y., Li, Y., Zhang, L., Huang, H., et al., J. Colloid Interface Sci., 2012, vol. 368, pp. 540–546.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Zahoor, M., Ullah, A. & Alam, S. Removal of Enrofloxacin from Water through Magnetic Nanocomposites Prepared from Pineapple Waste Biomass. Surf. Engin. Appl.Electrochem. 55, 536–547 (2019). https://doi.org/10.3103/S1068375519050156
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1068375519050156