Abstract
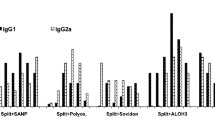
The objective of the study is to carry out preclinical research on the immunogenicity and protective properties of the trivalent influenza vaccine AdeVac-Flu in laboratory mice. The AdeVac-Flu vaccine is a mixture of recombinant human adenoviruses of the fifth serotype expressing the hemagglutinin genes of the A/California/07/2009(H1N1), A/Perth/16/2009(H3N2), and B/Brisbane/60/2008 influenza vaccine strains, as well as the molecular adjuvant Immunomax. The Ultravac live influenza vaccine (Microgen, Russia) was used as a reference. To study the protective properties of the vaccine, mice of the line Balb/c were immunized once intranasally and, 28 days after immunization, infected with lethal doses of influenza viruses A H1N1, H3N2, and B. The level of viral shedding from the lungs of mice was assessed on the third and sixth days after infection by titration of lung homogenates on MDCK cell culture. The level of specific antibodies to influenza viruses was determined by indirect ELISA, HAI, and SVN. It was shown that the trivalent influenza vaccine AdeVac-Flu is more immunogenic than the reference drug. The levels of protection against homologous strains of influenza viruses are either slightly less than or comparable to those of the reference drug. In this case, it was shown that the AdeVac-Flu, like the live influenza vaccine, effectively protects mice from a heterologous strain of the influenza virus, although the mice exhibited excretion of the virus from the lungs and a slight decrease in body weight (less than 10%). It has been shown that the AdeVac-Flu vaccine based on recombinant human adenoviruses of the fifth serotype expressing hemagglutinin genes of A/California/07/2009 (H1N1), A/Perth/16/2009 (H3N2), and B/Brisbane/60/2008 influenza viruses has immunogenicity and protective properties in relation to both homologous and heterologous (within the subtype) strains of the influenza virus.


Similar content being viewed by others
REFERENCES
Paules, C. and Subbarao, K., Influenza, Lancet, 2017, vol. 390, pp. 697–708. https://doi.org/10.1016/S0140-6736(17)30129-0
Zhang, Y., Xu, C., Zhang, H., Liu, G.D., Xue, C., and Cao, Y., Targeting Hemagglutinin: Approaches for broad protection against the influenza A virus, Viruses, 2019, vol. 11, no. 5, p. E405. https://doi.org/10.3390/v11050405
Sedova, E.S., Shcherbinin, D.N., Migunov, A.I., Smirnov, Yu.A., Logunov, D.Yu., Shmarov, M.M., et al., Recombinant influenza vaccines, ActaNaturae, 2012, vol. 4, no. 4, pp. 17–27.
Tompkins, S.M., Lin, Y., Leser, G.P., Kramer, K.A., Haas, D.L., Howerth, E.W., et al., Recombinant parainfluenza virus 5 (PIV5) expressing the influenza A virus hemagglutinin provides immunity in mice to influenza A virus challenge, Virology, 2007, vol. 362, pp. 139–150. https://doi.org/10.1016/j.virol.2006.12.005
Ertl, H.C., Viral vectors as vaccine carriers, Curr. Opin. Virol., 2016, vol. 21, pp. 1–8. https://doi.org/10.1016/j.coviro.2016.06.001
Hoelscher, M.A., Singh, N., Garg, S., Jayashankar, L., Veguilla, V., Pandey, A., et al., A broadly protective vaccine against globally dispersed clade 1 and clade 2 H5N1 influenza viruses, J. Infect. Dis., 2008, vol. 197, no. 8, pp. 1185–1188. https://doi.org/10.1086/529522
Melidou, A., Gioula, G., Exindari, M., Chatzidimitriou, D., and Diza-Mataftsi, E., Influenza A(H5N1): an overview of the current situation, Eurosurveillance, 2009, vol. 14, no. 20. https://doi.org/10.2807/ese.14.20.19216-en
Throsby, M., van den Brink, E., Jongeneelen, M., Poon, L.L., Alard, P., Cornelissen, L., et al., Heterosubtypic neutralizing monoclonal antibodies cross-protective against H5N1 and H1N1 recovered from human IgM+ memory B cells, PLoS One, 2008, vol. 3, no. 12, p. e3942. https://doi.org/10.1371/journal.pone.0003942
Shmarov, M.M., Sedova, E.S., Verkhovskaya, L.V., Rudneva, I.A., Bogacheva, E.A., Barykova, Y.A., et al., induction of a protective heterosubtypic immune response against the influenza virus by using recombinant adenoviral vectors expressing hemagglutinin of the influenza H5 virus, ActaNaturae, 2010, vol. 2, no. 1, pp. 111–118.
Ataullakhanov, R.I., Pichugin, A.V., Shishkova, N.M., Masternak, T.B., Malkina, E.Yu., Ulyanova, L.I., et al., Cell mechanisms of the immunomodulating action produced by “Immunomax” drug, Immunologiya, 2005, vol. 26, no. 2, pp. 111–120.
Bagaev, A.V., Pichugin, A.V., Lebedeva, E.S., Lysenko, A.A., Shmarov, M.M., Logunov, D.Yu., et al., Regulation of the target protein (transgene) expression in the adenovirus vector using agonists of toll-like receptors, ActaNaturae, 2014, vol. 6, no. 4, pp. 27–39.
Bagaev, A.V., Pichugin, A.V., Lebedeva, E.S., Lysenko, A.A., Shmarov, M.M., Logunov, D.Yu., et al., Influence of TLR-agonists on expression by antigen-presenting cells of the target protein antigen encoded in adenoviral vector, Immunologiya, 2015, vol. 36, no. 4, pp. 188–195.
MR (Methodological Recommendations) no. 3.3.2.1758-03: Methods for determining the quality indicators of immunobiological drugs for the prevention and diagnosis of influenza, Moscow: 2003.
MR (Methodological Recommendations): Detection of antibodies to influenza A (H5N1) in the sera of humans and animals under natural infection and vaccination process in the reaction of microneutralization, Moscow, 2009.
Stepanova, L.A., Sergeeva, M.V., Shuklina, M.A., Shaldzhyan, A.A., Potapchuk, M.V., Korotkov, A.V., et al., A fusion protein based on the second subunit of hemagglutinin of influenza A/H2N2 viruses provides cross immunity, ActaNaturae, 2016, vol. 8, no. 2, pp. 116–126.
Tutykhina, I.L., Sedova, E.S., Gribova, I.Y., Ivanova, T.I., Vasilev, L.A., and Rutovskaya, M.V., Passive immunization with a recombinant adenovirus expressing an HA (H5)-specific single-domain antibody protects mice from lethal influenza infection, Antiviral Res., 2013, vol. 97, pp. 318–328. https://doi.org/10.1016/j.antiviral.2012.12.021
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
Conflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Translated by A. Ostyak
Sedova E.S., https://orcid.org/0000-0001-6959-9988; e-mail: sedovaes@yandex.ru
Stepanova L.A., https://orcid.org/0000-0002-8833-9409
Lysenko A.A., https://orcid.org/0000-0002-0230-1822
Shcherbinin D.N., https://orcid.org/0000-0002-8518-1669
Verkhovskaya L.V., https://orcid.org/0000-0002-4731-1629
Tsybalova L.M., https://orcid.org/0000-0002-1193-5907
Shmarov M.M., https://orcid.org/0000-0002-5268-1296
About this article
Cite this article
Sedova, E.S., Stepanova, L.A., Lysenko, A.A. et al. A Study of the Immunogenicity and Protective Properties of Recombinant Influenza Vaccine. Mol. Genet. Microbiol. Virol. 35, 159–167 (2020). https://doi.org/10.3103/S0891416820030088
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0891416820030088