Abstract
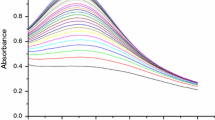
Silver nanoparticle (AgNP) has been synthesized using adrenaline. Adrenaline readily undergoes an autoxidation reaction in an alkaline medium with the dissolved oxygen to form adrenochrome, thus behaving as a mild reducing agent for the dissolved oxygen. This reducing behavior of adrenaline when employed to reduce Ag+ ions yielded a large enhancement in the intensity of absorbance in the visible region. Transmission electron microscopy (TEM) and X-ray diffraction (XRD) studies have been performed to confirm the surface morphology of AgNPs. Further, the metallic nanoparticles with size greater than 2 nm caused a strong and broad absorption band in the UV-visible spectrum called surface plasmon band or Mie resonance. The formation of AgNPs caused the large enhancement in the absorbance values with λmax at 436 nm through the excitation of the surface plasmon band. The formation of AgNPs was adopted to for the quantitative assessment of adrenaline using spectrophotometry with lower detection limit and higher precision values.
Similar content being viewed by others
References
A. Henglein and D. Meisel, J. Phys. Chem. B, 1998, 102, 8364.
R. Jana, T. K. Sau, and T. Pal, J. Phys. Chem. B, 1999, 103, 115.
K. Mallick, M. Witcomb, and M. Scurrell, Mater. Chem. Phys., 2006, 97, 283.
X. P. Miao, K. Han, H. Sun, J. Yin, J. Zhao, B. Wang, and Y. Tang, Appl. Mater. Interfaces, 2014, 6, 8667.
K. M. M. A. El-Nour, A. Eftaiha, A. Al-Warthan, and R. A. A. Ammar, Arab. J. Chem., 2010, 3, 135.
X. Gan, T. Liu, X. Zhu, and G. Li, Anal. Sci., 2004, 20, 1271.
H. Wang, D. Chen, Y. Wei, Y. Chang, and J. Zhao, Anal. Sci., 2011, 27, 937.
H. W. Park, S. M. Alam, S. H. Lee, M. M. Karim, S. M. Wabaidur, M. Kang, and J. H. Choi, Luminescence, 2009, 24, 367.
S. Yamamoto, K. Fujiwara, and H. Watarai, Anal. Sci., 2004, 20, 1347.
J. C. Riboh, A. J. Haes, A. D. Mcfarland, C. R. Yonzon, and R. P. Van Duyne, J. Phy. Chem. B, 2003, 107, 1772.
T. Yamashima and J. Takamine, J. Med. Biogr., 2003, 11, 95.
K. H. Berecek and M. J. Brody, Am. J. Physiol., 1982, 242, H593.
Y. K. Kothari and K. Srinivasulu, Asian J. Chem., 1989, 1, 42.
Z. A. Alothman, M. R. Siddiqui, and M. A. Khan, Asian J. Chem., 2013, 25, 7229.
N. Rahman, M. R. Siddiqui, and S. N. H. Azmi, Chem. Pharm. Bull., 2006, 546, 26.
Y.-C. Xie, H.-W. Huang, Q.-M. Zhang, and S.-H. Jin, Chem. Res. Chin. Univ., 2009, 25, 433.
A. K. Mishra, A. Mishra, and P. Chattopadhyay, Arch. Appl. Sci. Res., 2010, 2, 251.
A. Mishra, A. Upadhyay, A. Patra, S. Chaudhury, and P. Chattopadhyay, Sci. Pharm., 2009, 77, 367.
L. K. Abdulrahman, A. M. Al-Abachi, and M. H. Al-Qaissy, Anal. Chim. Acta, 2005, 538, 331.
H. J. Vieira and O. Fatibello-Filho, Ecletica Quim., 2004, 29, 79.
C. Ji, J. Walton, Y. Su, and M. Tella, Anal. Chim. Acta, 2010, 670, 84.
B. J. Petteys, K. S. Graham, M. L. Parnas, C. Holt, and E. L. Frank, Clin. Chim. Acta, 2012, 413, 1459.
M. J. Hamzah, A. B. M. Mahood, and S. A. Abid, J. Karbala Univ., 2009, 7, 9.
M. E. Al-Kommos, F. A. Mohamed, and A. S. Khedr, Talanta, 1990, 37, 625.
M. R. H. Nezhad, J. Tashkhourian, and J. Khodaveisi, J. Iran. Chem. Soc., 2010, 7, S83.
H. W. Park, S. M. Alam, S. H. Lee, M. M. Karim, S. M. Wabaidur, M. Kang, and J. H. Choi, Luminescence, 2009, 24, 367.
J. Riboh, A. Haes, A. McFarland, C. Yonzon, and R. Van Duyne, J. Phys. Chem. B, 2003, 107, 1772.
M. Malinsky, K. Kelly, G. Schatz, and R. Van Duyne, J. Am. Chem. Soc., 2001, 123, 1471.
Validation of analytical procedures: text and methodology Q2(R1). Current Step 4 version Parent Guideline dated 27 October 1994. (Complementary Guideline on Methodology dated 6 November 1996 incorporated in November 2005).
International conference on Harmonisation, ICH Harmonised Tripartite Guideline - Text on Validation of Analytical Procedures, Fed. Regist. 60, 11260 (1995).
M. Z. A. Rafiquee, M. R. Siddiqui, M. S. Ali, H. A. Al-Lohedan, and Z. A. Al-Othman, Bioprocess Biosyst. Eng., 2014, doi: 10.1007/s00449-014-1311-5.
R. Baron, M. Zayats, and I. Willner, Anal. Chem., 2005, 77, 1566.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research College of Science Research Center, King Saud University, Riyadh, Saudi Arabia, for supporting this project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Siddiqui, M.R., Rafiquee, M.Z.A., Wabaidur, S.M. et al. Synthesis of Silver Nanoparticle: A New Analytical Approach for the Quantitative Assessment of Adrenaline. ANAL. SCI. 31, 437–443 (2015). https://doi.org/10.2116/analsci.31.437
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.31.437