Abstract
Background
The treatment of difficult-to-treat wounds can be challenging. Although a number of approaches have been investigated, the healing process may be slow and unsatisfactory. An alternative approach is the use of a continuous sheet of skin cells applied over a wound which may improve cell implantation and patient recovery.
Objectives
To analyse the gene expression profile of fibroblast/keratinocyte co-culture on poly(tri[ethylene glycol] ethyl ether methacrylate) (P[TEGMA-EE]), a thermoresponsive biocompatible surface.
Materials and Methods
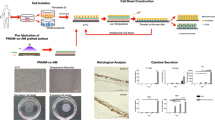
Cultures were grown for 72 hours as a continuous layer on P(TEGMA-EE). Assays for genotoxicity, cell morphology, and fluorescence-assisted flow cytometry were performed to exclude adverse effects. A gene expression profile related to the extracellular matrix was investigated by microarray analysis.
Results
for fibroblast monocultures and fibroblast/keratinocyte co-cultures maintained for 72 hours on P(TEGMA-EE), no change in morphology or specific surface markers, or DNA damage (comet assay) was observed, relative to control surface. Moreover, no detrimental impact was ascertained based on microarray analysis. In response to lowered temperature, the detachment of a continuous cell layer sheet from the thermoresponsive surface was observed. When gene expression was compared between fibroblasts cultured alone and co-cultured with keratinocytes on P(TEGMA-EE), 10 genes were shown to be differentially expressed. Of these genes, six were significantly differentially expressed between cultures grown on P(TEGMA-EE) and human skin samples.
Conclusion
Our results indicate that P(TEGMA-EE) is fully biocompatible and is therefore a suitable surface for successful preparation and recovery of two-layered fibroblast/keratinocyte co-culture as a continuous sheet of cells.
Similar content being viewed by others
References
Ehrenreich M, Ruszczak Z. Tissue-engineered temporary wound coverings. Important options for the clinician. Acta Dermatovenerol Alp Pannonica Adriat 2006; 15: 5–13.
Pham C, Greenwood J, Cleland H, Woodruff P, Maddern G. Bioengineered skin substitutes for the management of burns: a systematic review. Burns 2007; 33: 946–57.
Bechetoille N, Dezutter-Dambuyant C, Damour O, Andre V, Orly I, Perrier E. Effects of solar ultraviolet radiation on engineered human skin equivalent containing both Langerhans cells and dermal dendritic cells. Tissue Eng 2007; 13: 2667–79.
Blanpain C. Stem cells: skin regeneration and repair. Nature 2010; 464: 686–7.
Marston WA. Dermagraft, a bioengineered human dermal equivalent for the treatment of chronic nonhealing diabetic foot ulcer. Expert Rev Med Devices 2004; 1: 21–31.
Orlando G, Wood KJ, De Coppi P, et al. Regenerative medicine as applied to general surgery. Ann Surg 2012; 255: 867–80.
Everts PA, Jakimowicz JJ, van Beek M, et al. Reviewing the structural features of autologous platelet-leukocyte gel and suggestions for use in surgery. Eur Surg Res 2007; 39: 199–2007.
Kurkuri MD, Nussio MR, Deslandes A, Voelcker NH. Thermosensitive copolymer coatings with enhanced wettability switching. Langmuir 2008; 24: 4238–44.
Hom DB, Linzie BM, Huang TC. The healing effects of autologous platelet gel on acute human skin wounds. Arch Facial Plast Surg 2007; 9: 174–83.
Ikada Y. Challenges in tissue engineering. J R Soc Interface 2008; 3: 589–601.
Ebara M, Yamato M, Hirose M, et al. Copolymerization of 2-carboxyisopropylacrylamide with N-isopropylacrylamide accelerates cell detachment from grafted surfaces by reducing temperature. Biomacromolecules 2003; 4: 344–9.
Kikuchi A, Okano T. Nanostructured designs of biomedical materials: applications of cell sheet engineering to functional regenerative tissues and organs. J Control Release 2005; 101: 69–84.
Nitschke M, Gramm S, Gotze T, et al. Thermo-responsive poly(NiPAAm-co-DEGMA) substrates for gentle harvest of human corneal endothelial cell sheets. J Biomed Mater Res A 2007; 80: 1003–10.
Nagase K, Kobayashi J, Okano T. Temperature-responsive intelligent interfaces for biomolecular separation and cell sheet engineering. J R Soc Interface 2009; 6: S293–309.
Yamato M, Akiyama Y, Kobayashi J, Yang J, Kikuchi A, Okano T. Temperature-responsive cell culture surfaces for regenerative medicine with cell sheet engineering. Prog Polym Sci 2007; 32: 1123–33.
Harimoto M, Yamato M, Hirose M, et al. Novel approach for achieving double-layered cell sheets co-culture: overlaying endothelial cell sheets onto monolayer hepatocytes utilizing temperature-responsive culture dishes. J Biomed Mater Res 2002; 62: 464–70.
Kim K, Ohashi K, Utoh R, Kano K, Okano T. Preserved liverspecific functions of hepatocytes in 3D co-culture with endothelial cell sheets. Biomaterials 2012; 33: 1406–13.
Kessel S, Schmidt S, Muller R, et al. Thermoresponsive PEG-based polymer layers: surface characterization with AFM force measurements. Langmuir 2010; 26: 3462–7.
Uhlig K, Wischerhoff E, Lutz JF, et al. Monitoring cell detachment on PEG-based thermoresponsive surfaces using TIRF microscopy. Soft Matter 2010; 6: 4262–7.
Uhlig K, Boysen B, Lankenau A, et al. On the influence of the architecture of poly(ethylene glycol)-based thermoresponsive polymers on cell adhesion. Biomicrofluidics 2012; 6: 24129.
Wischerhoff E, Uhlig K, Lankenau A, et al. Controlled cell adhesion on PEG-based switchable surfaces. Angew Chem Int Ed 2008; 47: 5666–8.
Wischerhoff E, Glatzel S, Uhlig K, Lankenau A, Lutz JF, Laschewsky A. Tuning the thickness of polymer brushes grafted from nonlinearly growing multilayer. Langmuir 2009; 25: 5949–56.
Dworak A, Utrata-Wesolek A, Szweda D, et al. Poly(tri[ethylene glycol] ethyl ether methacrylate)-coated surfaces for controlled fibroblasts culturing. ACS Appl Mater Interfaces 2013; 5: 2197–207.
Dworak A, Utrata-Wesolek A, Oleszko N, et al. Poly(2-substituted-2-oxazoline) surfaces for dermal fibroblasts adhesion and detachment. J Mater Sci Mater Med 2014; 25: 1149–63.
Oleszko N, Wałach W, Utrata-Wesolek A, et al. Controlling the crystallinity of thermoresponsive poly(2-oxazoline)-based nanolayers to cell adhesion and detachment. Biomacromolecules 2015; 16: 2805–13.
Rosenberger A, Rossler U, Hornhardt S, et al. Validation of a fully automated COMET assay: 1.75 million single cells measured over a 5-year period. DNA Repair 2011; 10: 322–37.
Collins AR. The comet assay for DNA damage and repair: principles, applications, and limitations? Mol Biotechnol 2004; 26: 249–61.
Kawecki M, Kraut M, Klama-Baryla A, et al. Transfer of fibroblast sheets cultured on thermoresponsive dishes with membranes. J Mater Sci Mater Med 2016; 27: 1–12.
Saalbach A, Aust G, Haustein UF, Herrmann K, Anderegg U. The fibroblast-specific MAb AS02: a novel tool for detection and elimination of human fibroblasts. Cell Tissue Res 1997; 290: 593–9.
Saalbach A, Kraft R, Herrmann K, Haustein UF, Anderegg U. The monoclonal antibody AS02 recognizes a protein on human fibroblasts being highly homologous to Thy-1. Arch Dermatol Res 1998; 290: 360–6.
Theocharidis G, Drymoussi Z, Kao AP, et al. Type VI collagen regulates dermal matrix assembly and fibroblast motility. J Invest Dermatol 2016; 136: 74–83.
Horrocks C, Duncan JI, Oliver AM, Thomson AW. Adhesion molecule expression in psoriatic skin lesions and the influence of cyclosporin A. Clin Exp Immunol 1991; 84: 157–62.
Schleiffenbaum B, Spertini O, Tedder TF. Soluble L-selectin is present in human plasma at high levels and maintains functional activity. J Cell Biol 1992; 199: 229–38.
Wick M, Burger C, Brusselbach S, Lucibello FC, Muller R. A novel member of human tissue inhibitor of metalloproteinases (TIMP) gene family is regulated during G1 progression, mitogenic stimulation, differentiation, and senescence. J Biol Chem 1994; 269: 18953–60.
Butler GS, Apte SS, Willenbrock F, Murphy G. Human tissue inhibitor of metalloproteinases 3 interacts with both the N-and C-terminal domains of gelatinases A and B. Regulation by polyanions. J Biol Chem 1999; 274: 10846–51.
Baker AH, Murphy G, Newby AC. Inhibition of invasion and induction of apoptotic cell death of cancer cell lines by overexpression of TIMP-3. Br J Cancer 1999; 79: 1347–55.
Gaidarova S, Jiménez SA. Inhibition of basal and transforming growth factor-beta-induced stimulation of COL1A1 transcription by the DNA intercalators, mitoxantrone and WP631, in cultured human dermal fibroblasts. J Biol Chem 2002; 277: 38737–45.
Kim ES, Sohn YW, Moon A. TGF-beta-induced transcriptional activation of MMP-2 is mediated by activating transcription factor (ATF)2 in human breast epithelial cells. Cancer Lett 2007; 252: 147–56.
Crawford SE, Stellmach V, Murphy-Ullrich JE, et al. Thrombospondin-1 is a major activator of TGF-beta1 in vivo. Cell 1998; 93: 1159–70.
Yung S, Lee CY, Zhang Q, Lau SK, Tsang RC, Chan TM. Elevated glucose induction of thrombospondin-1 up-regulates fibronectin synthesis in proximal renal tubular epithelial cells through TGF-beta1 dependent and TGF-beta1 independent pathways. Nephrol Dial Transplant 2006; 21: 1504–13.
Yu Q, Stamenkovic I. Cell surface-localized matrix metalloproteinase-9 proteolytically activates TGF-beta and promotes tumor invasion and angiogenesis. Genes Dev 2000; 14: 163–76.
Young GD, Murphy-Ullrich JE. The tryptophan-rich motifs of the thrombospondin type 1 repeats bind VLAL motifs in the latent transforming growth factor-beta complex. J Biol Chem 2004; 279: 47633–42.
Pellegrini M, Marcotte EM, Thompson MJ, Eisenberg D, Yeates TO. Assigning protein functions by comparative genome analysis: protein phylogenetic profiles. Proc Natl Acad Sci USA 1999; 96: 4285–8.
Ellerbroek SM, Wu YI, Stack MS. Type I collagen stabilization of matrix metalloproteinase-2. Arch Biochem Biophys 2001; 390: 51–6.
Marcotte EM, Pellegrini M, Thompson MJ, Yeates TO, Eisenberg D. A combined algorithm for genome-wide prediction of protein function. Nature 1999; 402: 83–6.
Safina A1, Vandette E, Bakin AV. ALK5 promotes tumor angiogenesis by upregulating matrix metalloproteinase-9 in tumor cells. Oncogene 2007; 26: 2407–22.
Bartscht T, Lehnert H, Gieseler F, Ungefroren H. The Src family kinase inhibitors PP2 and PP1 effectively block TGF-beta1-induced cell migration and invasion in both established and primary carcinoma cells. Pharmacol 2012; 70: 221–30.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Lesiak, M., Sieroń, Ł., Gutmajster, E. et al. Fibroblast and keratinocyte crosstalk: the effect of a poly(tri[ethylene glycol] ethyl ether methacrylate) thermoresponsive surface on short-term co-culture. Eur J Dermatol 29, 126–140 (2019). https://doi.org/10.1684/ejd.2019.3561
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2019.3561