Abstract
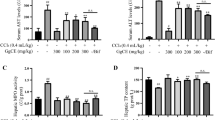
The present study was undertaken to investigate the possible protective effect of L-carnosine (CAR), an endogenous dipeptide of alanine and histidine, on carbon tetrachloride (CCl4)-induced hepatic injury. Liver injury was induced in male Sprague-Dawley rats by intraperitoneal (i.p.) injections of CCl4, twice weekly for six weeks. CAR was administered to rats daily, at dose of 250 mg/kg, i.p. At the end of six weeks, blood and liver tissue specimens were collected. Results show that CAR treatment attenuated the hepatic morphological changes, necroinflammation and fibrosis induced by CCl4, as indicated by hepatic histopathology scoring. In addition, CAR treatment significantly reduced the CCl4-induced elevation of liver-injury parameters in serum. CAR treatment also combated oxidative stress; possibly by restoring hepatic nuclear factor erythroid 2-related factor 2 (Nrf-2) levels. Moreover, CAR treatment prevented the activation of hepatic stellate cells (HSCs), as indicated by reduced α-smooth muscle actin (α-SMA) expression in the liver, and decreased hepatic inflammation as demonstrated by a reduction in hepatic tumor necrosis factor-α (TNF-α) and restoration of interleukin-10 (IL-10) levels. In conclusion, CCl4-induced hepatic injury was alleviated by CAR treatment. The results suggest that these beneficial, protective effects are due, at least in part, to its anti-oxidant, anti-inflammatory and anti-fibrotic activities.
Similar content being viewed by others
References
Huang X, Wang X, Lv Y, Xu L, Lin J, Diao Y. Protection effect of kallistatin on carbon tetrachloride-induced liver fibrosis in rats via antioxidative stress. PLoS One 2014; 18:9.
Balsano C, Alisi A, Nobili V. Liver fibrosis and therapeutic strategies: the goal for improving metabolism. Curr Drug Targets 2009; 10: 505–12.
Luedde T, Schwabe RF. NF-κB in the liver–linking injury, fibrosis and hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 2011; 8: 108–18.
Gäbele E, Brenner DA, Rippe RA. Liver fibrosis: signals leading to the amplification of the fibrogenic hepatic stellate cell. Front Biosci 2003; 1: 69–77.
Zhou WC, Zhang QB, Qiao L. Pathogenesis of liver cirrhosis. World J Gastroenterol 2014; 20: 7312–24.
Brenner DA. Molecular pathogenesis of liver fibrosis. Trans Am Clin Climatol Assoc 2009; 120: 361–8.
Tan Z, Qian X, Jiang R, et al. IL-17A plays a critical role in the pathogenesis of liver fibrosis through hepatic stellate cell activation. J Immunol 2013; 191: 1835–44.
Zhao X, Song JL, Kil JH, Park KY. Bamboo salt attenuates CCl4-induced hepatic damage in Sprague-Dawley rats. Nutr Res Pract 2013; 7: 273–80.
Yeh YH, Hsieh YL, Lee YT. Effects of yam peel extract against carbon tetrachloride-induced hepatotoxicity in rats. J Agric Food Chem 2013; 61: 7387–96.
Weber LW, Boll M, Stampfl A. Hepatotoxicity and mechanism of action of haloalkanes: carbon tetrachloride as a toxicological model. Crit Rev Toxicol 2003; 33: 105–36.
Kumar H, Kim IS, More SV, Kim BW, Choi DK. Natural productderived pharmacological modulators of Nrf2/ARE pathway for chronic diseases. Nat Prod Rep 2014; 31: 109–39.
Scannevin RH, Chollate S, Jung MY, et al. Fumarates promote cytoprotection of central nervous system cells against oxidative stress via the nuclear factor (erythroid-derived 2)-like 2 pathway. J Pharmacol Exp Ther 2012; 341: 274–84.
Ditte Z, Ditte P, Labudova M, et al. Carnosine inhibits carbonic anhydrase IX-mediated extracellular acidosis and suppresses growth of HeLa tumor xenografts. BMC Cancer 2014: 14–358.
Zhang Z, Miao L, Wu X, et al. Carnosine Inhibits the Proliferation of Human Gastric Carcinoma Cells by Retarding Akt/mTOR/p70S6K Signaling. J Cancer 2014; 5: 382–9.
Aydin AF, Küskü-Kiraz Z, Doğru-Abbasoğlu S, Güllüoğlu M, Uysal M, Koçak-Toker N. Effect of carnosine against thioacetamideinduced liver cirrhosis in rat. Peptides 2010; 31: 67–71.
Rashid I, van Reyk DM, Davies MJ. Carnosine and its constituents inhibit glycation of low-density lipoproteins that promots foamcell formation in vitro. FEBS Lett 2007; 581: 1067–70.
Riedl E, Pfister F, Braunagel M, et al. Carnosine prevents apoptosis of glomerular cells and podocyte loss in STZ diabetic rats. Cell Physiol Biochem 2011; 28: 279–88.
Hipkiss AR, Brownson C, Carrier MJ. Carnosine, the anti-ageing, anti-oxidant dipeptide, may react with protein carbonyl groups. Mech Ageing Dev 2001; 122: 1431–45.
Bellia F, Vecchio G, Cuzzocrea S, Calabrese V, Rizzarelli E. Neuroprotective features of carnosine in oxidative driven diseases. Mol Aspects Med 2011; 32: 258–66.
Hipkiss AR. Could carnosine or related structures suppress Alzheimer’s disease? J Alzheimers Dis 2007; 11: 229–40.
Artun BC, Küskü-Kiraz Z, Güllüoğlu M, Cevikbaş U, Koçak-Toker N, Uysal M. The effect of carnosine pretreatment on oxidative stress and hepatotoxicity in binge ethanol administered rats. Hum Exp Toxicol 2010; 29: 659–65.
Giriş M, Doğru-Abbasoğlu S, Kumral A, Olgaç V, Koçak-Toker N, Uysal M. Effect of carnosine alone or combined with α-tocopherol on hepatic steatosis and oxidative stress in fructose-induced insulinresistant rats. J Physiol Biochem 2014; 70: 385–95.
Mehmetçik G, Ozdemirler G, Koçak-Toker N, Cevikbaş U, Uysal M. Role of carnosine in preventing thioacetamide-induced liver injury in the rat. Peptides 2008; 29:425.
Aydín AF, Küskü-Kiraz Z, Doğru-Abbasoğlu S, Uysal M. Effect of carnosine treatment on oxidative stress in serum, apoB-containing lipoproteins fraction and erythrocytes of aged rats. Pharmacol Rep 2010: 733–9.
Elsherbiny NM, Abd El Galil KH, Gabr MM, Al-Gayyar MM, Eissa LA, El-Shishtawy MM. Reno-protective effect of NECA in diabetic nephropathy: implication of IL-18 and ICAM-1. Eur Cytokine Netw 2012; 23: 78–86.
DeChatelet LR, McCall CE, McPhail LC, Johnston Jr. RB. Superoxide dismutase activity in leukocytes. J Clin Invest 1974; 53: 1197–201.
Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys 1959; 82: 70–7.
Ishak K, Baptista A, Bianchi L, et al. Histological grading and staging of chronic hepatitis. J Hepatol 1995; 22: 696–9.
Maheshwari DT, Yogendra Kumar MS, Verma SK, Singh VK, Singh SN. Antioxidant and hepatoprotective activities of phenolic rich fraction of Seabuckthorn (Hippophae rhamnoides L.) leaves. Food Chem Toxicol 2011; 49: 2422–8.
Singh N, Kamath V, Narasimhamurthy K, Rajini PS. Protective effect of potato peel extract against carbon tetrachloride-induced liver injury in rats. Environ Toxicol Pharmacol 2008; 26: 241–6.
Shim JY, Kim MH, Kim HD, Ahn JY, Yun YS, Song JY. Protective action of the immunomodulator ginsan against carbon tetrachloride-induced liver injury via control of oxidative stress and the inflammatory response. Toxicol Appl Pharmacol 2010; 242: 318–25.
Weber LW, Boll M, Stampfl A. Hepatotoxicity and mechanism of action of haloalkanes: carbon tetrachloride as a toxicological model. Crit Rev Toxicol 2003; 33: 105–36.
Ma JQ, Ding J, Zhang L, Liu CM. Protective effects of ursolic acid in an experimental model of liver fibrosis through Nrf2/ARE pathway. Clin Res Hepatol Gastroenterol 2015; 39: 188–97.
Martin-Aragón S, de las Heras B, Sanchez-Reus MI, Benedi J. Pharmacological modification of endogenous antioxidant enzymes by ursolic acid on tetrachloride-induced liver damage in rats and primary cultures of rat hepatocytes. Exp Toxicol Pathol 2001; 53: 199–206.
Feng Y, Siu KY, Ye X, et al. Hepatoprotective effects of berberine on carbon tetrachloride-induced acute hepatotoxicity in rats. Chin Med 2010; 18: 5–33.
Al-Olayan EM, El-Khadragy MF, Aref AM, Othman MS, Kassab RB, Abdel Moneim AE. The potential protective effect of Physalis peruviana L. against carbon tetrachloride-induced hepatotoxicity in rats is mediated by suppression of oxidative stress and downregulation of MMP-9 expression. Oxid Med Cell Longev 2014; 2014: 381413.
Kim MY, Kim EJ, Kim YN, Choi C, Lee BH. Effects of α-lipoic acid and L-carnosine supplementation on antioxidant activities and lipid profiles in rats. Nutr Res Pract 2011; 5: 421–8.
Kumar H, Kim IS, More SV, Kim BW, Choi DK. Natural productderived pharmacological modulators of Nrf2/ARE pathway for chronic diseases. Nat Prod Rep 2014; 31: 109–39.
Xu W, Hellerbrand C, Köhler UA, Bugnon P, et al. The Nrf2 transcription factor protects from toxin-induced liver injury and fibrosis. Lab Invest 2008; 88: 1068–78.
Wu S, Yue Y, Tian H, et al. Carthamus red from Carthamus tinctorius L. exerts antioxidant and hepatoprotective effect against CCl-induced liver damage in rats via the Nrf2 pathway. J Ethnopharmacol 2013; 148: 570–8.
Li JP, Gao Y, Chu SF, et al. Nrf2 pathway activation contributes to anti-fibrosis effects of ginsenoside Rg1 in a rat model of alcohol-and CCl4-induced hepatic fibrosis. Acta Pharmacol Sin 2014; 35: 1031–44.
Li N, Xia T, Nel AE. The role of oxidative stress in ambient particulate matter-induced lung diseases and its implications in the toxicity of engineered nanoparticles. Free Radic Biol Med 2008; 44: 1689–99.
Niture SK, Kaspar JW, Shen J, Jaiswal AK. Nrf2 signaling and cell survival. Toxicol Appl Pharmacol 2010; 244: 37–42.
Rocha SW, de Franşa ME, Rodrigues GB, et al. Diethylcarbamazine reduces chronic inflammation and fibrosis in carbon tetrachloride-(CCl4-) induced liver injury in mice. Mediators Inflamm 2014; 2014: 696383.
Shin DS, Kim KW, Chung HY, Yoon S, Moon JO. Effect of sinapic acid against carbon tetrachloride-induced acute hepatic injury in rats. Arch Pharm Res 2013; 36: 626–33.
O’Shea JJ, Murray PJ. Cytokine signaling modules in inflammatory responses. Immunity 2008; 28: 477–87.
Arai T, Hiromatsu K, Kobayashi N, et al. IL-10 is involved in the protective effect of dibutyryl cyclic adenosine monophosphate on endotoxin-induced inflammatory liver injury. J Immunol 1995; 155: 5743–9.
Son DO, Satsu H, Kiso Y, Totsuka M, Shimizu M. Inhibitory effect of carnosine on interleukin-8 production in intestinal epithelial cells through translational regulation. Cytokine. 2008; 42: 265–76.
Sahin S, Oter S, Burukoğlu D, Sutken E. The effects of carnosine in an experimental rat model of septic shock. Med Sci Monit Basic Res 2013; 19: 54–61.
Al-Shabanah OA, Alam K, Nagi MN, Al-Rikabi AC, Al-Bekairi AM. Protective effect of aminoguanidine, a nitric oxide synthase inhibitor, against carbon tetrachloride induced hepatotoxicity in mice. Life Sci 2000; 66: 265–70.
Coleman JW. Nitric oxide in immunity and inflammation. Int Immunopharmacol 2001; 1: 1397–406.
Chen T, Zamora R, Zuckerbraun B, Billiar TR. Role of nitric oxide in liver injury. Curr Mol Med 2003; 3: 519–26.
Teraoka R, Shimada T, Aburada M. The molecular mechanisms of the hepatoprotective effect of gomisin A against oxidative stress and inflammatory response in rats with carbon tetrachloride-induced acute liver injury. Biol Pharm Bull 2012; 35: 171–7.
Xiao J, Liong EC, Ching YP, et al. Lycium barbarum polysaccharides protect mice liver from carbon tetrachloride-induced oxidative stress and necroinflammation. J Ethnopharmacol 2012; 139: 462–70.
De Minicis S, Rychlicki C, Agostinelli L, et al. Semaphorin 7A contributes to TGF-β-mediated liver fibrogenesis. Am J Pathol 2013; 183: 820–30.
López-Reyes AG, Arroyo-Curras N, Cano BG, et al. Black bean extract ameliorates liver fibrosis in rats with CCl4-induced injury. Ann Hepatol 2008; 7: 130–5.
Soliman KM, Abdel Aziz M, Nassar YH, Abdel-Sattar S, El-Ansary A. Effects of carnosine on bilharzial infestation in hamsters: biochemical and histochemical studies. Comp Biochem Physiol B Biochem Mol Biol 2002; 131: 535–42.
Cuzzocrea S, Genovese T, Failla M, et al. Protective effect of orally administered carnosine on bleomycin-induced lung injury. Am J Physiol Lung Cell Mol Physiol 2007; 292: 1095–104.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Alsheblak, M.M., Elsherbiny, N.M., El-Karef, A. et al. Protective effects of L-carnosine on CCl4 -induced hepatic injury in rats. Eur Cytokine Netw 27, 6–15 (2016). https://doi.org/10.1684/ecn.2016.0372
Published:
Issue Date:
DOI: https://doi.org/10.1684/ecn.2016.0372