Abstract
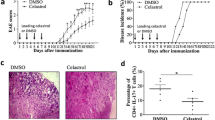
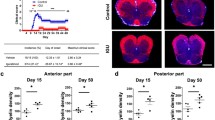
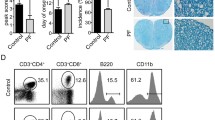
The present study was conducted to clarify the therapeutic effect of cornuside on experimental autoimmune encephalomyelitis (EAE) and its influence on T helper 17 (Th17) cell and regulatory T (Treg) cell infiltration into the central nervous system. Rats were randomly placed into four treatment groups: control, EAE, EAE+cornuside, and EAE+prednisolone. The neurological function scores of rats were assessed daily. On the second day after EAE rats began to show neurological deficit symptoms, the four groups were treated with normal saline, normal saline, cornuside (150 mg/kg), and prednisolone (5 mg/kg), respectively. The treatment was discontinued after two weeks, and the spinal cord was obtained for hematoxylin and eosin (H&E) and luxol fast blue staining, as well as retinoic acid receptor-related orphan receptor γ (RORγ) and forkhead box protein P3 (Foxp3) immunohistochemical staining. Blood was collected for Th17 and Treg cell flow cytometry testing, and the serum levels of interleukin (IL)-17A, IL-10, transforming growth factor-β (TGF-β), IL-6, IL-23, and IL-2 were measured via enzyme-linked immunosorbent assay (ELISA). Compared with rats in the EAE group, rats in the EAE+cornuside and EAE+prednisolone groups began to recover from neurological deficits earlier, and had a greater degree of improvement of symptoms. Focal inflammation, demyelination, and RORγ-positive cell infiltration were reduced by cornuside or prednisolone treatment, whereas the Foxp3-positive cell numbers were not significantly different. Meanwhile, the number of Th17 cells and the IL-17A, IL-6, and IL-23 levels were lower in the blood after cornuside or prednisolone treatment, whereas the number of Treg cells or the levels of IL-10, TGF-β, and IL-2 were not markedly different. Cornuside can alleviate symptoms of EAE neurological deficits through its anti-inflammatory and immunosuppressive effects, and Th17 cells may be one of its therapeutic targets.
Abstract
目的
为了明确山茱萸新苷对实验性自身免疫性脑脊髓炎(EAE)的治疗作用, 以及对Th17和Treg细胞浸润中枢神经系统(CNS)的影响。
创新点
本研究提示山茱萸新苷可以改善EAE大鼠的神经功能缺损症状, 减轻EAE大鼠炎症浸润及脱髓鞘, 并抑制Th17细胞浸润。
方法
使用豚鼠脊髓匀浆乳剂皮下注射Lewis大鼠诱导EAE, 每天进行神经功能评分。待EAE大鼠开始出现神经功能缺损症状的第2天, 4组大鼠(对照组、EAE组、EAE/山茱萸新苷组和EAE/泼尼松龙组)分别接受生理盐水、生理盐水、山茱萸新苷(150 mg/kg)、泼尼松龙(5 mg/kg)治疗, 2周后停止治疗。对大鼠脊髓进行HE和LFB染色, 以及RORγ和Foxp3免疫组化染色, 并通过流式细胞检测血液中Th17和Treg细胞数量, 用酶联免疫吸附法(ELISA)检测血清中白介素17A(IL-17A)、IL-10、转化生长因子β(TGF-β)、IL-6, IL-23和IL-2水平。
结论
山茱萸新苷可以缓解EAE症状, 这可能通过抗炎抗免疫作用产生, 而Th17细胞可能是其发挥作用的靶标之一。因此, 山茱萸新苷存在治疗多发性硬化(MS)的潜在可能性。
Similar content being viewed by others
References
Balasa R, Barcutean L, Balasa A, et al., 2020. The action of Th17 cells on blood brain barrier in multiple sclerosis and experimental autoimmune encephalomyelitis. Hum Immunol, 81(5):237–243. https://doi.org/10.1016/j.humimm.2020.02.009
Burrows DJ, McGown A, Jain SA, et al., 2019. Animal models of multiple sclerosis: from rodents to zebrafish. Mult Scler J, 25(3):306–324. https://doi.org/10.1177/1352458518805246
Cerboni S, Gehrmann U, Preite S, et al., 2020. Cytokine-regulated Th17 plasticity in human health and diseases. Immunology, 163(1):13280. https://doi.org/10.1111/imm.13280
Ding HY, Xie YN, Dong Q, et al., 2019. Roles of hyaluronan in cardiovascular and nervous system disorders. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 20(5):428–436. https://doi.org/10.1631/jzus.B1900155
Dobson R, Giovannoni G, 2019. Multiple sclerosis—a review. Eur J Neurol, 26(1):27–40. https://doi.org/10.1111/ene.13819
Dong Y, Feng ZL, Chen HB, et al., 2018. Corni Fructus: a review of chemical constituents and pharmacological activities. Chin Med, 13:34. https://doi.org/10.1186/s13020-018-0191-z
Durant L, Watford WT, Ramos HL, et al., 2010. Diverse targets of the transcription factor STAT3 contribute to T cell pathogenicity and homeostasis. Immunity, 32(5):605–615. https://doi.org/10.1016/j.immuni.2010.05.003
Fan YP, Wu W, 2014. Syndrome factors of multiple sclerosis in 500 patients. J Beijing Univ Tradit Chin Med, 37(1): 68–72 (in Chinese). https://doi.org/10.3969/j.issn.1006-2157.2014.01.015
Fan YP, Wang SQ, 2018. Standard for clinical diagnosis and treatment of traditional chinese medicine for multiple sclerosis/neuromyelitis optica. J Cap Med Univ, 39(6): 833–835 (in Chinese). https://doi.org/10.3969/j.issn.1006-7795.2018.06.008
Fasching P, Stradner M, Graninger W, et al., 2017. Therapeutic potential of targeting the Th17/Treg axis in autoimmune disorders. Molecules, 22(1):134. https://doi.org/10.3390/molecules22010134
Hauser SL, Cree BAC, 2020. Treatment of multiple sclerosis: a review. Am J Med, 133(12):1380–1390.E2. https://doi.org/10.1016/j.amjmed.2020.05.049
Huang J, Zhang YW, Dong L, et al., 2018. Ethnopharmacology, phytochemistry, and pharmacology of Cornus officinalis sieb. et Zucc. J Ethnopharmacol, 213:280–301. https://doi.org/10.1016/j.jep.2017.11.010
Kleinewietfeld M, Hafler DA, 2014. Regulatory T cells in autoimmune neuroinflammation. Immunol Rev, 259(1):231–244. https://doi.org/10.1111/imr.12169
Kunkl M, Frascolla S, Amormino C, et al., 2020. T helper cells: the modulators of inflammation in multiple sclerosis. Cells, 9(2):482. https://doi.org/10.3390/cells9020482
Lassmann H, Bradl M, 2017. Multiple sclerosis: experimental models and reality. Acta Neuropathol, 133(2):223–244. https://doi.org/10.1007/s00401-016-1631-4
Lee GR, 2018. The balance of Th17 versus Treg cells in auto-immunity. Int J Mol Sci, 19(3):730. https://doi.org/10.3390/ijms19030730
Li HH, Hu FL, Zhang YL, et al., 2020. Comparative efficacy and acceptability of disease-modifying therapies in patients with relapsing-remitting multiple sclerosis: a systematic review and network meta-analysis. J Neurol, 267(12):3489–3498. https://doi.org/10.1007/s00415-019-09395-w
Li ZF, Nie LL, Chen LP, et al., 2019. Rapamycin relieves inflammation of experimental autoimmune encephalomyelitis by altering the balance of Treg/Th17 in a mouse model. Neurosci Lett, 705:39–45. https://doi.org/10.1016/j.neulet.2019.04.035
McCall B, 2019. Alemtuzumab to be restricted pending review, says EMA. Lancet, 393(10182):1683. https://doi.org/10.1016/S0140-6736(19)30935-3
McCool R, Wilson K, Arber M, et al., 2019. Systematic review and network meta-analysis comparing ocrelizumab with other treatments for relapsing multiple sclerosis. Mult Scler Relat Disord, 29:55–61. https://doi.org/10.1016/j.msard.2018.12.040
Monaco S, Nicholas R, Reynolds R, et al., 2020. Intrathecal inflammation in progressive multiple sclerosis. Int J Mol Sci, 21(21):8217. https://doi.org/10.3390/ijms21218217
Moser T, Akgün K, Proschmann U, et al., 2020. The role of Th17 cells in multiple sclerosis: therapeutic implications. Autoimmun Rev, 19(10):102647. https://doi.org/10.1016/j.autrev.2020.102647
Oh J, Vidal-Jordana A, Montalban X, 2018. Multiple sclerosis: clinical aspects. Curr Opin Neurol, 31(6):752–759. https://doi.org/10.1097/WCO.0000000000000622
Park JS, Lee J, Lim MA, et al., 2014. JAK2-STAT3 blockade by AG490 suppresses autoimmune arthritis in mice via reciprocal regulation of regulatory T cells and Th17 cells. J Immunol, 192(9):4417–4424. https://doi.org/10.4049/jimmunol.1300514
Pawlak M, Ho AW, Kuchroo VK, 2020. Cytokines and transcription factors in the differentiation of CD4+ T helper cell subsets and induction of tissue inflammation and autoimmunity. Curr Opin Immunol, 67:57–67. https://doi.org/10.1016/j.coi.2020.09.001
Pegoretti V, Swanson KA, Bethea JR, et al., 2020. Inflammation and oxidative stress in multiple sclerosis: consequences for therapy development. Oxid Med Cell Longev, 2020:7191080. https://doi.org/10.1155/2020/7191080
Pitarokoili K, Ambrosius B, Gold R, 2017. Lewis rat model of experimental autoimmune encephalomyelitis. Curr Protoc Neurosci, 81:9.61.1–9.61.20. https://doi.org/10.1002/cpns.36
Pulido-Valdeolivas I, Andorrà M, Gómez-Andrés D, et al., 2020. Retinal and brain damage during multiple sclerosis course: inflammatory activity is a key factor in the first 5 years. Sci Rep, 10:13333. https://doi.org/10.1038/s41598-020-70255-z
Qu Z, Zheng N, Zhang YF, et al., 2016. Preventing the BDNF and NGF loss involved in the effects of cornel iridoid glycoside on attenuation of experimental autoimmune encephalomyelitis in mice. Neurol Res, 38(9):831–837. https://doi.org/10.1080/01616412.2016.1200766
Qu Z, Zheng N, Wei YZ, et al., 2019. Effect of cornel iridoid glycoside on microglia activation through suppression of the JAK/STAT signalling pathway. J Neuroimmunol, 330: 96–107. https://doi.org/10.1016/j.jneuroim.2019.01.014
Ruiz F, Vigne S, Pot C, 2019. Resolution of inflammation during multiple sclerosis. Semin Immunopathol, 41(6):711–726. https://doi.org/10.1007/s00281-019-00765-0
Schneider C, Schuetz G, Zollner TM, 2009. Acute neuroinflammation in lewis rats—a model for acute multiple sclerosis relapses. J Neuroimmunol, 213(1–2):84–90. https://doi.org/10.1016/j.jneuroim.2009.05.015
Segal BM, 2019. The diversity of encephalitogenic CD4+ T cells in multiple sclerosis and its animal models. J Clin Med, 8(1):120. https://doi.org/10.3390/jcm8010120
Stenager E, 2019. A global perspective on the burden of multiple sclerosis. Lancet Neurol, 18(3):227–228. https://doi.org/10.1016/S1474-4422(18)30498-8
Xie XJ, Ye YF, Zhou L, et al., 2010. Th17 promotes acute rejection following liver transplantation in rats. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 11(11):819–827. https://doi.org/10.1631/jzus.B1000030
Yin LL, Chen YY, Qu Z, et al., 2014. Involvement of JAK/STAT signaling in the effect of cornel iridoid glycoside on experimental autoimmune encephalomyelitis amelioration in rats. J Neuroimmunol, 274(1–2):28–37. https://doi.org/10.1016/j.jneuroim.2014.06.022
Zhao PY, Wang YQ, Liu XH, et al., 2018. Bu Shen Yi Sui capsule promotes remyelination correlating with Sema3A/NRP-1, LIF/LIFR and Nkx6.2 in mice with experimental autoimmune encephalomyelitis. J Ethnopharmacol, 217:36–48. https://doi.org/10.1016/j.jep.2018.02.014
Zhao ST, Wang CZ, 2018. Regulatory T cells and asthma. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 19(9):663–673. https://doi.org/10.1631/jzus.B1700346
Acknowledgments
This work was supported by the Traditional Chinese Medical Science and Technology Project of Zhejiang Province (No. 2019ZA063) and the Scientific Research Fund of Zhejiang Chinese Medical University (No. 2019ZY09), China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Rongbo ZHANG and Qiang YUAN designed the study. Jin LIU and Bin XU established the animal models. Rongbo ZHANG and Jin LIU performed the experimental research, and wrote and edited the manuscript. You WU and Shunli LIANG contributed to the data analysis. All authors have read and approved the final manuscript and, therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Rongbo ZHANG, Jin LIU, Bin XU, You WU, Shunli LIANG, and Qiang YUAN declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
Rights and permissions
About this article
Cite this article
Zhang, R., Liu, J., Xu, B. et al. Cornuside alleviates experimental autoimmune encephalomyelitis by inhibiting Th17 cell infiltration into the central nervous system. J. Zhejiang Univ. Sci. B 22, 421–430 (2021). https://doi.org/10.1631/jzus.B2000771
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000771