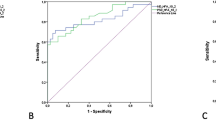
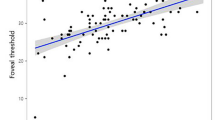
Abstract
To investigate associations between central visual function and inner retinal structure in primary open-angle glaucoma (POAG). This study enrolled 78 POAG patients and 58 healthy controls. POAG was classified into early glaucoma and moderate to advanced glaucoma. The following tests were performed on all participants: isolated-check visual evoked potential (icVEP) testing, 24–2 standard automated perimetry (SAP), and Cirrus optical coherence tomography (OCT) examinations. Signal-to-noise ratio (SNR) measures obtained from icVEP responses to isolated checks presented at four depths of modulation (DOMs; 8%, 14%, 22%, and 32%) were explored. Mean macular sensitivity (mMS) was assessed by calculating the mean sensitivities of central 12 SAP points. Ganglion cell layer+ inner plexiform layer thickness (GCL+IPLT) and peripapillary retinal nerve fiber layer thickness (pRNFLT) were measured by OCT scanning. For each group of subjects, linear relationships among the following measures were analyzed: SNR, mMS, GCL+IPLT, and pRNFLT. SNR, mMS, GCL+IPLT, and pRNFLT were all more significantly decreased in glaucoma than in controls (P<0.001). A significant positive association was found between SNR at 14% DOM and GCL+IPLT at the inferior sector in early glaucoma (r=0.465, P=0.004). In moderate to advanced glaucoma, significant correlations were found between SNR at 32% DOM and mean GCL+IPLT (r=0.364, P=0.023), superior GCL+IPLT (r=0.358, P=0.025), and mean pRNFLT (r=0.396, P=0.025). In addition, in moderate to advanced glaucoma, there were significant correlations between mMS and all relevant measures of retinal thickness (r=0.330−0.663, P<0.010). In early glaucoma, significant correlations were found between mean mMS and minimum GCL+IPLT (r=0.373, P=0.023), and between inferior mMS and superior GCL+IPLT (r=0.470, P=0.003). Linear models provided a good explanation for the relationship between SNR and inner retinal thickness (IRT), whereas nonlinear models better explained the relationship between mMS and IRT. In early glaucoma, both SNR and mMS were related moderately and significantly to IRT, whereas in moderate to advanced glaucoma, mMS was more strongly correlated with IRT than SNR.
摘要
目 的
探讨原发性开角型青光眼 (POAG) 患者黄斑区视网膜内层结构和功能的关系.
创新点
POAG 的潜在原因是视网膜神经节细胞 (RGC) 的丢失. 虽然在传统上我们可以通过测量视网膜后极部约 30° 的结构和功能来评估青光眼性视神经损害, 但是 50% 的 RGC 存在于黄斑区 4.5 mm 范围内. 本研究关注黄斑区约 10°范围视网膜结构和功能的关系, 有助于更早地监测到青光眼性视神经损害.
方 法
本研究纳入了 78 例 POAG 患者及 58 例健康对照者, 其中 POAG 分为早期青光眼 (EG) 和中晚期青光眼 (AG). 所有受试者均进行了以下检测: 分离格栅视觉诱发电位 (icVEP)、 标准自动视野计 (SAP) 及光学相干断层扫描 (OCT). icVEP 检测时给予 8%、14%、22% 及 32% 对比度刺激, 采集相应的信躁比 (SNR). 黄斑区视野敏感度 (mMS) 通过计算黄斑中心 12 点的敏感度平均值获得. OCT 扫描包括视网膜神经节细胞层+内丛状层的平均厚度 (GCL+IPLT) 及视盘周围神经纤维层平均厚度 (pRNFLT). 我们比较了各组间 SNR、 mMS、 GCL+IPLT 及 pRNFLT 值, 并分析了结构性指标和功能性指标之间的相关性.
结 论
POAG 组患者的 SNR、 mMS、GCL+IPLT 及 pRNFLT 均较正常对照组显著下降 (所有 P 值 <0.001). 在早期青光眼中, SNR 及 mMS 均与视网膜内层厚度呈中度相关; 而在中晚期青光眼中, mMS 与视网膜内层厚度呈高度相关.
Similar content being viewed by others
References
Bartz-Schmidt KU, Thumann G, Jonescu-Cuypers CP, et al., 1999. Quantitative morphologic and functional evaluation of the optic nerve head in chronic open-angle glaucoma. Surv Ophthalmol, 44(Suppl 1):S41–S53. https://doi.org/10.1016/S0039-6257(99)00076-4
Budenz DL, Rhee P, Feuer WJ, et al., 2002. Comparison of glaucomatous visual field defects using standard full threshold and Swedish interactive threshold algorithms. Arch Ophthalmol, 120(9):1136–1141. https://doi.org/10.1016/j.clinbiochem.2005.11.019
Curcio CA, Allen KA, 1990. Topography of ganglion cells in human retina. J Comp Neurol, 300(1):5–25. https://doi.org/10.1002/cne.903000103
de Moraes CG, Sun A, Jarukasetphon R, et al., 2019. Association of macular visual field measurements with glaucoma staging systems. JAMA Ophthalmol, 137(2):139–145. https://doi.org/10.1001/jamaophthalmol.2018.5398
Garway-Heath DF, Caprioli J, Fitzke FW, et al., 2000. Scaling the hill of vision: the physiological relationship between light sensitivity and ganglion cell numbers. Invest Ophthalmol Vis Sci, 41(7):1774–1782.
Glovinsky Y, Quigley HA, Dunkelberger GR, 1991. Retinal ganglion cell loss is size dependent in experimental glaucoma. Invest Ophthalmol Vis Sci, 32(3):484–491.
Glovinsky Y, Quigley HA, Pease ME, 1993. Foveal ganglion cell loss is size dependent in experimental glaucoma. Invest Ophthalmol Vis Sci, 34(2):395–400.
Greenstein VC, Seliger S, Zemon V, et al., 1998. Visual evoked potential assessment of the effects of glaucoma on visual subsystems. Vision Res, 38(12):1901–1911. https://doi.org/10.1016/S0042-6989(97)00348-9
Gutowitz H, Zemon V, Victor J, et al., 1986. Source geometry and dynamics of the visual evoked potential. Electroencephalogr Clin Neurophysiol, 64(4):308–327. https://doi.org/10.1016/0013-4694(86)90155-0
Harwerth RS, Carter-Dawson L, Shen F, et al., 1999. Ganglion cell losses underlying visual field defects from experimental glaucoma. Invest Ophthalmol Vis Sci, 40(10):2242–2250.
Harwerth RS, Vilupuru AS, Rangaswamy NV, et al., 2007. The relationship between nerve fiber layer and perimetry measurements. Invest Ophthalmol Vis Sci, 48(2):763–773. https://doi.org/10.1167/iovs.06-0688
Harwerth RS, Wheat JL, Fredette MJ, et al., 2010. Linking structure and function in glaucoma. Prog Retin Eye Res, 29(4):249–271. https://doi.org/10.1016/j.preteyeres.2010.02.001
Hood DC, Anderson SC, Wall M, et al., 2007. Structure versus function in glaucoma: an application of a linear model. Invest Ophthalmol Vis Sci, 48(8):3662–3668. https://doi.org/10.1167/iovs.06-1401
Hood DC, Raza AS, de Moraes CGV, et al., 2013. Glaucomatous damage of the macula. Prog Retin Eye Res, 1: 1–21. https://doi.org/10.1016/j.preteyeres.2012.08.003
Kerrigan-Baumrind LA, Quigley HA, Pease ME, et al., 2000. Number of ganglion cells in glaucoma eyes compared with threshold visual field tests in the same persons. Invest Ophthalmol Vis Sci, 41(3):741–748.
Kim KE, Park KH, Jeoung JW, et al., 2014. Severity-dependent association between ganglion cell inner plexiform layer thickness and macular mean sensitivity in open-angle glaucoma. Acta Ophthalmol, 92(8):e650–e656. https://doi.org/10.1111/aos.12438
Mwanza JC, Oakley JD, Budenz DL, et al., 2011. Macular ganglion cell-inner plexiform layer: automated detection and thickness reproducibility with spectral domain-optical coherence tomography in glaucoma. Invest Ophthalmol Vis Sci, 52(11):8323–8329. https://doi.org/10.1167/iovs.11-7962
Nouri-Mahdavi K, Nowroozizadeh S, Nassiri N, et al., 2013. Macular ganglion cell/inner plexiform layer measurements by spectral domain optical coherence tomography for detection of early glaucoma and comparison to retinal nerve fiber layer measurements. Am J Ophthalmol, 156(6): 1297–1307.e2. https://doi.org/10.1016/j.ajo.2013.08.001
Quigley HA, 1999. Neuronal death in glaucoma. Prog Retin Eye Res, 18(1):39–57. https://doi.org/10.1016/S1350-9462(98)00014-7
Quigley HA, Dunkelberger GR, Green WR, 1989. Retinal ganglion cell atrophy correlated with automated perimetry in human eyes with glaucoma. Am J Ophthalmol, 107(5): 453–464. https://doi.org/10.1016/0002-9394(89)90488-1
van Buskirk EM, Cioffi GA, 1992. Glaucomatous optic neuropathy. Am J Ophthalmol, 113(4):447–452. https://doi.org/10.1016/s0002-9394(14)76171-9
Xu LJ, Zhang L, Li SL, et al., 2017. Accuracy of isolated-check visual evoked potential technique for diagnosing primary open-angle glaucoma. Doc Ophthalmol, 135(2): 107–119. https://doi.org/10.1007/s10633-017-9598-6
Yanashima K, 1982. Surface distribution of steady-state cortical potentials evoked by visual half-field stimulation. Graefes Arch Clin Exp Ophthalmol, 218(3):118–123. https://doi.org/10.1007/BF02215648
Zemon V, Eisner W, Gordon J, et al., 1995. Contrast-dependent responses in the human visual system: childhood through adulthood. Int J Neurosci, 80(1–4):181–201. https://doi.org/10.3109/00207459508986100
Zemon V, Tsai JC, Forbes M, et al., 2008. Novel electro-physiological instrument for rapid and objective assessment of magnocellular deficits associated with glaucoma. Doc Ophthalmol, 117(3):233–243. https://doi.org/10.1007/s10633-008-9129-6
Author information
Authors and Affiliations
Contributions
Yuan-bo LIANG participated in the conception of the study, analysis of data, revision and final approval of the manuscript. Li-juan XU participated in the design of study, acquisition and analysis of data, drafting of the manuscript and revision, and the final approval of the version. Sha-ling LI participated in the acquisition and analysis of data. Vance ZEMON performed the analysis of data and revision of manuscript. Yan-qian XIE performed the analysis of data. All authors have read and approved the final manuscript. Therefore, the authors have full access to all the data in the study and take responsibility for the integrity and security of the data.
Corresponding author
Ethics declarations
Li-juan XU, Sha-ling LI, Yan-qian XIE, and Yuan-bo LIANG declare that they have no conflict of interest.
Vance ZEMON, as a principal in VeriSci Corp. (Raritan, NJ, USA), has received a financial interest in Huzhou Medconova Medical Technology Co., Ltd., China.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5). All participants signed the consent form approved by the Eye Hospital of Wenzhou Medical University, Wenzhou, China.
Additional information
Project supported by the Wenzhou Medical University (No. QTJ13009), the Health Innovation Talents in Zhejiang Province (2016, No. 25), and the Eye Hospital of Wenzhou Medical University (the Value of Isolated-Check Visual Evoked Potential in Glaucoma Diagnosis and Monitoring), China
Rights and permissions
About this article
Cite this article
Xu, Lj., Li, Sl., Zemon, V. et al. Central visual function and inner retinal structure in primary open-angle glaucoma. J. Zhejiang Univ. Sci. B 21, 305–314 (2020). https://doi.org/10.1631/jzus.B1900506
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1900506
Key words
- Isolated-check visual evoked potential (icVEP)
- Primary open-angle glaucoma (POAG)
- Optical coherence tomography (OCT)
- Standard automated perimetry (SAP)