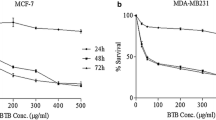
Abstract
In order to study the molecular mechanisms of green tea polyphenols (GTPs) in treatment or prevention of breast cancer, the cytotoxic effects of GTPs on five human cell lines (MCF-7, A549, Hela, PC3, and HepG2 cells) were determined and the antitumor mechanisms of GTPs in MCF-7 cells were analyzed. The results showed that GTPs exhibited a broad spectrum of inhibition against the detected cancer cell lines, particularly the MCF-7 cells. Studies on the mechanisms revealed that the main modes of cell death induced by GTPs were cell cycle arrest and mitochondrialmediated apoptosis. Flow cytometric analysis showed that GTPs mediated cell cycle arrest at both G1/M and G2/M transitions. GTP dose dependently led to apoptosis of MCF-7 cells via the mitochondrial pathways, as evidenced by induction of chromatin condensation, reduction of mitochondrial membrane potential (ΔΨm), improvement in the generation of reactive oxygen species (ROS), induction of DNA fragmentation, and activations of caspase-3 and caspase-9 in the present paper.
摘要
目的
评估茶多酚对人乳腺癌细胞MCF-7 凋亡的影响, 并探讨了其作用机制。
创新点
全面考察了茶多酚对抗乳腺癌的分子机制,为茶 多酚作为抗肿瘤辅助药物提供理论依据。
方法
首先选取不同组织来源的五种人肿瘤细胞(人肝 癌细胞HepG2、人肺癌细胞A549、人前列腺癌 细胞PC3、人宫颈癌细胞Hela、人乳腺癌细胞 MCF-7)作为体外模型,以MTT 法检测茶多酚 对其增殖抑制作用。然后,选用最敏感细胞 MCF-7 为研究对象,采用流式细胞术检测茶多酚 对细胞周期分布的影响,用Hoechst 3328 染色法 观察茶多酚对细胞核形态的影响,用JC-1 染色法 观察茶多酚对细胞线粒体跨膜电位的影响,用双 氯荧光素(DCFH-DA)染色法观察茶多酚对细 胞活性氧(ROS)水平的影响,用凝胶电泳DNA 片段测定法(DNA ladder)观察茶多酚处理后细 胞DNA 断裂情况,用蛋白质印迹法(Western blot)检测茶多酚对细胞凋亡关键蛋白caspase-3 和caspase-9 表达的影响,全面探讨了茶多酚体外 抗肿瘤机制。
结论
实验结果显示,茶多酚能够通过诱导细胞周期阻 滞和线粒体凋亡抑制MCF-7 细胞增殖。茶多酚诱 导线粒体凋亡的途径是使线粒体跨膜电位下降, 促使MCF-7 细胞内ROS 生成,促使细胞DNA 断裂和促进细胞内caspase-3 和caspase-9 的活化。
Similar content being viewed by others
References
Baregamian, N., Song, J., Bailey, C.E., et al., 2009. Tumor necrosis factor-α and apoptosis signal-regulating kinase 1 control reactive oxygen species release, mitochondrial autophagy and c-Jun N-terminal kinase/p38 phosphorylation during necrotizing enterocolitis. Oxid. Med. Cell. Longev., 2(5):297–306. http://dx.doi.org/10.4161/oxim.2.5.9541
Boyle, P., Levin, B., 2008. World Cancer Report 2008. International Agency for Research on Cancer, Lyon, France. British Standards Institutio., 2005. BS ISO 14502-1:2005. Determination of substances characteristic of green and black tea. Content of total polyphenols in tea. Colorimetric method using Folin-Ciocalteu reagent. British Standards Institution, London, UK, p.1–6. http://shop.bsigroup.com/ProductDetail/?pid=000000000030153463
Chakraborty, D., Bishayee, K., Ghosh, S., et al., 2012. [6]-Gingerol induces caspase 3 dependent apoptosis and autophagy in cancer cells: drug-DNA interaction and expression of certain signal genes in HeLa cells. Eur. J. Pharmacol., 694(1-3):20–29. http://dx.doi.org/10.1016/j.ejphar.2012.08.001
Cotran, R.S., Kumar, V., Collins, T., et al., 1999. Robbins Pathologic Basis of Disease, 6th Ed. WB Saunders, Philadelphia.
Crews, C.M., Mohan, R., 2000. Small-molecule inhibitors of the cell cycle. Curr. Opin. Chem. Biol., 4(1):47–53. http://dx.doi.org/10.1016/S1367-5931(99)00050-2
Ferlay, J., Shin, H., Bray, F., et al., 2010. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer, 127(12):2893–2917. http://dx.doi.org/10.1002/ijc.25516
González, S.E.F., Anguiano, E.A., Herrera, A.M., et al., 2013. Cytotoxic, pro-apoptotic, pro-oxidant, and non-genotoxic activities of a novel copper(II) complex against human cervical cancer. Toxicology, 314(1):155–165. http://dx.doi.org/10.1016/j.tox.2013.08.018
Hail, N.Jr., Carter, B.Z., Konopleva, M., 2006. Apoptosis effector mechanisms: a requiem performed in different keys. Apoptosis, 11(6):889–904. http://dx.doi.org/10.1007/s10495-006-6712-8
Hail, N.Jr., Cortes, M., Drake, E.N., et al., 2008. Cancer chemoprevention: a radical perspective. Free Radical Biol. Med., 45(2):97–110. http://dx.doi.org/10.1016/j.freeradbiomed.2008.04.004
Herold, M.J., Kuss, A.W., Kraus, C., et al., 2002. Mitochondriadependent caspase-9 activation is necessary for antigen receptor-mediated effector caspase activation and apoptosis in WEHI 231 lymphoma cells. J. Immunol., 168(8): 3902–3909. http://dx.doi.org/10.4049/jimmunol.168.8.3902
Hortobagyi, G.N., Ames, F.C., Buzdar, A.U., et al., 1988. Management of stage III primary breast cancer with primary chemotherapy, surgery, and radiation therapy. Cancer, 62(12):2507–2516. http://dx.doi.org/10.1002/1097-0142(19881215)62:12<2507::AID-CNCR2820621210>3.0.CO;2-D
Huang, X.C., Wang, M., Wang, H.S., et al., 2014. Synthesis and antitumor activities of novel dipeptide derivatives derived from dehydroabietic acid. Bioorg. Med. Chem. Lett., 24(6):1511–1518. http://dx.doi.org/10.1016/j.bmcl.2014.02.001
Jiang, X.G., 2014. Harnessing the immune system for the treatment of breast cancer. J. Zhejiang Univ.-Sci. B (Biomed. & Biotechnol.), 15(1):1–15. http://dx.doi.org/10.1631/jzus.B1300264
Khan, N., Mukhtar, H., 2007. Tea polyphenols for health promotion. Life Sci., 81(7):519–533. http://dx.doi.org/10.1016/j.lfs.2007.06.011
Li, L.L., Cao, W.Q., Zheng, W.J., et al., 2012. Ruthenium complexes containing 2,6-bis(benzimidazolyl)pyridine derivatives induce cancer cell apoptosis by triggering DNA damage-mediated p53 phosphorylation. Dalton Trans., 41(41):12766–12772. http://dx.doi.org/10.1039/C2DT30665D
Li, T., Zhu, J., Guo, L., et al., 2013. Differential effects of polyphenols-enriched extracts from hawthorn fruit peels and fleshes on cell cycle and apoptosis in human MCF-7 breast carcinoma cells. Food Chem., 141(2):1008–1018. http://dx.doi.org/10.1016/j.foodchem.2013.04.050
Liang, H.H., Huang, H.H., Kwok, K.C., 1999. Properties of tea-polyphenol-complexed bromelain. Food Res. Int., 32(8):545–551. http://dx.doi.org/10.1016/S0963-9969(99)00129-5
Liu, H.X., Li, X.L., Dong, C.F., 2015. Epigenetic and metabolic regulation of breast cancer stem cells. J. Zhejiang Univ.-Sci. B (Biomed. & Biotechnol.), 16(1):10–17. http://dx.doi.org/10.1631/jzus.B1400172
Liu, S.M., Huang, H.H., 2015. Assessments of antioxidant effect of black tea extract and its rationals by erythrocyte haemolysis assay, plasma oxidation assay and cellular antioxidant activity (CAA) assay. J. Funct. Foods, 18(Part B):1095–1105. http://dx.doi.org/10.1016/j.jff.2014.08.023
Lu, G., Xiao, H., You, H., et al., 2008. Synergistic inhibition of lung tumorigenesis by a combination of green tea polyphenols and atorvastatin. Clin. Cancer Res., 14(15): 4981–4988. http://dx.doi.org/10.1158/1078-0432.CCR-07-1860
Luo, H.T., Tang, L.L., Tang, M., et al., 2006. Phase IIa chemoprevention trial of green tea polyphenols in high-risk individuals of liver cancer: modulation of urinary excretion of green tea polyphenols and 8-hydroxydeoxyguanosine. Carcinogenesis, 27(2):262–268. http://dx.doi.org/10.1093/carcin/bgi147
Pan, G.H., O'Rourke, K., Dixit, V.M., 1998. Caspase-9, Bcl-XL, and Apaf-1 form a ternary complex. J. Biol. Chem., 273(10):5841–5845. http://dx.doi.org/10.1074/jbc.273.10.5841
Pandey, M., Shukla, S., Gupta, S., 2010. Promoter demethylation and chromatin remodeling by green tea polyphenols leads to re-expression of GSTP1 in human prostate cancer cells. Int. J. Cancer, 126(11):2520–2533. http://dx.doi.org/10.1002/ijc.24988
Pianetti, S., Guo, S., Kavanagh, K.T., et al., 2002. Green tea polyphenol epigallocatechin-3 gallate inhibits Her-2/neu signaling, proliferation, and transformed phenotype of breast cancer cells. Cancer Res., 62(3):652–655.
Quan, Z.W., Gu, J., Dong, P., et al., 2010. Reactive oxygen species-mediated endoplasmic reticulum stress and mitochondrial dysfunction contribute to cirsimaritin-induced apoptosis in human gallbladder carcinoma GBC-SD cells. Cancer Lett., 295(2):252–259. http://dx.doi.org/10.1016/j.canlet.2010.03.008
Radad, K., Rausch, W.D., Gille, G., 2006. Rotenone induces cell death in primary dopaminergic culture by increasing ROS production and inhibiting mitochondrial respiration. Neurochem. Int., 49(4):379–386. http://dx.doi.org/10.1016/j.neuint.2006.02.003
Rezaei, P.F., Fouladdel, S., Hassani, S., et al., 2012. Induction of apoptosis and cell cycle arrest by pericarp polyphenolrich extract of Baneh in human colon carcinoma HT29 cells. Food Chem. Toxicol., 50(3-4):1054–1059. http://dx.doi.org/10.1016/j.fct.2011.11.012
Rogalska, A., Koceva-Chyla, A., Józwiak, Z., 2008. Aclarubicininduced ROS generation and collapse of mitochondrial membrane potential in human cancer cell lines. Chem. Biol. Interact., 176(1):58–70. http://dx.doi.org/10.1016/j.cbi.2008.07.002
Stewart, B.W., Wild, C.P., 2014. World Cancer Report 2014. World Health Organization, International Agency for Research on Cancer, Lyon, France.
Strasser, A., O'Connor, L., Dixit, V.M., 2000. Apoptosis signaling. Annu. Rev. Biochem., 69:217–245. http://dx.doi.org/10.1146/annurev.biochem.69.1.217
Sun, X.M., MacFarlane, M., Zhuang, J., et al., 1999. Distinct caspase cascades are initiated in receptor-mediated and chemical-induced apoptosis. J. Biol. Chem., 274(8):5053–5060. http://dx.doi.org/10.1074/jbc.274.8.5053
Thangapazham, R.L., Singh, A.K., Sharma, A., et al., 2007. Green tea polyphenols and its constituent epigallocatechin gallate inhibits proliferation of human breast cancer cells in vitro and in vivo. Cancer Lett., 245(1-2):232–241. http://dx.doi.org/10.1016/j.canlet.2006.01.027
Yang, C.S., Lambert, J.D., Sang, S., 2009. Antioxidative and anti-carcinogenic activities of tea polyphenols. Arch. Toxicol., 83(1):11–21. http://dx.doi.org/10.1007/s00204-008-0372-0
Zhang, J.Y., Wu, H.Y., Xia, X.K., et al., 2007. Anthracenedione derivative 1403P-3 induces apoptosis in KBand KBv200 cells via reactive oxygen species-independent mitochondrial pathway and death receptor pathway. Cancer Biol. Ther., 6(9):1409–1417. http://dx.doi.org/10.4161/cbt.6.9.4543
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Research Fund for the Doctoral Program of Higher Education of China (No. 20120172110017) and the National Natural Science Foundation of China (Nos. 31471673 and 31271978)
ORCID: Shu-min LIU, http://orcid.org/0000-0001-7053-5551
Rights and permissions
About this article
Cite this article
Liu, Sm., Ou, Sy. & Huang, Hh. Green tea polyphenols induce cell death in breast cancer MCF-7 cells through induction of cell cycle arrest and mitochondrial-mediated apoptosis. J. Zhejiang Univ. Sci. B 18, 89–98 (2017). https://doi.org/10.1631/jzus.B1600022
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1600022
Key words
- Green tea polyphenol (GTP)
- Breast cancer
- MCF-7 cells
- Mitochondrial-mediated apoptosis
- Cell death
- Cell cycle arrest