Abstract
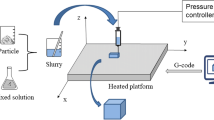
Tissue-engineered scaffolds are widely researched for their potential to replace damaged or diseased bone. Similarly, this study reports 3D printing of multi-walled carbon nanotubes (MWCNTS) reinforced calcium silicate (2 CaO⋅SiO2) bone scaffold structures using Robocasting. The scaffold structures were 3D printed with 0.5, 1, and 2 wt% of MWCNTS in 2 CaO⋅SiO2 and carboxymethyl cellulose (CMC) solution as the binder. The printed structures were sintered at 1000 °C for 1 h in a vacuum to ensure the retention of MWCNTS. The addition of 2 wt% of MWCNTS in Calcium silicate structures exhibited an increase in compressive strength (18.57 ± 0.349 MPa) compared to pure Calcium silicate scaffold structures. In addition, the developed scaffolds were found to be hydrophilic, cytocompatible, enhance cell proliferation, and stable for 28 days under in vitro conditions.
Graphical abstract








Similar content being viewed by others
Data availability
All data acquired during experimentation are disclosed and discussed in the manuscript.
References
R. Dimitriou, E. Jones, D. McGonagle, P.V. Giannoudis, Bone regeneration: current concepts and future directions. BMC Med. 9, 1–10 (2011). https://doi.org/10.1186/1741-7015-9-66
W. Wang, K.W.K. Yeung, Bone grafts and biomaterials substitutes for bone defect repair: a review. Bioact. Mater. 2, 224–247 (2017). https://doi.org/10.1016/j.bioactmat.2017.05.007
R. Manonmani, T.M. Sridhar, Sintering temperature effects on nano triphasic bioceramic composite coated 316L SS for corrosion resistance, adhesion strength, and cell proliferation on implants. J. Mater. Res. 35, 580–590 (2020). https://doi.org/10.1557/jmr.2020.22
H. Qu, H. Fu, Z. Han, Y. Sun, Biomaterials for bone tissue engineering scaffolds: a review. RSC Adv. 9, 26252–26262 (2019). https://doi.org/10.1039/c9ra05214c
J.A. Ramírez, V. Ospina, A.A. Rozo et al., Influence of geometry on cell proliferation of PLA and alumina scaffolds constructed by additive manufacturing. J. Mater. Res. 34, 3757–3765 (2019). https://doi.org/10.1557/jmr.2019.323
M.N. Collins, G. Ren, K. Young et al., Scaffold fabrication technologies and structure/function properties in bone tissue engineering. Adv. Funct. Mater. 31, 2010609 (2021). https://doi.org/10.1002/adfm.202010609
K.K. Moncal, D.N. Heo, K.P. Godzik et al., 3D printing of poly(ε-caprolactone)/poly(D, L-lactide-co-glycolide)/hydroxyapatite composite constructs for bone tissue engineering. J. Mater. Res. 33, 1972–1986 (2018). https://doi.org/10.1557/jmr.2018.111
A.M. Deliormanll, A.H. Deliormanll, Finite element method simulation for the prediction of mechanical properties of three-dimensional periodic bioactive glass scaffolds. J. Aust. Ceram. Soc. 53, 299–307 (2017). https://doi.org/10.1007/s41779-017-0037-7
S. Lamnini, F. Baino, G. Montalbano et al., Printability of carboxymethyl cellulose/glass-containing inks for robocasting deposition in reversible solid oxide cell applications. Mater. Lett. 318, 132239 (2022). https://doi.org/10.1016/j.matlet.2022.132239
L. Rueschhoff, W. Costakis, M. Michie et al., Additive manufacturing of dense ceramic parts via direct ink writing of aqueous alumina suspensions. Int. J. Appl. Ceram. Technol. 13, 821–830 (2016). https://doi.org/10.1111/ijac.12557
T. Cebe, N. Ahuja, F. Monte et al., Novel 3D-printed methacrylated chitosan-laponite nanosilicate composite scaffolds enhance cell growth and biomineral formation in MC3T3 pre-osteoblasts. J. Mater. Res. 35, 58–75 (2020). https://doi.org/10.1557/jmr.2018.260
A. Paterlini, S. Le Grill, F. Brouillet et al., Robocasting of self-setting bioceramics: from paste formulation to 3D part characteristics. Open Ceram. 5, 100070 (2021). https://doi.org/10.1016/j.oceram.2021.100070
R.L. Walton, E.R. Kupp, G.L. Messing, Additive manufacturing of textured ceramics: a review. J. Mater. Res. 36, 3591–3606 (2021). https://doi.org/10.1557/s43578-021-00283-6
K.Y. Tsai, H.Y. Lin, Y.W. Chen, C.Y. Lin, T.T. Hsu, C.T. Kao, Laser sintered magnesium-calciumsilicate/poly-ε-caprolactone scaffold for bone tissue engineering. Materials 10, 65 (2017). https://doi.org/10.3390/ma10010065
A. Sathain, P. Monvisade, P. Siriphannon, Bioactive alginate/carrageenan/calcium silicate porous scaffolds for bone tissue engineering. Mater. Today Commun. 26, 102165 (2021). https://doi.org/10.1016/j.mtcomm.2021.102165
Y. Zhang, Y. Yu, F. Dolati, I.T. Ozbolat, Effect of multiwall carbon nanotube reinforcement on coaxially extruded cellular vascular conduits. Mater. Sci. Eng. C 39, 126–133 (2014). https://doi.org/10.1016/j.msec.2014.02.036
S. Li, Y. Su, H. Jin et al., Effects of carbon nanotube content on morphology of SiCp(CNT) hybrid reinforcement and tensile mechanical properties of SiCp(CNT)/Al composites. J. Mater. Res. 32, 1239–1247 (2017). https://doi.org/10.1557/jmr.2017.12
K.P. Thomas Heinze, Studies on the synthesis and characterization of carboxymethylcellulose. Die Angew. Makromol Chem. 266, 37–45 (1999). https://doi.org/10.4156/jcit.vol8.issue6.73
A. Benchabane, K. Bekkour, Rheological properties of carboxymethyl cellulose (CMC) solutions. Colloid Polym. Sci. 286, 1173–1180 (2008). https://doi.org/10.1007/s00396-008-1882-2
A. Mahajan, A. Kingon, Á. Kukovecz et al., Studies on the thermal decomposition of multiwall carbon nanotubes under different atmospheres. Mater. Lett. 90, 165–168 (2013). https://doi.org/10.1016/j.matlet.2012.08.120
C. Li, X. Liu, J. Yi et al., Effects of sintering parameters on the microstructure and mechanical properties of carbon nanotubes reinforced aluminum matrix composites. J. Mater. Res. 31, 3757–3765 (2016). https://doi.org/10.1557/jmr.2016.436
G. Mata-Osoro, J.S. Moya, C. Pecharroman, Transparent alumina by vacuum sintering. J. Eur. Ceram. Soc. 32, 2925–2933 (2012). https://doi.org/10.1016/j.jeurceramsoc.2012.02.039
G.J. Arputhavalli, S. Agilan, P. Saravanan, Influence of sintering temperature on microstructure, magnetic properties of vacuum sintered Co (-Zn)-Ni-Al alloys. Mater. Lett. 233, 177–180 (2018). https://doi.org/10.1016/j.matlet.2018.08.152
J. Ravoor, A study on retention of MWCNT in robocasted MWCNT-HAP scaffold structures using vacuum sintering technique and their characteristics. Ceram. Int. 48, 31289–31298 (2022). https://doi.org/10.1016/j.ceramint.2022.06.204
N. Golafshan, E. Vorndran, S. Zaharievski et al., Tough magnesium phosphate-based 3D-printed implants induce bone regeneration in an equine defect model. Biomaterials 261, 120302 (2020). https://doi.org/10.1016/j.biomaterials.2020.120302
I. Denry, O.-M. Goudouri, J.A.H. Jefferey Harless, Rapid vacuum sintering: a novel technique for fabricating fluorapatite ceramic scaffolds for bone tissue engineering Isabelle. J. Biomed. Mater. Res. B 106, 291–299 (2018). https://doi.org/10.1002/jbm.b.33825.Rapid
R. Choudhary, S. Koppala, S. Swamiappan, Bioactivity studies of calcium magnesium silicate prepared from eggshell waste by sol-gel combustion synthesis. J. Asian Ceram. Soc. 3, 173–177 (2015). https://doi.org/10.1016/j.jascer.2015.01.002
B. Yu, Z. Liu, C. Ma et al., Ionic liquid modified multi-walled carbon nanotubes as lubricant additive. Tribol. Int. 81, 38–42 (2015). https://doi.org/10.1016/j.triboint.2014.07.019
R. Atchudan, A. Pandurangan, J. Joo, Effects of nanofillers on the thermo-mechanical properties and chemical resistivity of epoxy nanocomposites. J. Nanosci. Nanotechnol. 15, 4255–4267 (2015). https://doi.org/10.1166/jnn.2015.9706
Y.R. Son, S.J. Park, Green preparation and characterization of graphene oxide/carbon nanotubes-loaded carboxymethyl cellulose nanocomposites. Sci. Rep. 8, 2–11 (2018). https://doi.org/10.1038/s41598-018-35984-2
M. Tamaddon, S. Samizadeh, L. Wang et al., Intrinsic osteoinductivity of porous titanium scaffold for bone tissue engineering. Int. J. Biomater. (2017). https://doi.org/10.1155/2017/5093063
M. Rasoulianboroujeni, A. Yadegari, S. Tajik, L. Tayebi, Development of a modular reinforced bone tissue engineering scaffold with enhanced mechanical properties. Mater. Lett. 318, 132170 (2022). https://doi.org/10.1016/j.matlet.2022.132170
W. Wang, G. Caetano, W.S. Ambler et al., Enhancing the hydrophilicity and cell attachment of 3D printed PCL/graphene scaffolds for bone tissue engineering. Materials 9, 992 (2016). https://doi.org/10.3390/ma9120992
W.Y. Wang, J.Y. Shi, J.L. Wang et al., Preparation and characterization of PEG-g-MWCNTs/PSf nano-hybrid membranes with hydrophilicity and antifouling properties. RSC Adv. 5, 84746–84753 (2015). https://doi.org/10.1039/c5ra16077d
T. Werder, J.H. Walther, R.L. Jaffe et al., On the water-carbon interaction for use in molecular dynamics simulations of graphite and carbon nanotubes. J. Phys. Chem. B 107, 1345–1352 (2003). https://doi.org/10.1021/jp0268112
Z. Mohammadalizadeh, S. Karbasi, S. Arasteh, Physical, mechanical and biological evaluation of poly (3-hydroxybutyrate)-chitosan/MWNTs as a novel electrospun scaffold for cartilage tissue engineering applications. Polym. Technol. Mater. 59, 417–429 (2020). https://doi.org/10.1080/25740881.2019.1647244
R. Eivazzadeh-Keihan, A. Maleki, M. de la Guardia et al., Carbon based nanomaterials for tissue engineering of bone: building new bone on small black scaffolds: a review. J. Adv. Res. 18, 185–201 (2019). https://doi.org/10.1016/j.jare.2019.03.011
M.J. Ditty, D. Ezhilarasan, Β-sitosterol induces reactive oxygen species-mediated apoptosis in human hepatocellular carcinoma cell line. Avicenna J. Phytomed. 11, 541–550 (2021). https://doi.org/10.22038/AJP.2021.17746
M. Wu, T. Wang, W. Zha et al., Effects of nano-SiO2 particles on physio-chemical properties of bioactive tricalcium silicate cements. J. Aust. Ceram. Soc. 57, 9–20 (2021). https://doi.org/10.1007/s41779-020-00489-7
P. Siriphannon, S. Hayashi, A. Yasumori, K. Okada, Preparation and sintering of CaSiO3 from coprecipitated powder using NaOH as precipitant and its apatite formation in simulated body fluid solution. J. Mater. Res. 14, 529–536 (1999). https://doi.org/10.1557/JMR.1999.0076
P. Coussot, Rheometry of Pastes, Suspensions, and Granular Materials: Applications in Industry and Environment (Wiley, New York, 2005)
ASTM International (2013) Standard Test Methods for Density of Compacted or Sintered Powder Metallurgy (PM) Products Using Archimedes’ Principle. ASTM B962-13 1-7
Q.L. Loh, C. Choong, Three-dimensional scaffolds for tissue engineering applications: role of porosity and pore size. Tissue Eng. Part B 19, 485–502 (2013). https://doi.org/10.1089/ten.teb.2012.0437
ASTM C1424-10, Standard test method for monotonic compressive strength of advanced ceramics at ambient temperature. ASTM Int. 08, 1–13 (2010). https://doi.org/10.1520/C1424-15R19.ization
S. Amirkhani, R. Bagheri, A. Zehtab Yazdi, Effect of pore geometry and loading direction on deformation mechanism of rapid prototyped scaffolds. Acta Mater. 60, 2778–2789 (2012). https://doi.org/10.1016/j.actamat.2012.01.044
F. Alam, V.R. Shukla, K.M. Varadarajan, S. Kumar, Microarchitected 3D printed polylactic acid (PLA) nanocomposite scaffolds for biomedical applications. J. Mech. Behav. Biomed. Mater. 103, 103576 (2020). https://doi.org/10.1016/j.jmbbm.2019.103576
ASTM International (2017) Standard Test Method for in vitro Degradation Testing of Hydrolytically Degradable Polymer Resins and Fabricated Forms for Surgical. ASTM B1635-16 7
ASTM International (2019) Standard Guide for Quantifying Cell Viability and Related Attributes within within Biomaterial Scaffolds. ASTM F2739-19 8
K. Liu, P. Liu, R. Liu, X. Wu, Dual AO/EB staining to detect apoptosis in osteosarcoma cells compared with flow cytometry. Med. Sci. Monit. Basic Res. 21, 15–20 (2015). https://doi.org/10.12659/MSMBR.893327
Acknowledgments
The authors thank DST-SERB, Government of India for completely funding this work and VIT, Vellore for providing all the characterization facilities, vacuum furnace, and rheometer.
Funding
This work has been funded by DST-SERB, Government of India under the Start-up research Grant (SRG/2019/002038).
Author information
Authors and Affiliations
Contributions
JR—Writing of original draft, review and editing, Experimentation, Data acquisition and analysis. SRE—Funding acquisition, Conceptualizing, Designing the study, Supervision, Writing- review and editing. DK—Experimentation, Data acquisition and analysis. UP—Experimentation, Data acquisition and analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ravoor, J., Selvam, R.E., Karuppan, D. et al. Development of robocasted MWCNTS-calcium silicate 3D structures for bone regeneration applications with retention of MWCNTS using vacuum sintering technique. Journal of Materials Research 38, 2389–2400 (2023). https://doi.org/10.1557/s43578-023-00968-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/s43578-023-00968-0