Summary
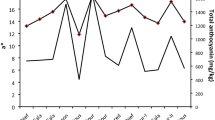
In this article, a simple and sensitive stress-induced high-performance thin-layer chromatography (HPTLC) method is proposed for the quantitative analysis of piperine (a bioactive marker) in the alcoholic extracts of five different marketed powders (P1—P5) and dried seeds (P6) of black pepper (Piper nigrum). HPTLC was performed on glass-backed silica gel 60 F254 HPTLC plates with toluene, ethyl acetate, and glacial acetic acid (7:3:0.5; v/v) as the mobile phase. Scanning and quantification of the developed HPTLC plate (λ = 280 nm) furnished compact and intense spots of piperine at R F = 0.40 ± 0.01. For the proposed method, the estimated regression equation (Y) and r 2 were 9.473X + 2262.23 and 0.997, respectively, in the range of 100–900 ng, the limit of detection (LOD) and limit of quantification (LOQ) were 19.63 and 59.47 ng, respectively. The quantified piperine values (% w/w) in P1, P2, P3, P4, P5, and P6 were 1.75, 2.71, 1.21, 1.67, 2.45, and 1.80, respectively. In the stress study, the treatment of piperine with acid, alkali, and hydrogen peroxide showed its complete degradation. On the other hand, there was no effect of dry or moist heat, and photochemical and ultraviolet light (λ = 254 nm) on the stability of piperine. The present maiden report on a validated HPTLC method for the stress induced analysis of piperine, therefore, would help in the selection of good-quality marketed products. Furthermore, the developed method may help in selecting or rejecting the in-process procedural conditions for making the products and formulations more efficacious and safe.
Similar content being viewed by others
References
N. Kanaki, M. Dave, H. Padh, M.A. Rajani, J. Nat. Med. 62 (2008) 281–283.
Y.Q. Pei, Epilepsia 24 (1983) 177–181.
S.K. Gupta, P. Bansal, R.K. Velpandian Bhardwaj, Pharmacol. Res. 41 (2000) 657–662.
M. Fu, Z.H. Sun, H.C. Zuo, Biol. Pharm. Bull. 33 (2010) 598–603.
S. Atal, R.P. Agrawal, S. Vyas, P. Phadnis, N. Rai, Acta Pol. Pharm. 69 (2012) 965–969.
D.Y. Ouyang, L.H. Zeng, H. Pan, L.H. Xu, Y. Wang, K.P. Liu, X.H. He, Food Chem. Toxicol. 60 (2013) 424–430.
O.B. Al-Baghdadi, N.I. Prater, C.J. Van der Schyf, W.J. Geldenhuys, Bioorg. Med. Chem. Lett. 22 (2012) 7183–7188.
R.F. Tayyem, D.D. Heath, W.K. Al-Delaimy, C.L. Rock, Nutr. Cancer 55 (2006) 126–131.
P.H. Nikam, J. Kareparamban, A. Jadhav, V. Kadam, J. App. Pharm. Sci. 2 (2012) 38–44.
P. Alam, A.J. Al-Rehaily, N.A. Siddiqui, E. S. Al-Sheddi, F. Shakeel, J. Planar Chromatogr. 28 (2015) 54–60.
P. Alam, O.A. Basudan, N.A. Siddiqui, S.I. Alqasoumi, M.S. Abdel- Kader, Abd El Raheim M. Donia, P. Alam, J. P lanar C hromatogr. 28 (2015) 30–35.
P. Alam, N.A. Siddiqui, O.A. Basudan, A.J. Al-Rehaily, S.I. Alqasoumi, P. Alam, M.S. Abdel-Kader, Abd El Raheim M. Donia, F. Shakeel, Afr. J. Tradit. Complement. Altern. Med. 12 (2015) 57–67.
N.A. Siddiqui, P. Alam, A.J. Al-Rehaily, M.M. Al-Oqail, J. Chromatogr. Sci. 53 (2015) 824–829.
M.F. Alajmi, P. Alam, N.A. Siddiqui, O.A. Basudan, A. Hussain, Pak. J. Pharm. Sci. 28 (2015) 2213–2220.
Y.T. Kamal, M. Singh, R. Parveen, S. Ahmad, S. Baboota, I. Ali, K.M. Siddiqui, S.M. Arif Zaidi, Biomed. Chromatogr. 26 (2012) 1183–1190.
S. Ramaswamy, G. Kuppuswamy, P. Dwarampudi, M. Kadiyala, L. Menta, E. Kannan, Pak. J. Pharm. Sci. 27 (2014) 901–906.
N.S. Jeganathan, K. Kannan, R. Manavalan, H.R. Vasanthi, Afr. J. Tradit. Complement. Altern. Med. 5 (2008) 131–140.
K.L. Jain, R.K. Patel, H.P. Patel, J. AOAC Int. 97 (2014) 773–777.
International Conference on Harmonization (ICH) of Technical Requirements for Registration of Pharmaceuticals for Human Use, Harmonised Triplicate Guideline on Validation of Analytical Procedures: Text and Methodology Q2 (R1), Complementary Guideline on Methodology incorporated in November 2005 by the ICH Steering Committee, IFPMA, Geneva.
P. Alam, N.A. Siddiqui, A.J. Al-Rehaily, M.F. Alajmi, O.A. Basudan, T.H. Khan, J. Planar Chromatogr. 27 (2014) 204–209.
U.K. Patil, A. Singh, A.K. Chakraborty, Int. J. Recent Adv. Pharm. Res. 4 (2011) 16–23.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alam, P. Stress-Induced Analysis of Piperine Variability in Different Marketed Black Pepper Powders and Seeds by a Validated High-Performance Thin-Layer Chromatography Method. JPC-J Planar Chromat 30, 251–258 (2017). https://doi.org/10.1556/1006.2017.30.4.3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/1006.2017.30.4.3