Summary
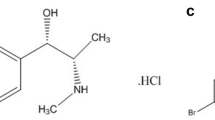
Simple, accurate, precise, sensitive, and validated high-performance liquid chromatography (HPLC) and thin-layer chromatography (TLC)-densitometric methods were developed for the simultaneous determination of carbinoxamine (CAR), pholcodine (PHL), and ephedrine (EPH) in antitussive syrup. In method A, reversed-phase (RP)-HPLC analysis was performed on an Inertsil CN-3 column (250 mm × 4.6 mm, 5 µm), using a mobile phase consisting of acetonitrile-distilled water (pH 3.5) using orthophosphoric acid in the ratio 70:30 (v/v) and flow rate of 1.5 mL min−1. Quantitation was achieved with ultraviolet (UV) detection at 220 nm. In method B, TLC analysis was carried out on an aluminum-backed sheet of silica gel 60 F254 layer using chloroform-propanol-ammonia (6:4:0.1, v/v) as the mobile. Quantification was carried out with UV detection at 245 nm. The validation of the proposed methods was applied according to the International Conference on Harmonization (ICH) guidelines. The suggested methods were successfully applied for the determination of the cited drugs in bulk powder and commercial dosage form.
Similar content being viewed by others
References
K. Parfitt, Martindale: The Complete Drug Reference, 33 edn., The Pharmaceutical Press, London, 2002.
S.C. Sweetman, Martindale: The Complete Drug Reference, The Pharmaceutical Press, London, 2009, 1236–1237.
L. Monferrer-Pons, J. Esteve-Romero, G. Ramis-Ramos, M. Garcia-Alvarez-Coque, Anal. Lett. 29 (1996) 1399–1413.
I. Shoukrallah, Acta Pharm. Jugosl. 41 (1991) 107–115.
I. Shoukrallah, Anal. Lett. 24 (1991) 2043–2058.
A. Ramadan H. Mandil, Anal. Biochem. 353 (2006) 133–137.
J. Araujo, J. Boyer, M. Probecker, STP Pharma Sci. 4 (1988) 598–602.
C.D.A. Bastos, M.A. de Oliveira, Química Nova 32 (2009) 1951–1955.
I.M. Palabiyik, F. Onur, J. Anal. Chem. 67 (2012) 56–63.
A.M. Mansour, J. AOAC Int. 81 (1997) 958–962.
Y.F. Tang, H.L. Wu, S.M. Wu, S.H. Chen, H.S. Kou, J. Microcolumn Sep. 12 (2000) 366–370.
D. Hoffman, M. Leveque, T. Thomson, J. Pharm. Sci. 72 (1983) 1342–1344.
A. Ramadan, S. Antakli, I. Mahmoud, Res. J. Aleppo Uni. 56 (2007) 97.
B.G. Bhavani, B. Gurupadayya, S. Sharfuddin, J. App. Chem. 6 (2014) 46–51.
Z.R. Chen, D.M. Siebert, A.A. Somogyi, F. Bochner, J. Chromatogr. B 424 (1988) 170–176.
A.T. Andresen, M. Krogh, K.E. Rasmussen, J. Chromatogr. B 582 (1992) 123–130.
M, Johansen, F. Tønnesen, K.E. Rasmussen, J. Chromatogr. B 573 (1992) 283–288.
A. Low, R. Taylor, J. Chromatogr. B 663 (1995) 225–233.
A. Petkovska, H. Babunovska, M. Stefova, Maced. J. Chem. Chem. Eng. 30 (2011) 139–150.
W.-G. Zhu, M. Li, Chem. Bioeng. 7 (2011) 025.
J. Sheng, Q. Ping, J. Lei, H. Ju, C. Song, D. Zhang, Anal. Lett. 45 (2012) 652–664.
M. Johansen, K.E. Rasmussen, A.S. Christophersen, J. Chromatogr. B 532 (1990) 277–284.
C. Meadway, S. George, R. Braithwaite, Forensic Sci. Int. 127 (2002) 131–135.
O.M. Denk, G.G. Skellern, D.G. Watson, J. Pharm. Pharmacol. 54 (2002) 87–98.
M. El-Shahat, M. Badei, A. Daifullah, J. Chem. Technol. Biot. 54 (1992) 175–181.
Z. Şentürk, N. Erk, S.A. Özkan, C. Akay, Ş. Cevheroğlu, J. Pharm. Biomed. 29 (2002) 291–298.
P.C. Falcó, A.S. Cabeza, C.M. Legua, Anal. Lett. 27 (1994) 531–547.
N. Okamura, H. Miki, T. Harada, S. Yamashita, Y. Masaoka, Y. Nakamoto, M. Tsuguma, H. Yoshitomi, A. Yagi, J. Pharm. Biomed. 20 (1999) 363–372.
R.A. Niemann, M.L. Gay, J. Agric. Food Chem. 51 (2003) 5630–5638.
Y.-M. Liu, S.-J. Sheu, J. Chromatogr. A 600 (1992) 370–372.
C.L. Flurer, L.A. Lin, R. Duane Satzger, K.A. Wolnik, J. Chromatogr. B 669 (1995) 133–139.
L. Mateus-Avois, P. Mangin, M. Saugy, J. Chromatogr. B 791 (2003) 203–216.
K.W. Phinney, T. Ihara, L.C. Sander, J. Chromatogr. A 1077 (2005) 90–97.
J. Betz, M. Gay, M. Mossoba, S. Adams, B. Portz, J. AOAC Int. 80 (1996) 303–315.
E. Marchei, M. Pellegrini, R. Pacifici, P. Zuccaro, S. Pichini, J. Pharm. Biomed. 41 (2006) 1633–1641.
R.L. Sheppard, X. Tong, J. Cai, J.D. Henion, Anal. Chem. 67 (1995) 2054–2058.
M.L. Gay, R.A. Niemann, S.M. Musser, J. Agric. Food Chem. 54 (2006) 285–291.
R.M. Abdallah, Nat. Prod. Sci. 12 (2006) 55–61.
British Pharmacopoeia, The Stationery Office on behalf of the Medicines and Healthcare Products Regulatory Agency (MHRA), © Crown Copyright, Vol. I & II, London, 2012.
United States Pharmacopoeia Commission, United States Pharmacopeia – National Formulary, United States Pharmacopeial Inc., Rockville, MD, 2004.
ICH Q2A, Note for Guidance on Validation of Analytical Methods: Definition and Terminology, International Conference on Harmonization, IFPMA, Geneva, 1994.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moustafa, A.A., Salem, H., Hegazy, M. et al. Simultaneous Determination of Carbinoxamine, Pholcodine, and Ephedrine in Antitussive Preparation by High-Performance Liquid Chromatography and Thin-Layer Chromatography-Densitometry. JPC-J Planar Chromat 28, 307–315 (2015). https://doi.org/10.1556/1006.2015.28.4.7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/1006.2015.28.4.7