Abstract
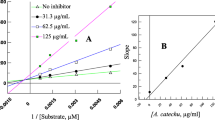
Control of hyperglycemia is an important treatment in metabolic disorders such as type II diabetes and obesity. α-Amylase, as the first enzyme of glucose release from dietary polysaccharides, is a potential target to identify new sources of novel anti-obesity and anti-diabetic drugs. In this work, different herbal extracts as α-amylase inhibitors were studied by measuring the rate of the cleavage of a maltooligomer substrate 2-chloro-4-nitrophenyl-β-D-maltoheptoside. Measurement of chromophore containing products after reversed phase HPLC separation was used for α-amylase activity measurement. Rates of hydrolysis catalysed by human salivary α-amylase were determined in the presence and absence of lyophilised water extracts of eleven herbs. Remarkable bioactivities were found for extracts of Cinnamomum zeylanicum Blume (bark), Camellia sinensis L. (leaf), Ribes nigrum L. (leaf), Laurus nobilis L. (leaf), Vaccinium macrocarpon Aiton (fruit) and Syzygium aromaticum L. (bud). Determined IC50 values were in 0.017–41 μg/ml range for these six selected plant extracts. Our results confirm the applicability of this HPLC-based method for the quick and reliable comparison of plants as α-amylase inhibitors.
Article PDF
Similar content being viewed by others
References
Adefegha, S. A., Oboh, G. (2012). In vitro inhibition activity of polyphenol-rich extracts fro. Syzygium aromaticum (L.) Merr. & Perry (Clove) buds against carbohydrate hydrolyzing enzymes linked to type 2 diabetes and Fe2+-induced lipid peroxidation in rat pancreas. Asian Pac. J. Trop. Biomed. 2, 774–781.
Adisakwattana, S., Lerdsuwankij, O., Poputtachai, U., Minipun, A., Suparpprom, C. (2011) Inhibitory activity of cinnamon bark species and their combination effect with acarbose against intestina l alphaglucosidase and pancreatic alpha-amylase. Plant Foods Hum. Nut. 66, 143–148.
Beejmohun, V., Peytavy-Izard, M., Mignon, C., Muscente-Paque, D., Deplanque, X., Ripoll, C., Chapal, N. (2014) Acute effect of Ceylon cinnamon extract on postprandial glycemia: alpha-amylase inhibition, starch tolerance test in rats, and randomized crossover clinical trial in healthy volunteers. BMC Complement. Alter. Med. 14, 351.
Brayer, G. D., Luo, Y., Withers, S. G. (1995) The structure of human pancreatic alpha-amylase at 1.8 A resolution and comparisons with related enzymes. Protein Sci. 4, 1730–1742.
Brayer, G. D., Sidhu, G., Maurus, R., Rydberg, E. H., Braun, C., Wang, Y., Nguyen, N. T., Overall, C. M., Withers, S. G. (2000) Subsite mapping of the human pancreatic alpha-amylase active site through structural, kinetic, and mutagenesis techniques. Biochemistry 39, 4778–4791.
Farkas, E., Janossy, L., Harangi, J., Kandra, L., Liptak, A. (1997) Synthesis of chromogenic substrates of alpha-amylases on a cyclodextrin basis. Carbohydr. Res. 303, 407–415.
Gao, J., Xu, P., Wang, Y., Wang, Y., Hochstetter, D. (2013) Combined effects of green tea extracts, green tea polyphenols or epigallocatechin gallate with acarbose on inhibition against alpha-amylase and alpha-glucosidas. in vitro. Molecules 18, 11614–11623.
Gusakov, A. V., Kondratyeva, E. G., Sinitsyn, A. P. (2011) Comparison of two methods for assaying reducing sugars in the determination of carbohydrase activities. Int. J. Anal. Chem, article ID: 283658.
Helmstädter, A., Schuster, N. (2010) Vaccinium myrtillus as an antidiabetic medicinal plant research through the ages. Pharmazie 65, 315–321.
Kandra, L., Gyemant, G. (2000) Examination of the active sites of human salivary alpha-amylase (HSA). Carbohydr. Res. 329, 579–585.
Kang, J. G., Park, C. Y. (2012) Anti-obesity-drugs: A review about their effects and safety. Diabetes Metab. 36, 13–25.
Khan, A., Zaman, G., Anderson, R. A. (2009) Bay leaves improve glucose and lipid profile of people with type 2 diabetes. J. Clin. Biochem. Nutr. 44, 52–56.
Lehmann, U., Robin, F. (2007) Slowly digestible starch–its structure and health implications: A review. Trends Food Sci. Tech. 18, 346–355.
Lehoczki, G., Szabo, K., Takacs, I., Kandra, L., Gyemant, G. (2016) Simple ITC method for activity and inhibition studies on human salivary alpha-amylase. J. Enzyme Inhib. Med. Ch. 6, 1648–1653.
Marsh, K., Brand-Miller, J. C. (2008) State of the art reviews: Glycemic index, obesity, and chronic disease. Am. J. Lifestyle Med. 2, 142–150.
McDougall, G. J., Stewart, D. (2005) The inhibitory effects of berry polyphenols on digestive enzymes. BioFactors 23, 189–195.
Miao, M., Jiang, B., Jiang, H., Zhang, T., Li, X. (2015) Interaction mechanism between green tea extract and human alpha-amylase for reducing starch digestion. Food Chem. 186, 20–25.
Najafian, M., Ebrahim-Habibi, A., Yaghmaei, P., Parivar, K., Larijani, B. (2010) Core structure of flavonoids precursor as an antihyperglycemic and antihyperlipidemic agent: a. in vivo study in rats. Acta Biochim. Pol. 57, 553–560.
Pinto, Mda, S., Ghaedian, R., Shinde, R., Shetty, K. (2010) Potential of cranberry powder for management of hyperglycemia using in vitro Models. J. Med. Food. 13, 1036–1044.
Rossomando, E. F. (1998) HPLC in enzymatic analysis. In. Methods of Biochemical Anaysis, John Wiley & Sons, pp. 1–24.
Salehi, P., Asghari, B., Esmaeili, M. A., Dehghan, H., Ghazi, I. (2013) α-Glucosidase and α-amylase inhibitory effect and antioxidant activity of ten plant extracts traditionally used in Iran for diabetes. J. Med. Plant. Res. 7, 257–266.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Takács, I., Takács, Á., Pósa, A. et al. HPLC Method for Measurement of Human Salivary α-Amylase Inhibition by Aqueous Plant Extracts. BIOLOGIA FUTURA 68, 127–136 (2017). https://doi.org/10.1556/018.68.2017.2.1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/018.68.2017.2.1