Abstract
Background
The population aged ≥90 years is increasing worldwide, yet nearly 50% of elderly breast cancer (BC) patients receive suboptimal treatments, resulting in high rates of BC-related mortality. We analyzed clinical and survival outcomes of nonagenarian BC patients to identify effective treatment strategies.
Methods
This single-institution retrospective cohort study analyzed patients aged ≥90 years diagnosed with stage I–III BC between 2007 and 2018. Patients were categorized into three treatment groups: traditional surgery (TS), performed according to local guidelines; current-standard surgery (CS), defined as breast surgery without axillary surgery (in concordance with 2016 Choosing Wisely guidelines) and/or cavity shaving; and non-surgical treatment (NS). Clinicopathological features were recorded and recurrence rates and survival outcomes were analyzed.
Results
We collected data from 113 nonagenarians with a median age of 93 years (range 90–99). Among these patients, 43/113 (38.1%) underwent TS, 34/113 (30.1%) underwent CS, and 36/113 (31.9%) underwent NS. The overall recurrence rate among surgical patients was 10.4%, while the disease progression rate in the NS group was 22.2%. Overall survival was significantly longer in surgical patients compared with NS patients (p = 0.04). BC-related mortality was significantly higher in the NS group than in the TS and CS groups (25.0% vs. 0% vs. 7.1%, respectively; p = 0.01). There were no significant differences in overall survival and disease-free survival between the TS and CS groups (p = 0.6 and p = 0.8, respectively), although the TS group experienced a significantly higher overall postoperative complication rate (p < 0.001).
Conclusions
Individualized treatment planning is essential for nonagenarian BC patients. Surgery, whenever feasible, remains the treatment of choice, with CS emerging as the best option for the majority of patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The proportion of the elderly population is increasing worldwide. In 2020, the population aged 90 years or older increased by 10% compared with 50 years earlier,1 with faster growth of people aged >80 years of age compared with those >65 years of age.2 Breast cancer (BC) incidence has also risen with age. In 2020, 45% of new BC cases were diagnosed in women aged over 65 years, with one in nine of these cases occurring in nonagenarians.3 Nearly 50% of elderly BC patients receive suboptimal or unconventional treatments,4 reflecting a lack of standardized therapeutic approaches,5 mainly due to under-enrollment in specific clinical trials.6 Despite pre-existing comorbidities and lower life expectancy,7 40% of women aged ≥80 years with BC die from cancer-related causes,8 mainly attributed to less aggressive management, resulting in poorer disease-free survival (DFS) and overall survival (OS) rates.9 Surgery remains the gold-standard treatment for stage I and II BC in elderly patients.10 Both mastectomy and breast conserving surgery achieve superior local control compared with primary endocrine therapy;11 however, an appropriate management should consider patient frailty and background as integral parts of treatment planning.12 A comprehensive geriatric assessment is crucial to identify elderly patients who can tolerate treatments while balancing factors influencing clinical and survival outcomes.13 We evaluated the clinical and survival outcomes in patients aged ≥90 years newly diagnosed with BC, and to assess which therapeutic approach is associated with the best results.
Methods
This single-center, retrospective cohort study was conducted at the Veneto Institute of Oncology. We included consecutive patients aged ≥90 years with newly diagnosed stage I–III BC treated between January 2007 and December 2018. Patients with stage IV disease were excluded. A minimum follow-up period of 5 years was guaranteed whenever the survival period allowed. This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines14 and the study protocol was approved by the local Ethics Committee (CESC-IOV 2023-12).
Clinical Features and Geriatric Assessment
Complete clinical features were collected for all the patients from our institutional database.
All patients underwent comprehensive geriatric evaluation, which included the Karnofsky Performance Status (KPS) scale to evaluate functional abilities; the Eastern Cooperative Oncology Group Performance Status (ECOG PS) to measure functional status; the Age-not Charlson Comorbidity Index (AN-CCI) to categorize comorbidities and evaluate mortality risk (excluding age due to the uniformly aged very elderly population); the Mini-Mental State Examination (MMSE) for cognitive function assessment; the Geriatric Depression Scale (GDS) for psychological evaluation; and body mass index (BMI) to assess the nutritional status.
Therapeutic Approaches
All therapeutic approaches were described (surgery, hormone therapy [HT], chemotherapy, and radiotherapy) and the following surgical aspects were examined: type of breast surgery (mastectomy vs. wide local excision [WLE]); axillary surgery (sentinel lymph node biopsy [SLNB] and/or axillary lymph node dissection [ALND]); margin status of the specimen (defined as positive if ‘ink on tumor’ was present, and close if any margin width was <2 mm); and postoperative complications. The TNM staging system was adopted15 and tumor subtypes were categorized according to the World Health Organization biomolecular classification.16 Within the medical therapies, we comprehensively documented the treatment pathway, timing, and the administered drugs. Any clinical response to medical therapies was recorded according to the Response Evaluation Criteria in Solid Tumors (RECIST) criteria.17 Radiotherapy treatment fields were specified.
Treatment Groups
Patients were stratified into three distinct groups according to the treatment received: traditional surgery (TS), current-standard surgery (CS), and non-surgical treatment (NS). TS was defined as per local guidelines, mainly adopted before 2016,18 and involving either mastectomy or WLE with margin cavity shaving19 possibly combined with axillary surgery, such as SLNB and/or ALND. CS was defined as either mastectomy or WLE alone, without axillary surgery, even for staging purposes only (according to the 2016 Society of Surgical Oncology Choosing Wisely guidelines),20 or additional surgical procedures (e.g. margin cavity shaving).
Study Endpoints and Statistical Analysis
The main endpoints were OS, DFS, progression-free survival (PFS) and recurrence rate (RR). DFS analysis was limited to surgical patients, while PFS analysis was exclusive to NS patients. Categorical variables are expressed as numbers (%) and compared using the Chi-square or Fisher's exact tests. Continuous variables are expressed as median ± interquartile range (IQR) and compared using the Wilcoxon or Kruskal–Wallis rank-sum test.
Given the advanced age of the cohort, the median follow-up time was estimated using the reverse Kaplan–Meier estimator,21 which combines Korn’s potential follow-up advantage to overcome the bias of underestimating the follow-up time.
OS, DFS, and PFS were evaluated using Kaplan–Meier survival analysis, employing the log-rank test to compare different patient groups. Cancer-specific survival (CSS) was assessed through competing risk analysis, considering deaths from non-BC causes as competing risks. The Gray test was used to compare the surgical and non-surgical groups.
A multiple Cox proportional hazards regression model was built to assess whether any differences in OS could be attributed to the type of surgery (TS vs. CS). To partially overcome the limitations of retrospective studies and address potential sources of bias, this model was adjusted for well-established BC prognostic factors. This was achieved by considering the number of events that occurred and trying to mitigate any collinearity between our predictors, i.e., age, type of surgery, Charlson score, and tumor stage. The results are expressed as hazard ratios (HR) with 95% confidence intervals (CI) and associated p-values. A p-value <0.05 was considered statistically significant. Statistical analyses were performed using R software.22
Results
This study included 113 nonagenarian Caucasian patients living in Italy, of whom only one patient was male. Of these patients, 43 (38.1%) underwent TS, 34 (30.1%) underwent CS, and 36 (31.9%) did not undergo surgery (and represent the NS group).
Geriatric Assessment
The median age of the entire cohort was 93 years (range 90–99). The most common comorbidities were hypertension (66.4%), osteoporosis (29.2%), diabetes (24.8%), and atrial fibrillation (23.0%) [Table 1]. Approximately half of the patients presented with a good clinical condition, as indicated by Karnofsky scores of 80–100 (54.9%), ECOG scores of 0–1 (54.9%), and AN-CCI scores of 0–1 (48.6%). Cognitive function was normal in 54.9% of patients, and 74.3% exhibited normal humoral status according to the GDS.
Neoplastic Features and Staging
Five patients presented with bilateral tumors. A palpable mass was the clinical presentation in 115/118 cases (97.5%), with 28.8% also showing visible findings and 22.0% showing palpable axillary nodes. Most tumors were invasive carcinomas of no special type (78.8%) and were categorized as Luminal A (84.7%). Neither ductal carcinoma in situ nor triple-negative BC cases were recorded. The global median tumor size was 27 mm, with significant differences among the groups (28 mm in the TS group, 21 mm in the AS group, and 31 mm in the NS group; p < 0.001). At presentation, half of the cases (50.0%) were classified as T2 neoplasia, while T4 accounted for 18.6% of cases. Lymph node metastases were detected in 22.0% of cases (Table 1).
Primary Systemic Therapy
Primary systemic therapy (PST), always hormone-based, was administered to 48/113 patients (42.5%), of whom 12/48 (25.0%) underwent subsequent surgery, while 36 (31.9% of the entire cohort) did not (Table 2).
HT as a definitive treatment was delivered to 31.9% of patients, with a median duration of 24 months, predominantly using aromatase inhibitors (AIs; 86.1%). Among these patients, 30.6% had to switch type of HT due to adverse effects, most commonly arthralgia. Partial clinical response was documented in 66.7% of patients, while two patients achieved complete clinical response; disease progression was recorded in 22.2% of patients, while two patients showed stable disease. Neoadjuvant HT was administered to 15.6% of the surgical patients, mainly using AIs (91.7%), for a median duration of 19 months. This approach was chosen for patients who were initially non-surgical candidates due to transient comorbidities, those who needed a reduction in BC dimensions to become operable, or for patients who initially refused surgery. Of these patients, 66.7% showed partial response, whereas 33.3% experienced disease progression. The following surgical treatment was TS and CS for 8 and 4 of these patients, respectively.
Chemotherapy was not administered to any patients, neither in the neoadjuvant setting nor as part of an exclusive medical treatment regimen. This decision was mainly due to the frailty of this specific age group of patients. After multidisciplinary discussion, it was determined that the risks outweighed the potential benefits, even in cases of human epidermal growth factor receptor 2-positive tumors, where trastuzumab would have posed a risk of severe cardiotoxicity, especially in patients already suffering from heart disease.
Surgical Treatment
Seventy-seven patients (68.1% of the entire cohort, corresponding to 79 tumors) underwent surgery, of whom 65 (84.4%) underwent upfront surgery. Mastectomy was performed in 39.2% of tumors, while 60.8% underwent WLE. Mastectomy was the most common procedure in the TS group (27/44, 61.4%), while WLE was preferred in the CS group (31/35, 88.6%; p < 0.001). Axillary surgery was only performed in the TS group, with SLNB performed in 30/44 cN0 cases (68.2%) and ALND in all 12 cN+ patients. In two cases, sentinel lymph nodes were positive and ALND was consequently performed.
The overall positive and close margin rates were 11.4% and 13.9%, respectively, and no significant differences were observed between the two surgical groups (p = 0.151). Re-operation for margin clearance was performed in three TS selected cases with positive margins, while no close margin was considered for additional surgery.
The overall rate of patients with at least one postoperative complication was 51.9%, with breast seroma being the most frequent (22.8% of all procedures), occurring after 41.9% of mastectomies and 10.4% of WLEs (p < 0.001). The TS group experienced a significantly higher postoperative complication rate, including minor complications, than the CS group (74.4% vs. 23.5%; p < 0.001), with a reported axillary lymphocele rate of 31.8%, mainly after ALND (71.4% vs. 13.3% after SLNB; p < 0.01). Mild arm lymphedema was recorded after ALND in 18.2% of TS procedures. All postoperative complications were conservatively managed and no surgical re-intervention was required. The 30-day postoperative mortality rate was zero.
Adjuvant Therapies
Adjuvant HT was administered to 72.7% of surgical patients, with a median duration of 34 months; in 7.1% of cases, adjuvant HT was suspended due to adverse effects. AIs were used in 91.1% of patients. None of the patients received adjuvant chemotherapy. Adjuvant radiotherapy was recommended for 21/77 surgical patients (27.3%), but only 9.1% adhered to the treatment.
Recurrences and Disease Progression
Among all surgical patients, the RR was 10.4%, whereas the NS group experienced an overall (local and/or distant) disease progression rate of 22.2% (p = 0.049). Recurrence management consisted of additional surgery in the TS group only, performed in three of five recorded recurrences (60.0%). Specifically, this included two WLEs in patients initially staged IIB who subsequently developed ipsilateral BC recurrence, and one ALND in a stage IIIB patient with axillary recurrence.
HT was the treatment used for 80% of TS group recurrences and 33.3% of CS group recurrences. Additionally, 50% of NS patients experiencing disease progression underwent drug switching during the course of HT. Supportive care as the sole recurrence treatment was provided to 66.7% and 37.5% of CS and NS patients, respectively.
Survival Outcomes
Over a median estimated follow-up of 77 months (real median follow-up of 42 months and a loss to follow-up rate of 7.4%), 54.9% (62/113) of the cohort died, with only 12.9% of these deaths caused by BC. BC-related mortality was significantly higher in the NS group than in the TS and CS groups (25% vs. 0% and 7.1%, respectively; p = 0.01). The leading causes of non-BC-related deaths were heart failure (43.5%), respiratory failure (19.4%), and cancer at other sites (12.9%).
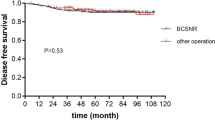
The median global OS was 52 months, with median DFS and median PFS not reached in any of the three groups (Fig. 1). Surgical patients experienced a significantly higher OS than NS patients (p = 0.04) (Fig. 2a). When considering non-BC-related deaths as a competing risk, surgical patients showed a significantly lower BC-related mortality rate (p = 0.002), with no notable difference in mortality from other causes between the groups (p = 0.8) (Fig. 2b). No significant differences in OS or DFS were observed between the TS and CS groups (p = 0.6 and p = 0.8, respectively) (Fig. 3). A specific survival analysis revealed no significant difference in OS (medians of 66 and 51 months, respectively) and DFS between clinically node-negative (cN0) patients at diagnosis undergoing TS or CS (p = 0.7 and p = 0.8, respectively) (Fig. 4).
A Cox Model analysis, including all the surgical patients, revealed that the Charlson score at diagnosis was the only significant predictor of poorer OS (HR 1.24, 95% CI 1.03–1.48; p = 0.022). Conversely, age, tumor stage, and, most notably, the type of surgery did not significantly affect the OS (Table 3).
Discussion
The very elderly population remains significantly underrepresented in BC clinical trials due to challenges in their enrollment in prospective studies. This has led to a lack of evidence and limited recommendations for their optimal clinical management.5 Despite the increasing incidence of BC with age, older patients frequently face underdiagnosis, understaging, and undertreatment compared with their younger counterparts, resulting in increased mortality rates.23,24 To the best of our knowledge, this study represents the largest single-center analysis of BC treatment among nonagenarians to date. Similar to prior studies,23 almost all BC cases in our cohort (97.5%) were palpable. This highlights the impact of screening omission in elderly women, consistently with specific guidelines25,26 for this age group, resulting in delayed diagnosis.
Comprehensive Geriatric Assessment
In the United States, individuals aged ≥ 90 years have a life expectancy of over 4.5 years,27 while in Europe, women aged 92–93 years have an overall 11.4% chance of reaching 100.28 Despite the advanced age at diagnosis, nearly half of our patients were in good clinical condition, making them potential candidates for surgery. Among these fragile patients, it is crucial to identify those who can tolerate the treatments, achieving survival benefits. Consequently, we conducted a comprehensive geriatric assessment for all elderly patients to predict adverse events and to determine the most suitable treatment plans. Our analysis identified the AN-CCI as the sole prognostic factor for OS within the surgical cohort, underlining its utility to evaluate a patient’s suitability for surgery.
Benefits of Surgery
Rao et al. demonstrated that BC surgery offered survival advantages over primary HT in patients aged ≥ 80 years, with a significant decrease in local RR and improved 5-year survival rates.29 Similarly, we observed a lower RR (10.4% vs. 22.2%) and a better 5-year OS (51% vs. 36%) in surgical patients compared with the NS group. Di Lascio et al. retrospectively analyzed 58 BC patients aged ≥ 89 years and found that among surgical patients, 56% underwent mastectomy and 71% had axillary surgery, resulting in a 10% relapse rate and a median survival of 50 months.30 Our experience showed a median OS of 52 months, with lower axillary surgery (54.4%) and mastectomy (39.2%) rates; however, the mastectomy rate was significantly higher than that observed in the younger population with BC.31 This is likely due to a higher clinical T stage, contraindications to radiation therapy, and the omission of level II oncoplastic techniques in this demographic.
Axillary Surgery
Elderly BC patients are less likely to undergo axillary surgery due to the lack of long-term survival benefit32 and increased risk of specific morbidities.33
Recently, the SOUND trial,34 along with other randomized trials, investigated SLNB omission in cN0 younger patients,35 suggesting that its implementation in the elderly is even more warranted. In our cohort, axillary surgery was performed in only 36.6% of patients, with no OS advantage for cN0 patients, supporting its omission. The International Breast Cancer Study Group demonstrated that avoiding axillary surgery in women aged ≥60 years diagnosed with cN0 BC improved their quality of life without adversely affecting DFS or OS.36
A 2016 consensus statement from the Society of Surgical Oncology20 declared SNLB avoidable in clinically node-negative women aged ≥70 years with early-stage hormone receptor-positive, HER2-negative invasive BC; however, our experience shows that current clinical practice still diverges from these recommendations.37 In our population, 49.2% of cN0 patients underwent SLNB (the majority were performed before 2016), while all 12 cN+ patients underwent ALND. A recent study involving 125 patients aged ≥65 years with favorable BC undergoing breast-conserving surgery without SLNB reported only 1.6% axillary recurrences, further supporting this approach.38
Future studies could explore the feasibility of totally omitting axillary surgery in nonagenarians even with involved axillary lymph nodes, relying on systemic therapy only. This strategy could reduce post-surgical complications without negatively affecting OS. Alternatively, only the excision of macroscopically involved lymph nodes in patients, or tailored axillary dissection, may be considered, as already being investigated by the TAXIS trial,39 and hopefully by further prospective studies.
Radiotherapy
Adjuvant radiotherapy slightly reduces the risk of local recurrence, but does not significantly improve OS in older populations.40,41,42 It is crucial to balance its benefits against potential adverse effects, particularly among elderly patients.43 The St. Gallen International Consensus Guidelines recommend adjuvant radiotherapy for older women with a life expectancy exceeding 10 years.44 Consequently, the omission of radiotherapy for nonagenarians is justifiable because of their shorter life expectancies and the high incidence of contraindications due to their comorbidities. In our experience, radiotherapy was advised in selected patients (27.3%), although adherence was notably low (9.1%).
Hormone Therapy
Fennessy et al. reported reduced OS and increased BC-related mortality in patients aged over 70 years treated with tamoxifen alone, compared with those receiving surgery plus tamoxifen. Significant differences emerged after 5 years, suggesting tamoxifen as a potential option for elderly estrogen receptor-positive BC patients with a life expectancy of <5 years.45 The Italian GRETA trial showed increased local disease progression in the tamoxifen-alone arm compared with the tamoxifen plus surgery arm, without differences in OS or BC-specific survival.46 In our cohort, 31.9% of patients received HT alone, experiencing significantly higher global relapse and BC-related mortality rates than surgical patients (22.2% vs. 10.4%, and 25.0% vs. 1.33%, respectively). A specific survival analysis revealed significantly improved OS in surgical patients compared with that in the NS group, even when considering only BC-related deaths.
Chemotherapy
In our experience, chemotherapy was never administered to nonagenarian BC patients due to their frailty and comorbidities. Elkin et al. demonstrated a reduction in all-cause mortality by approximately 16% among older patients with estrogen receptor-negative BC who received systemic chemotherapy;47 however, older patients may have an increased risk of cardiac toxicity, treatment-induced myelodysplasia, or acute leukemia.48,49 Consequently, the indications for chemotherapy in patients aged ≥90 years remain very rare.
Study Limitations
This study has several limitations. First, its retrospective, single-institution design may limit the generalizability of the findings. Despite representing the largest cohort within this specific age group reported in the literature, the sample size is relatively small. Additionally, the population lacks homogeneity due to different comorbidities, increased competing causes of death, discrepancies in therapy adherence and tolerance, and uneven distribution of cancer stages across groups. The better outcomes found in the surgical population may at least partially be due to selection bias, as surgical patients may present with a more favorable tumor stage. However, the Age-Adjusted Charlson Comorbidity Index was not significantly different among the three treatment groups.
Conclusions
With the increasing incidence of BC in the oldest old population and the global aging trend, it is imperative to collect stronger evidence on surgical and oncological outcomes for BC patients aged ≥90 years. Rigorous geriatric assessment and a specialized multidisciplinary approach should guide treatment decisions. Our findings underscore the need to tailor the surgical indications for nonagenarians, supporting omission of axillary surgery and highlighting the survival advantages of breast surgery over non-surgical approaches. Consequently, whenever feasible, surgery should be the primary treatment of choice, even within this growing patient demographic, with alternative therapies reserved for patients not suitable for surgery. Aging should not be considered a disease in itself.
Data Availability
All relevant data are included within this paper and its supporting information files. Anything else required is available upon request.
References
Knoema. World - Total population aged 90 years and over. Available at: https://knoema.com/atlas/World/topics/Demographics/Age/Population-aged-90-years
United Nations. World Social Report 2023: leaving no one behind in an ageing world. Sales No. E.23.IV.2. New York, NY: United Nations; 2023.
SEER. Cancer Stat Facts: Female Breast Cancer. Available at: https://seer.cancer.gov/statfacts/html/breast.html
Lavelle K, Todd C, Moran A, Howell A, Bundred N, Campbell M. Non-standard management of breast cancer increases with age in the UK: a population based cohort of women > or =65 years. Br J Cancer. 2007;96(8):1197–203. https://doi.org/10.1038/sj.bjc.6603709.
Bouchardy C, Rapiti E, Fioretta G, et al. Undertreatment strongly decreases prognosis of breast cancer in elderly women. J Clin Oncol. 2003;21:3580–7. https://doi.org/10.1200/JCO.2003.02.046.
Hutchins LF, Unger JM, Crowley JJ, et al. Underrepresentation of patients 65 years of age or older in cancer- treatment trials. N Engl J Med. 1999;341:2061–7. https://doi.org/10.1056/NEJM199912303412706.
Plichta JK, Thomas SM, Vernon R, et al. Breast cancer tumor histopathology, stage at presentation, and treatment in the extremes of age. Breast Canc Res Treat. 2020;180:227–35. https://doi.org/10.1007/s10549-020-05542-4.
Van De Water W, Markopoulos C, Van De Velde CJH, et al. Association between age at diagnosis and disease-specific mortality among postmenopausal women with hormone receptor-positive breast cancer. JAMA. 2012;307(6):590–7. https://doi.org/10.1001/jama.2012.84.
Angarita FA, Chesney T, Elser C, et al. Treatment patterns of elderly breast cancer patients at two Canadian cancer centers. Eur J Surg Oncol. 2015;41(5):625–34. https://doi.org/10.1016/j.ejso.2015.01.028.
Ferrucci M, Milardi F, Passeri D, et al. Intraoperative ultrasound-guided conserving surgery for breast cancer: no more time for blind surgery. Ann Surg Oncol. 2023;30(10):6201–14. https://doi.org/10.1245/s10434-023-13900-x.
Hind D, Wyld L, Reed MW. Surgery, with or without tamoxifen, vs tamoxifen alone for older women with operable breast cancer: cochrane review. Br J Cancer. 2007;96(7):1025–9. https://doi.org/10.1038/sj.bjc.6603600.
Shimozuma K. Quality of life assessment. Breast Cancer. 2002;9:100–6. https://doi.org/10.1007/BF02967573.
Taira N, Sawaki M, Takahashi M, et al. Comprehensive geriatric assessment in elderly breast cancer patients. Breast Cancer. 2010;17(3):183–9. https://doi.org/10.1007/s12282-009-0167-z.
Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, STROBE Initiative. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370(9596):1453–7. https://doi.org/10.1136/bmj.39335.541782.AD.
Zhu H, Doğan BE. American joint committee on cancer’s staging system for breast cancer, eighth edition: summary for clinicians. Eur J Breast Health. 2021;17(3):234–8. https://doi.org/10.4274/ejbh.galenos.2021.2021-4-3.
Tan PH, Ellis I, Allison K, WHO Classification of Tumours Editorial Board, et al. The 2019 World Health Organization classification of tumours of the breast. Histopathology. 2020;77(2):181–5. https://doi.org/10.1111/his.14091.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47. https://doi.org/10.1016/j.ejca.2008.10.026.
Cardoso F, Kyriakides S, Ohno S, et al. Early breast cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30(8):1194–220. https://doi.org/10.1093/annonc/mdz173.
Chagpar AB, Killelea BK, Tsangaris TN, et al. a randomized, controlled trial of cavity shave margins in breast cancer. N Engl J Med. 2015;373(6):503–10. https://doi.org/10.1056/NEJMoa1504473.
Society of Surgical Oncology. Choosing Wisely. Published 12 Jul 2016. Available at: https://www.choosingwisely.org/clinician-lists/sso-sentinel-node-biopsy-in-node-negative-women-70-and-over/.
Schemper M, Smith TL. A note on quantifying follow-up in studies of failure time. Control Clin Trials. 1996;17(4):343–6. https://doi.org/10.1016/0197-2456(96)00075-x.
R Core Team (2022). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available at: https://www.R-project.org/.22
Litvak DA, Arora R. Treatment of elderly breast cancer patients in a community hospital setting. Arch Surg. 2006;141(10):985–90. https://doi.org/10.1001/archsurg.141.10.985.
Schonberg MA, Marcantonio ER, Li D, et al. Breast cancer among the oldest old: tumor characteristics, treatment choices, and survival. J Clin Oncol. 2010;28(12):2038–45. https://doi.org/10.1200/JCO.2009.25.9796.
Schünemann HJ, Lerda D, Quinn C, et al. Breast cancer screening and diagnosis: a synopsis of the European breast guidelines. Ann Intern Med. 2020;172:46.
Ren W, Chen M, Qiao Y, Zhao F. Global guidelines for breast cancer screening: A systematic review. Breast. 2022;64:85–99. https://doi.org/10.1016/j.breast.2022.04.003.
Arias E, Bastian B, Xu J, et al. U.S. state life tables, 2018. Natl Vital Stat Rep. 2021;70(1):1–18.
Thinggaard M, McGue M, Jeune B, et al. Survival prognosis in very old adults. J Am Geriatr Soc. 2016;64(1):81–8. https://doi.org/10.1111/jgs.13838.
Rao VS, Jameel JK, Mahapatra TK, et al. Surgery is associated with lower morbidity and longer survival in elderly breast cancer patients over 80. Breast J. 2007;13(4):368–73. https://doi.org/10.1111/j.1524-4741.2007.00444.x.
Di Lascio S, Tognazzo E, Bigiotti S, et al. Breast cancer in the oldest old (≥ 89 years): tumor characteristics, treatment choices, clinical outcomes and literature review. Eur J Surg Oncol. 2021;47(4):796–803. https://doi.org/10.1016/j.ejso.2020.10.008.
Biganzoli L, Battisti NML, Wildiers H, et al. Updated recommendations regarding the management of older patients with breast cancer: a joint paper from the European Society of Breast Cancer Specialists (EUSOMA) and the International Society of Geriatric Oncology (SIOG). Lancet Oncol. 2021;22(7):e327–40. https://doi.org/10.1016/S1470-2045(20)30741-5.
Corso G, Magnoni F, Montagna G, et al. Long-term outcome and axillary recurrence in elderly women (≥70 years) with breast cancer: 10-years follow-up from a matched cohort study. Eur J Surg Oncol. 2021;47(7):1593–600. https://doi.org/10.1016/j.ejso.2021.02.027.
Mandelblatt JS, Edge SB, Meropol NJ, et al. Sequelae of axillary lymph node dissection in older women with stage 1 and 2 breast carcinoma. Cancer. 2002;95(12):2445–54. https://doi.org/10.1002/cncr.10983.
Gentilini OD, Botteri E, Sangalli C, et al. Sentinel lymph node biopsy vs no axillary surgery in patients with small breast cancer and negative results on ultrasonography of axillary lymph nodes: the SOUND randomized clinical trial. JAMA Oncol. 2023;9(11):1557–64. https://doi.org/10.1001/jamaoncol.2023.3759.
Reimer T. Omission of axillary sentinel lymph node biopsy in early invasive breast cancer. The Breast. 2023;67:124–8. https://doi.org/10.1016/j.breast.2023.01.002.
Rudenstam CM, Zahrieh D, Forbes JF, et al. Randomized trial comparing axillary clearance versus no axillary clearance in older patients with breast cancer: first results of International Breast Cancer Study Group Trial 10–93. J Clin Oncol. 2006;24(3):337–44. https://doi.org/10.1200/JCO.2005.01.5784.
Heidinger M, Maggi N, Dutilh G, et al. Use of sentinel lymph node biopsy in elderly patients with breast cancer - 10-year experience from a Swiss university hospital. World J Surg Oncol. 2023;21(1):176. https://doi.org/10.1186/s12957-023-03062-1.
Chung AP, Dang CM, Karlan SR, et al. A prospective study of sentinel node biopsy omission in women age ≥ 65 years with ER+ breast cancer. Ann Surg Oncol. 2024;31(5):3160–7. https://doi.org/10.1245/s10434-024-15000-w.
Weber WP, Matrai Z, Hayoz S, et al. Association of axillary dissection with systemic therapy in patients with clinically node-positive breast cancer. JAMA Surg. 2023;158(10):1013–21. https://doi.org/10.1001/jamasurg.2023.2840.
Hughes KS, Schnaper LA, Bellon JR, et al. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol. 2013;31(19):2382–7. https://doi.org/10.1200/JCO.2012.45.2615.
Matuschek C, Bölke E, Haussmann J, et al. The benefit of adjuvant radiotherapy after breast conserving surgery in older patients with low risk breast cancer- a meta-analysis of randomized trials. Radiat Oncol. 2017;12(1):60. https://doi.org/10.1186/s13014-017-0796-x.
Huang XZ, Chen Y, Chen WJ, et al. Effect of radiotherapy after breast-conserving surgery in older patients with early breast cancer and breast ductal carcinoma in situ: a meta-analysis. Oncotarget. 2017;8(17):28215–25. https://doi.org/10.18632/oncotarget.15998.
Darby SC, McGale P, Taylor CW, et al. Long-term mortality from heart disease and lung cancer after radiotherapy for early breast cancer: prospective cohort study of about 300,000 women in US SEER cancer registries. Lancet Oncol. 2005;6:557–65. https://doi.org/10.1016/S1470-2045(05)70251-5.
Burstein HJ, Curigliano G, Thürlimann B, et al. Customizing local and systemic therapies for women with early breast cancer: the St. Gallen International Consensus Guidelines for treatment of early breast cancer 2021. Ann Oncol. 2021;32(10):1216–35. https://doi.org/10.1016/j.annonc.2021.06.023.
Fennessy M, Bates T, MacRae K, et al. Late follow-up of a randomized trial of surgery plus tamoxifen versus tamoxifen alone in women aged over 70 years with operable breast cancer. Br J Surg. 2004;91(6):699–704. https://doi.org/10.1002/bjs.4603.
Mustacchi G, Scanni A, Capasso I, et al. Update of the phase III trial “GRETA” of surgery and tamoxifen versus tamoxifen alone for early breast cancer in elderly women. Future Oncol. 2015;11(6):933–41. https://doi.org/10.2217/fon.14.266.
Elkin EB, Hurria A, Mitra N, et al. Adjuvant chemotherapy and survival in older women with hormone receptor-negative breast cancer: assessing outcome in a population-based observational cohort. J Clin Oncol. 2006;24:2750–6. https://doi.org/10.1200/JCO.2005.03.6053.
Doyle JJ, Neugut AI, Jacobson JS, et al. Chemotherapy and cardiotoxicity in older breast cancer patients: a population-based study. J Clin Oncol. 2005;23:8597–605. https://doi.org/10.1200/JCO.2005.02.5841.
Muss HB, Berry DA, Cirrincione C, et al. Toxicity of older and younger patients treated with adjuvant chemotherapy for node-positive breast cancer: the Cancer and Leukemia Group B experience. J Clin Oncol. 2007;25:3699–704. https://doi.org/10.1200/JCO.2007.10.971.
Funding
This research was funded by the Italian Ministry of Health Ricerca Corrente.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Massimo Ferrucci, Daniele Passeri, Francesco Milardi, Andrea Francavilla, Matteo Cagol, Mariacristina Toffanin, Giacomo Montagna, and Alberto Marchet have no conflicts of interest to declare that may be relevant to the contents of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ferrucci, M., Passeri, D., Milardi, F. et al. Surgery Plays a Leading Role in Breast Cancer Treatment for Patients Aged ≥90 Years: A Large Retrospective Cohort Study. Ann Surg Oncol (2024). https://doi.org/10.1245/s10434-024-15790-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1245/s10434-024-15790-z