Abstract
Background
The extent of histological organ involvement (HOI) to organs and structures of a retroperitoneal liposarcoma may have prognostic implications. This study investigated incidence, characteristics, and risk association of HOI in these patients.
Patients and Methods
Data of patients who underwent multivisceral resection for primary liposarcoma (2009–2014) were retrospectively analyzed. HOI was the variable of interest and was classified into four degrees: absent (HOI-0), perivisceral (HOI-1), initial (HOI-2), and advanced (HOI-3). Primary endpoint was overall survival (OS). Secondary endpoint was disease-free survival (DFS). The prognostic value of HOI was adjusted for preoperative treatment and the Sarculator nomogram score.
Results
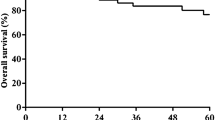
A total of 109 patients were included. HOI-0, HOI-1, HOI-2, and HOI-3 were detected in 9 (8.3%), 11 (10.1%), 43 (39.4%), and 46 (42.2%) patients. Median follow-up was 8.4 years [interquartile range (IQR) 7.2–9.6 years]. There were 68 recurrences and 50 patient deaths observed, resulting in a 10-year OS and DFS of 51.1% [95% confidence interval (CI) 41.9–62.1%] and 34.1% (95% CI 25.2–46.1%), respectively. Clinically relevant HOIs (HOI-2 and HOI-3) were found in 35/45 (77.8%) and 54/64 (84.4%) cases of well- and de-differentiated liposarcomas, respectively. On multivariable survival analysis, patients with HOI-3 had significantly shorter OS (HOI-3 vs HOI-0/HOI-1 HR 2.92; p = 0.012) and DFS (HOI-3 vs HOI-0/HOI-1 HR 2.23; p = 0.045), independently of the nomogram score (OS: HR 2.93; p < 0.001; DFS: HR 1.78; p = 0.003).
Conclusions
Initial and advanced HOIs are frequently detected in both well-differentiated and de-differentiated liposarcomas, supporting that multivisceral resection may be needed. HOI stratifies the risk of patients with primary retroperitoneal liposarcoma.



Similar content being viewed by others
Data availability
All relevant data are within the paper and its Supplementary Digital Content (SDC).
References
WHO Classification of Tumours Editorial Board. Soft tissue and bone tumours: International Agency for Research on Cancer. 2020.
Mussi C, Colombo P, Bertuzzi A, et al. Retroperitoneal sarcoma: is it time to change the surgical policy? Ann Surg Oncol. 2011;18:2136–42. https://doi.org/10.1245/s10434-011-1742-z.
Russo P, Kim Y, Ravindran S, Huang W, Brennan MF. Nephrectomy during operative management of retroperitoneal sarcoma. Ann Surg Oncol. 1997;4:421–4. https://doi.org/10.1007/BF02305556.
Toulmonde M, Bonvalot S, Méeus P, et al. Retroperitoneal sarcomas: patterns of care at diagnosis, prognostic factors and focus on main histological subtypes: a multicenter analysis of the French Sarcoma Group. Ann Oncol. 2014;25:735–42.
Pasquali S, Vohra R, Tsimopoulou I, Vijayan D, Gourevitch D, Desai A. Outcomes following extended surgery for retroperitoneal sarcomas: results from a UK referral centre. Ann Surg Oncol. 2015;22:3550–6.
Fairweather M, Wang J, Jo VY, Baldini EH, Bertagnolli MM, Raut CP. Incidence and adverse prognostic implications of histopathologic organ invasion in primary retroperitoneal sarcoma. J Am Coll Surg. 2017;224:876–83.
Fairweather M, Wang J, Jo VY, Baldini EH, Bertagnolli MM, Raut CP. Surgical management of primary retroperitoneal sarcomas: rationale for selective organ resection. Ann Surg Oncol. 2018;25:98–106. https://doi.org/10.1245/s10434-017-6136-4.
Wang Z, Wu J, Lv A, Li C, Li Z, Zhao M, Hao C. Infiltration characteristics and influencing factors of retroperitoneal liposarcoma: novel evidence for extended surgery and a tumor grading system. BioSci Trends. 2018;12:185–92.
Strauss DC, Renne SL, Gronchi A. Adjacent, adherent, invaded: a spectrum of biologic aggressiveness rather than a rationale for selecting organ resection in surgery of primary retroperitoneal sarcomas. Ann Surg Oncol. 2018;25:13–6. https://doi.org/10.1245/s10434-017-6137-3.
Tyler R, Lee M, Ierodiakonou V, et al. Prognostic implications of histological organ involvement in retroperitoneal sarcoma. BJS Open. 2021;5:zrab080. https://doi.org/10.1093/bjsopen/zrab080.
Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, Strobe Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg. 2014;12:1495–9.
Bonvalot S, Raut CP, Pollock RE, et al. Technical considerations in surgery for retroperitoneal sarcomas: position paper from E-Surge, a master class in sarcoma surgery, and EORTC-STBSG. Ann Surg Oncol. 2012;19:2981–91. https://doi.org/10.1245/s10434-012-2342-2.
Callegaro D, Fiore M, Gronchi A. Personalizing surgical margins in retroperitoneal sarcomas. Expert Rev Anticancer Ther. 2015;15:553–67.
Dingley B, Fiore M, Gronchi A. Personalizing surgical margins in retroperitoneal sarcomas: an update. Expert Rev Anticancer Ther. 2019;19:613–31.
Swallow CJ, Strauss DC, Bonvalot S, et al. Management of primary retroperitoneal sarcoma (rps) in the adult: an updated consensus approach from the Transatlantic Australasian RPS Working Group. Ann Surg Oncol. 2021;28:7873–88. https://doi.org/10.1245/s10434-021-09654-z.
Ray-Coquard I, Thiesse P, Ranchère-Vince D, et al. Conformity to clinical practice guidelines, multidisciplinary management and outcome of treatment for soft tissue sarcomas. Ann Oncol. 2004;15:307–15.
Choi JH, Ro JY. Retroperitoneal sarcomas: an update on the diagnostic pathology approach. Diagnostics. 2020;10:642.
Gronchi A, Miceli R, Shurell E, et al. Outcome prediction in primary resected retroperitoneal soft tissue sarcoma: histology-specific overall survival and disease-free survival nomograms built on major sarcoma center data sets. J Clin Oncol. 2013;31:1649–55.
Callegaro D, Miceli R, Gladdy RA. Prognostic models for RPS patients—attempting to predict patient outcomes. J Surg Oncol. 2018;117:69–78.
Cates JM. The AJCC 8th edition staging system for soft tissue sarcoma of the extremities or trunk: a cohort study of the SEER database. J Natl Compr Canc Netw. 2018;16:144–52.
Marbac M, Sedki M. Variable selection for model-based clustering using the integrated complete-data likelihood. Stat Comput. 2017;27:1049–63.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81.
Marubini E, Valsecchi MG. Analysing survival data from clinical trials and observational studies. New York: Wiley; 2004.
Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988;16:1141–54.
Dindo D. The Clavien–Dindo classification of surgical complications. In: Anonymous treatment of postoperative complications after digestive surgery. Springer; 2014. p. 13–7.
Bonvalot S, Gaignard E, Stoeckle E, et al. Survival benefit of the surgical management of retroperitoneal sarcoma in a reference center: a nationwide study of the French Sarcoma Group from the NetSarc database. Ann Surg Oncol. 2019;26:2286–93. https://doi.org/10.1245/s10434-019-07421-9.
Pasquali S, Bonvalot S, Tzanis D, Casali PG, Trama A, Gronchi A, RARECARENet Working Group. Treatment challenges in and outside a network setting: soft tissue sarcomas. Eur J Surg Oncol. 2019;45:31–9.
MacNeill AJ, Gronchi A, Miceli R, et al. Postoperative morbidity after radical resection of primary retroperitoneal sarcoma: a report from the Transatlantic RPS Working Group. Ann Surg. 2018;267:959–64.
Bonvalot S, Miceli R, Berselli M, et al. Aggressive surgery in retroperitoneal soft tissue sarcoma carried out at high-volume centers is safe and is associated with improved local control. Ann Surg Oncol. 2010;17:1507–14. https://doi.org/10.1245/s10434-010-1057-5.
van Houdt WJ, Raut CP, Bonvalot S, Swallow CJ, Haas R, Gronchi A. New research strategies in retroperitoneal sarcoma. The case of TARPSWG, STRASS and RESAR: making progress through collaboration. Curr Opin Oncol. 2019;31:310–6.
Callegaro D, Raut CP, Swallow CJ, Gronchi A. Retroperitoneal sarcoma: the Transatlantic Australasian Retroperitoneal Sarcoma Working Group Program. Curr Opin Oncol. 2021;33:301–8.
Kasper B, Baldi GG, Loong HHF, Trent J. EJSO educational special issue from the TARPSWG-Standard medical treatment and new options in retroperitoneal sarcoma. Eur J Surg Oncol. 2021. https://doi.org/10.1016/j.ejso.2021.12.465
Colombo C, Randall RL, Andtbacka RH, Gronchi A. Surgery in soft tissue sarcoma: more conservative in extremities, more extended in the retroperitoneum. Expert RevAnticancer Ther. 2012;12:1079–87.
Baldini EH. The conundrum of retroperitoneal liposarcomadto be more aggressive or less aggressive. Int J Radiat Oncol Biol Phys. 2017;98:269–70.
Gronchi A, Miceli R, Colombo C, et al. Frontline extended surgery is associated with improved survival in retroperitoneal low- to intermediate-grade soft tissue sarcomas. Ann Oncol. 2012;23:1067–73.
Bonvalot S, Gronchi A, Le Péchoux C, et al. Preoperative radiotherapy plus surgery versus surgery alone for patients with primary retroperitoneal sarcoma (EORTC-62092: STRASS): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21:1366–77.
Gronchi A, Miceli R, Allard MA, et al. Personalizing the approach to retroperitoneal soft tissue sarcoma: histology-specific patterns of failure and postrelapse outcome after primary extended resection. Ann Surg Oncol. 2015;22:1447–54. https://doi.org/10.1245/s10434-014-4130-7.
Tan MC, Brennan MF, Kuk D, et al. Histology-based classification predicts pattern of recurrence and improves risk stratification in primary retroperitoneal sarcoma. Ann Surg. 2016;263:593.
Gronchi A, Strauss DC, Miceli R, et al. Variability in patterns of recurrence after resection of primary retroperitoneal sarcoma (RPS): a report on 1007 patients from the Multi-institutional Collaborative RPS Working Group. Ann Surg. 2016;263:1002–9.
Funding
This study was supported by the following research grants: “5x1000 Founds” – 2016, Italian Ministry of Health – Institutional Grant BRI2017 from Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, GR-2019-12369175, FRRB-584-797, 60, and International Accelerator Award funded by AIRC [ID #24297]/Cancer Research UK [C56167/A29363]/Fundacion Científica, Asociacion Espanola Contra el Cancer [Foundation AECC-GEACC19007MA]. L.I. research is supported by Fondazione Policlinico Universitario Campus Bio-Medico (Sarcoma Research Fund).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
A.G. received compensation for Ad Boards from Novartis, Bayer, Lilly, PharmaMar, and SpringWorks, as well as honoraria from PharmaMar and Deciphera and research grants from PharmaMar and Nanobiotix. The other authors have declared that no competing interests exist.
Ethical approval
The study was approved by the Ethics Committee of Fondazione IRCCS Istituto Nazionale Tumori, Milan, Italy (INT 88/17).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Improta, L., Pasquali, S., Iadecola, S. et al. Organ Infiltration and Patient Risk After Multivisceral Surgery for Primary Retroperitoneal Liposarcomas. Ann Surg Oncol 30, 4500–4510 (2023). https://doi.org/10.1245/s10434-023-13314-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-13314-9