Abstract
Background
Appendiceal cancer with peritoneal metastases (ACPM) is a complex disease requiring multidisciplinary care. Cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion (CRS HIPEC) can significantly improve survival but requires evaluation by a surgical oncologist and significant treatment endurance. The impacts of socioeconomic status (SES) and other social determinants of health on rates of surgical evaluation and treatment have not been examined.
Methods
We conducted a retrospective cohort study examining all patients with ACPM from 2010 to 2018 in a regional healthcare system. Patient characteristics, oncologic details, treatment strategies, and survival were examined. The primary outcomes of interest were referral to Surgical Oncology, receipt of CRS HIPEC, and survival.
Results
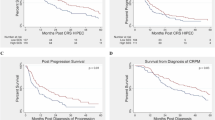
Of 194 patients identified, 94% had synchronous ACPM. The majority of patients (95%) were referred to surgical oncology. Advanced age was the only predictor of nonreferral (p < 0.001). A total of 147 patients (76%) ultimately underwent CRS HIPEC. After adjusting for medical and tumor characteristics, CRS HIPEC was less likely for patients who were unmarried [odds ratio (OR) 0.253, p = 0.004] or of low SES (OR 0.372, p = 0.03). On subanalysis of patients undergoing CRS HIPEC, median overall survival was worse for patients of low SES [51 months versus not reached (NR), p = 0.05], and this disparity persisted on multivariate analysis [hazard ratio (HR) = 2.278, p = 0.001].
Conclusions
This analysis is the first to evaluate barriers to CRS HIPEC for ACPM. While most patients were evaluated by a multidisciplinary team, nonmedical factors may play a role in the treatment received and ultimate outcomes. Addressing these disparities is crucial for ensuring equitable outcomes and improving patient care.



Similar content being viewed by others
References
Kirkegård J, Ladekarl M, Fristrup CW, Hansen CP, Sall M, Mortensen FV. Urban versus rural residency and pancreatic cancer survival: a Danish nationwide population-based cohort study. PLoS One. 2018;13(8):e0202486. https://doi.org/10.1371/journal.pone.0202486.
Lee RM, Liu Y, Gamboa AC, et al. Race, ethnicity, and socioeconomic factors in cholangiocarcinoma: what is driving disparities in receipt of treatment? J Surg Oncol. 2019;120(4):611–23. https://doi.org/10.1002/jso.25632.
Lutfi W, Zenati MS, Zureikat AH, Zeh HJ, Hogg ME. Health disparities impact expected treatment of pancreatic ductal adenocarcinoma nationally. Ann Surg Oncol. 2018;25(7):1860–7. https://doi.org/10.1245/s10434-018-6487-5.
Shapiro M, Chen Q, Huang Q, et al. Associations of socioeconomic variables with resection, stage, and survival in patients with early-stage pancreatic xancer. JAMA Surg. 2016;151(4):338–45. https://doi.org/10.1001/jamasurg.2015.4239.
Hoehn RS, Rieser CJ, Winters S, et al. A pancreatic cancer multidisciplinary clinic eliminates socioeconomic disparities in treatment and improves survival. Ann Surg Oncol. Feb 1 2021;doi:https://doi.org/10.1245/s10434-021-09594-8.
Swords DS, Mulvihill SJ, Brooke BS, Firpo MA, Scaife CL. Size and importance of socioeconomic status-based disparities in use of surgery in nonadvanced stage gastrointestinal cancers.
van Roest MH, van der Aa MA, van der Geest LG, de Jong KP. The impact of socioeconomic status, surgical resection and type of hospital on survival in patients with pancreatic cancer. A population-based study in the Netherlands. PLoS One. 2016;11(11):e0166449. doi:https://doi.org/10.1371/journal.pone.0166449.
Rieser CJ, Hoehn RS, Zenati M, et al. Impact of socioeconomic status on presentation and outcomes in colorectal peritoneal metastases following cytoreduction and chemoperfusion: persistent inequalities in outcomes at a high-volume center. Ann Surg Oncol. Mar 9 2021;doi:https://doi.org/10.1245/s10434-021-09627-2.
Miura J, Karakousis G. Improving access to specialized centers is not enough to mitigate socioeconomic disparities in complex oncologic surgery. Ann Surg Oncol. Feb 6 2021;doi:https://doi.org/10.1245/s10434-021-09631-6.
Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–51. https://doi.org/10.1016/0895-4356(94)90129-5.
Sugarbaker PH. Management of peritoneal-surface malignancy: the surgeon’s role. Langenbecks Arch Surg. 1999;384(6):576–87. https://doi.org/10.1007/s004230050246.
Slankamenac K, Graf R, Barkun J, Puhan MA, Clavien PA. The comprehensive complication index: a novel continuous scale to measure surgical morbidity. Ann Surg. 2013;258(1):1–7. https://doi.org/10.1097/SLA.0b013e318296c732.
Health UoWSoMaP. Area Deprivation Index 2.0. Downloaded from https://www.neighborhoodatlas.medicine.wisc.edu/ (accessed 3/1/2021).
Mora J, Krepline AN, Aldakkak M, et al. Adjuvant therapy rates and overall survival in patients with localized pancreatic cancer from high Area Deprivation Index neighborhoods. Am J Surg. Dec 3 2020;doi:https://doi.org/10.1016/j.amjsurg.2020.12.001.
Arias F, Chen F, Fong TG, et al. Neighborhood-level social disadvantage and risk of delirium following major surgery. J Am Geriatr Soc. 2020;68(12):2863–71. https://doi.org/10.1111/jgs.16782.
Swords DS, Mulvihill SJ, Brooke BS, Firpo MA, Scaife CL. Size and importance of socioeconomic status-based disparities in use of surgery in nonadvanced stage gastrointestinal cancers. Ann Surg Oncol. 2020;27(2):333–41. https://doi.org/10.1245/s10434-019-07922-7.
Tohme S, Kaltenmeier C, Bou-Samra P, Varley PR, Tsung A. Race and health disparities in patient refusal of surgery for early-stage pancreatic cancer: an NCDB cohort study. Ann Surg Oncol. 2018;25(12):3427–35. https://doi.org/10.1245/s10434-018-6680-6.
Kaltenmeier C, Malik J, Yazdani H, et al. Refusal of cancer-directed treatment by colon cancer patients: risk factors and survival outcomes. Am J Surg. 2020;220(6):1605–12. https://doi.org/10.1016/j.amjsurg.2020.04.022.
Votanopoulos KI, Bartlett D, Moran B, et al. PCI is not predictive of survival after complete CRS/HIPEC in peritoneal dissemination from high-grade appendiceal primaries. Ann Surg Oncol. 2018;25(3):674–8. https://doi.org/10.1245/s10434-017-6315-3.
Levinsky NC, Morris MC, Wima K, et al. Should we be doing cytoreductive surgery with HIPEC for signet ring cell appendiceal adenocarcinoma? A study from the US HIPEC Collaborative. J Gastrointest Surg. 2020;24(1):155–64. https://doi.org/10.1007/s11605-019-04336-4.
Munoz-Zuluaga CA, King MC, Diaz-Sarmiento VS, et al. Defining “complete cytoreduction” after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) for the histopathologic spectrum of appendiceal carcinomatosis. Ann Surg Oncol. 2020;27(13):5026–36. https://doi.org/10.1245/s10434-020-08844-5.
Acknowledgements
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under Award Number T32CA113263. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest for the authors of this manuscript.
Data Sharing and Data Accessibility
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval Statement
This study was approved by our institutional review board (IRB20100475).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rieser, C., Phelos, H., Zureikat, A. et al. Socioeconomic Barriers to CRS HIPEC for Appendiceal Cancer within a Regional Academic Hospital System. Ann Surg Oncol 29, 6593–6602 (2022). https://doi.org/10.1245/s10434-022-11949-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11949-8