Abstract
Introduction
Liver steatosis (LS) has been increasingly described in preoperative imaging of patients undergoing pancreaticoduodenectomy (PD). The aim of this study was to assess the impact of preoperative LS on complications after PD and identify possible contributors to LS development in this specific cohort.
Methods
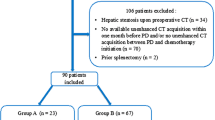
Pancreatic head adenocarcinoma (PDAC) patients scheduled for PD, with preoperative CT-imaging available were included in the study. LS was defined as mean liver density lower than 45 Hounsfield units. Patients showing preoperative LS were matched for patient age, gender, BMI, ASA score, neoadjuvant treatment, and vascular and multivisceral resections, based on propensity scores in a 1:2 ratio to patients with no LS. The primary outcome was postoperative complication severity at 90 days as measured by the comprehensive complication index (CCI)
Results
Overall, 247 patients were included in the study. Forty-three (17%) patients presented with LS at preoperative CT-scan. After matching, the LS group included 37 patients, whereas the non-LS group had 74 patients. LS patients had a higher mean (SD) CCI, 29.7 (24.5) versus 19.5 (22.5), p = 0.035, and a longer length of hospital stay, median [IQR] 12 [8–26] versus 8 [7–13] days, p = 0.006 compared with non-LS patients. On multivariate analysis, variables independently associated with CCI were: LS (16% increase, p = 0.048), male sex (19% increase, p = 0.030), ASA score ≥ 3 (26% increase, p = 0.002), fistula risk score (FRS) (28% increase for each point of FRS, p = 0.001) and vascular resection (20% increase, p = 0.019).
Conclusion
Preliminary evidence suggests that preoperative LS assessed by CT-scan influences complication severity in patients undergoing PD for PDAC.


Similar content being viewed by others
References
Brown EG, Yang A, Canter RJ, Bold RJ. Outcomes of pancreaticoduodenectomy: Where should we focus our efforts on improving outcomes? JAMA Surg. 2014. https://doi.org/10.1001/JAMASURG.2014.151.
Palumbo D, Tamburrino D, Partelli S, et al. Before sentinel bleeding: early prediction of postpancreatectomy hemorrhage (PPH) with a CT-based scoring system. Eur Radiol. 2021. https://doi.org/10.1007/S00330-021-07788-Y.
Uzunoglu FG, Reeh M, Vettorazzi E, et al. Preoperative Pancreatic Resection (PREPARE) score: a prospective multicenter-based morbidity risk score. Ann Surg. 2014. https://doi.org/10.1097/SLA.0000000000000946.
Junejo MA, Mason JM, Sheen AJ, et al. Cardiopulmonary exercise testing for preoperative risk assessment before pancreaticoduodenectomy for cancer. Ann Surg Oncol. 2014. https://doi.org/10.1245/S10434-014-3493-0.
Lovasik BP, Kron P, Clavien PA, Petrowsky H, Kooby DA. Pancreatectomy and body mass index: an international evaluation of cumulative postoperative complications using the comprehensive complications index. HPB (Oxford). 2019. https://doi.org/10.1016/J.HPB.2019.04.006.
Mungroop TH, Van Rijssen LB, Van Klaveren D, et al. Alternative Fistula Risk Score for Pancreatoduodenectomy (a-FRS): Design and International External Validation. Ann Surg. 2019. https://doi.org/10.1097/SLA.0000000000002620.
Chu CK, Mazo AE, Goodman M, et al. Preoperative diabetes mellitus and long-term survival after resection of pancreatic adenocarcinoma. Ann Surg Oncol. 2010. https://doi.org/10.1245/S10434-009-0789-6.
Gangi A, Lu SC. Chemotherapy-associated liver injury in colorectal cancer. Therap Adv Gastroenterol. 2020. https://doi.org/10.1177/1756284820924194.
De Meijer VE, Kalish BT, Puder M, IJzermans JNM. Systematic review and meta-analysis of steatosis as a risk factor in major hepatic resection. Br J Surg. 2010. https://doi.org/10.1002/BJS.7194.
Fagenson AM, Pitt HA, Moten AS, Karhadkar SS, Di Carlo A, Lau KN. Fatty liver: The metabolic syndrome increases major hepatectomy mortality. Surgery. 2021. https://doi.org/10.1016/J.SURG.2020.11.021.
Hashimoto Y, Sclabas GM, Takahashi N, et al. Dual-phase computed tomography for assessment of pancreatic fibrosis and anastomotic failure risk following pancreatoduodenectomy. J Gastrointest Surg. 2011. https://doi.org/10.1007/S11605-011-1687-3.
Ohgi K, Okamura Y, Sugiura T, et al. Pancreatic attenuation on computed tomography predicts pancreatic fistula after pancreaticoduodenectomy. HPB (Oxford). 2020. https://doi.org/10.1016/J.HPB.2019.05.008.
Kirihara Y, Takahashi N, Hashimoto Y, et al. Prediction of pancreatic anastomotic failure after pancreatoduodenectomy: The use of preoperative, quantitative computed tomography to measure remnant pancreatic volume and body composition. Ann Surg. 2013. https://doi.org/10.1097/SLA.0b013e31827827d0.
Jang M, Park HW, Huh J, et al. Predictive value of sarcopenia and visceral obesity for postoperative pancreatic fistula after pancreaticoduodenectomy analyzed on clinically acquired CT and MRI. Eur Radiol. 2019. https://doi.org/10.1007/S00330-018-5790-7.
Pecorelli N, Carrara G, De Cobelli F, et al. Effect of sarcopenia and visceral obesity on mortality and pancreatic fistula following pancreatic cancer surgery. Br J Surg. 2016. https://doi.org/10.1002/BJS.10063.
Xingjun G, Feng Z, Meiwen Y, et al. A score model based on pancreatic steatosis and fibrosis and pancreatic duct diameter to predict postoperative pancreatic fistula after pancreatoduodenectomy. BMC Surg. 2019. https://doi.org/10.1186/S12893-019-0534-4.
Flick KF, Al-Temimi MH, Maatman TK, et al. Hepatic steatosis after neoadjuvant chemotherapy for pancreatic cancer: incidence and implications for outcomes after pancreatoduodenectomy. J Gastrointest Surg. 2020. https://doi.org/10.1007/S11605-020-04723-2.
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg. 2014. https://doi.org/10.1016/j.ijsu.2014.07.013.
Graffy PM, Pickhardt PJ. Quantification of hepatic and visceral fat by CT and MR imaging: relevance to the obesity epidemic, metabolic syndrome and NAFLD. Br J Radiol. 2016. https://doi.org/10.1259/BJR.20151024.
Balzano G, Guarneri G, Pecorelli N, et al. Modelling centralization of pancreatic surgery in a nationwide analysis. Br J Surg. 2020. https://doi.org/10.1002/bjs.11716.
Pecorelli N, Nobile S, Partelli S, et al. Enhanced recovery pathways in pancreatic surgery: State of the art. World J Gastroenterol. 2016. https://doi.org/10.3748/wjg.v22.i28.6456.
Braga M, Pecorelli N, Ariotti R, et al. Enhanced recovery after surgery pathway in patients undergoing pancreaticoduodenectomy. World J Surg. 2014. https://doi.org/10.1007/s00268-014-2653-5.
Slankamenac K, Graf R, Barkun J, Puhan MA, Clavien PA. The comprehensive complication index: a novel continuous scale to measure surgical morbidity. Ann Surg. 2013. https://doi.org/10.1097/SLA.0b013e318296c732.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004. https://doi.org/10.1097/01.sla.0000133083.54934.ae.
Bassi C, Marchegiani G, Dervenis C, et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surg (United States). 2017. https://doi.org/10.1016/j.surg.2016.11.014.
Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery. 2007. https://doi.org/10.1016/j.surg.2007.05.005.
Wente MN, Veit JA, Bassi C, et al. Postpancreatectomy hemorrhage (PPH): an International Study Group of Pancreatic Surgery (ISGPS) definition. Surgery. 2007. https://doi.org/10.1016/j.surg.2007.02.001.
Berríos-Torres SI, Umscheid CA, Bratzler DW, et al. Centers for disease control and prevention guideline for the prevention of surgical site infection, 2017. JAMA Surg. 2017. https://doi.org/10.1001/JAMASURG.2017.0904.
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016. https://doi.org/10.1002/HEP.28431.
Dash A, Figler RA, Sanyal AJ, Wamhoff BR. Drug-induced steatohepatitis. Expert Opin Drug Metab Toxicol. 2017. https://doi.org/10.1080/17425255.2017.1246534.
Robinson SM, Wilson CH, Burt AD, Manas DM, White SA. Chemotherapy-associated liver injury in patients with colorectal liver metastases: a systematic review and meta-analysis. Ann Surg Oncol. 2012. https://doi.org/10.1245/S10434-012-2438-8.
White MA, Fong Y, Singh G. Chemotherapy-associated hepatotoxicities. Surg Clin North Am. 2016. https://doi.org/10.1016/J.SUC.2015.11.005.
McCormack L, Petrowsky H, Jochum W, Furrer K, Clavien PA. Hepatic steatosis is a risk factor for postoperative complications after major hepatectomy: a matched case-control study. Ann Surg. 2007. https://doi.org/10.1097/01.SLA.0000251747.80025.B7.
Zhou L, Xiao W, Li C, Gao Y, Gong W, Lu G. Impact of fatty pancreas on postoperative pancreatic fistulae: a meta-analysis. Front Oncol. 2021. https://doi.org/10.3389/FONC.2021.622282.
Partelli S, Andreasi V, Schiavo Lena M, et al. The role of acinar content at pancreatic resection margin in the development of postoperative pancreatic fistula and acute pancreatitis after pancreaticoduodenectomy. Surgery. 2021. https://doi.org/10.1016/j.surg.2021.03.047.
Mathur A, Pitt HA, Marine M, et al. Fatty pancreas: a factor in postoperative pancreatic fistula. Ann Surg. 2007. https://doi.org/10.1097/SLA.0B013E31814A6906.
Den Boer M, Voshol PJ, Kuipers F, Havekes LM, Romijn JA. Hepatic steatosis: a mediator of the metabolic syndrome. Lessons from animal models. Arterioscler Thromb Vasc Biol. 2004. https://doi.org/10.1161/01.ATV.0000116217.57583.6E.
Pickhardt PJ, Jee Y, O’Connor SD, Del Rio AM. Visceral adiposity and hepatic steatosis at abdominal CT: association with the metabolic syndrome. Am J Roentgenol. 2012. https://doi.org/10.2214/AJR.11.7361.
Taipale T, Seppälä I, Raitoharju E, et al. Fatty liver is associated with blood pathways of inflammatory response, immune system activation and prothrombotic state in Young Finns Study. Sci Rep. 2018. https://doi.org/10.1038/S41598-018-28563-Y.
Kurmann A, Wanner B, Martens F, et al. Hepatic steatosis is associated with surgical-site infection after hepatic and colorectal surgery. Surgery. 2014. https://doi.org/10.1016/J.SURG.2014.02.020.
Romeijn MM, Kolen AM, Holthuijsen DDB, et al. Effectiveness of a low-calorie diet for liver volume reduction prior to bariatric surgery: a systematic review. Obes Surg. 2021. https://doi.org/10.1007/S11695-020-05070-6.
Doherty DT, Coe PO, Rimmer L, Lapsia S, Krige A, Subar DA. Hepatic steatosis in patients undergoing resection of colorectal liver metastases: a target for prehabilitation? A narrative review. Surg Oncol. 2019. https://doi.org/10.1016/J.SURONC.2019.07.007.
Armstrong MJ, Gaunt P, Aithal GP, et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet. 2016. https://doi.org/10.1016/S0140-6736(15)00803-X.
Barth RJ, Mills JB, Suriawinata AA, et al. Short-term preoperative diet decreases bleeding after partial hepatectomy: results from a multi-institutional randomized controlled trial. Ann Surg. 2019. https://doi.org/10.1097/SLA.0000000000002709.
Waterland JL, McCourt O, Edbrooke L, et al. Efficacy of prehabilitation including exercise on postoperative outcomes following abdominal cancer surgery: a systematic review and meta-analysis. Front Surg. 2021. https://doi.org/10.3389/FSURG.2021.628848.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987. https://doi.org/10.1016/0021-9681(87)90171-8.
Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg. 1973. https://doi.org/10.1002/bjs.1800600817.
Acknowledgments
The authors are grateful to Fondazione Umberto Veronesi for supporting Dr. Guarneri’s research fellowship. The authors are grateful to Dr. Alice Wong Licinio for her English language review and editing.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Giovanni Guarneri’s research fellowship, unrelated to this study, was funded by Fondazione Umberto Veronesi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors have no related conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10434_2022_11946_MOESM2_ESM.tif
Supplementary Disease free survival (DFS) in patients with preoperative LS and in those without preoperative LS. Median DFS was 20 months for patients without preoperative LS compared with 14 months of those with preoperative LS (p=0.147) (TIF 157 KB)
Rights and permissions
About this article
Cite this article
Guarneri, G., Palumbo, D., Pecorelli, N. et al. The Impact of CT-Assessed Liver Steatosis on Postoperative Complications After Pancreaticoduodenectomy for Cancer. Ann Surg Oncol 29, 7063–7073 (2022). https://doi.org/10.1245/s10434-022-11946-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11946-x



