Abstract
Introduction
The safety and efficacy of cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) in peritoneal metastasis in palliative settings remain poorly investigated and understood. Chemotherapy-refractory patients often present with symptomatic disease. This study investigated the safety and survival outcomes of optimal CRS/HIPEC performed primarily for palliation.
Methods
Palliative CRS/HIPEC was defined as asymptomatic patients who did not respond to three or more lines of chemotherapy, progression on current chemotherapy, and/or any symptomatic disease progression, including ascites, bowel obstruction, and pain. Data collected included demographics, histology, length of stay (LOS), perioperative complications, perioperative mortality, adjuvant chemotherapy use, peritoneal recurrence, overall recurrence, and overall survival.
Results
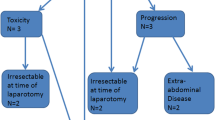
The median number of lines of chemotherapy received prior to CRS/HIPEC was 3.2, and 81% of patients were symptomatic. There were no postoperative deaths and the major complication rate was 22%. Ostomy creation and abdominal wall reconstruction were performed in 24% and 21% of patients, respectively. The median LOS was 11 days and successful palliation was achieved in 97% of patients. Overall survival was 13.5 months and factors associated with prolonged survival included optimal CRS (R1/R2a; p < 0.01) and the use of adjuvant chemotherapy (p < 0.001). Synchronous liver metastasis in the colon cancer subset did not negatively impact survival.
Conclusion
CRS/HIPEC was performed safely in the palliative setting in patients with symptomatic progressive disease receiving multiple lines of chemotherapy. Median survival exceeded 1 year and factors associated with longer survival were optimal CRS and adjuvant chemotherapy. Liver metastasis did not preclude survival benefit in colon cancer patients. CRS/HIPEC can be considered for palliation but should be performed at high-volume centers.


Similar content being viewed by others
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Morano WF, Khalili M, Chi DS, Bowne WB, Esquivel J. Clinical studies in CRS and HIPEC: trials, tribulations, and future directions: a systematic review. J Surg Oncol. 2018;117(2):245–59.
Verwaal VJ, van Ruth S, de Bree E, et al. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J Clin Oncol. 2003;21(20):3737–43.
Verma V, Sleightholm RL, Rusthoven CG, et al. Malignant peritoneal mesothelioma: national practice patterns, outcomes, and predictors of survival. Ann Surg Oncol. 2018;25(7):2018–26.
Spiliotis J, Halkia E, Lianos E, et al. Cytoreductive surgery and HIPEC in recurrent epithelial ovarian cancer: a prospective randomized phase III study. Ann Surg Oncol. 2015;22(5):1570–5.
Terzi C, Arslan NC, Canda AE. Peritoneal carcinomatosis of gastrointestinal tumors: Where are we now? World J Gastroenterol. 2014;20(39):14371–80.
Sadeghi B, Arvieux C, Glehen O, et al. Peritoneal carcinomatosis from non-gynecologic malignancies: results of the EVOCAPE 1 multicentric prospective study. Cancer. 2000;88(2):358–63.
Quenet F, Elias D, Roca L, et al. Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy versus cytoreductive surgery alone for colorectal peritoneal metastases (PRODIGE 7): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(2):256–66.
Bakrin N, Classe JM, Pomel C, Gouy S, Chene G, Glehen O. Hyperthermic intraperitoneal chemotherapy (HIPEC) in ovarian cancer. J Visc Surg. 2014;151(5):347–53.
Davies JM, O’Neil B. Peritoneal carcinomatosis of gastrointestinal origin: natural history and treatment options. Expert Opin Investig Drugs. 2009;18(7):913–9.
Jayne DG, Fook S, Loi C, Seow-Choen F. Peritoneal carcinomatosis from colorectal cancer. Br J Surg. 2002;89(12):1545–50.
Chu DZ, Lang NP, Thompson C, Osteen PK, Westbrook KC. Peritoneal carcinomatosis in nongynecologic malignancy. A prospective study of prognostic factors. Cancer. 1989;63(2):364–7.
Glehen O, Osinsky D, Beaujard AC, Gilly FN. Natural history of peritoneal carcinomatosis from nongynecologic malignancies. Surg Oncol Clin N Am. 2003;12(3):729–39.
Lambert LA, Wiseman J. Palliative management of peritoneal metastases. Ann Surg Oncol. 2018;25(8):2165–71.
Averbach AM, Sugarbaker PH. Recurrent intraabdominal cancer with intestinal obstruction. Int Surg. 1995;80(2):141–6.
Blair SL, Chu DZ, Schwarz RE. Outcome of palliative operations for malignant bowel obstruction in patients with peritoneal carcinomatosis from nongynecological cancer. Ann Surg Oncol. 2001;8(8):632–7.
Smeenk RM, Verwaal VJ, Zoetmulder FA. Learning curve of combined modality treatment in peritoneal surface disease. Br J Surg. 2007;94(11):1408–14.
Kusamura S, Baratti D, Deraco M. Multidimensional analysis of the learning curve for cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in peritoneal surface malignancies. Ann Surg. 2012;255(2):348–56.
Andreasson H, Lorant T, Pahlman L, Graf W, Mahteme H. Cytoreductive surgery plus perioperative intraperitoneal chemotherapy in pseudomyxoma peritonei: aspects of the learning curve. Eur J Surg Oncol. 2014;40(8):930–6.
Spiliotis J, Kalles V, Kyriazanos I, et al. CRS and HIPEC in patients with peritoneal metastasis secondary to colorectal cancer: the small-bowel PCI score as a predictor of survival. Pleura Peritoneum. 2019;4(4):20190018.
Elias D, Mariani A, Cloutier AS, et al. Modified selection criteria for complete cytoreductive surgery plus HIPEC based on peritoneal cancer index and small bowel involvement for peritoneal carcinomatosis of colorectal origin. Eur J Surg Oncol. 2014;40(11):1467–73.
Benizri EI, Bernard JL, Rahili A, Benchimol D, Bereder JM. Small bowel involvement is a prognostic factor in colorectal carcinomatosis treated with complete cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy. World J Surg Oncol. 2012;10:56.
Chandramohan A, Thrower A, Smith SA, Shah N, Moran B. “PAUSE”: a method for communicating radiological extent of peritoneal malignancy. Clin Radiol. 2017;72(11):972–80.
Randle RW, Swett KR, Swords DS, et al. Efficacy of cytoreductive surgery with hyperthermic intraperitoneal chemotherapy in the management of malignant ascites. Ann Surg Oncol. 2014;21(5):1474–9.
Valle SJ, Alzahrani NA, Alzahrani SE, Liauw W, Morris DL. Laparoscopic hyperthermic intraperitoneal chemotherapy (HIPEC) for refractory malignant ascites in patients unsuitable for cytoreductive surgery. Int J Surg. 2015;23(Pt A):176–80.
Jacquet P, Jelinek JS, Chang D, Koslowe P, Sugarbaker PH. Abdominal computed tomographic scan in the selection of patients with mucinous peritoneal carcinomatosis for cytoreductive surgery. J Am Coll Surg. 1995;181(6):530–8.
Cotte E, Passot G, Gilly FN, Glehen O. Selection of patients and staging of peritoneal surface malignancies. World J Gastrointest Oncol. 2010;2(1):31–5.
Paul Olson TJ, Pinkerton C, Brasel KJ, Schwarze ML. Palliative surgery for malignant bowel obstruction from carcinomatosis: a systematic review. JAMA Surg. 2014;149(4):383–92.
Baumgartner JM, Marmor R, Hsu A, Veerapong J, Kelly KJ, Lowy AM. Obstruction-free survival following operative intervention for malignant bowel obstruction in appendiceal cancer. Ann Surg Oncol. 2019;26(11):3611–7.
Kopanakis N, Argyriou EO, Vassiliadou D, et al. Quality of life after cytoreductive surgery and HIPEC: a single centre prospective study. J BUON. 2018;23(2):488–93.
Glehen O, Kwiatkowski F, Sugarbaker PH, et al. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: a multi-institutional study. J Clin Oncol. 2004;22(16):3284–92.
Levine EA, Stewart JH 4th, Russell GB, Geisinger KR, Loggie BL, Shen P. Cytoreductive surgery and intraperitoneal hyperthermic chemotherapy for peritoneal surface malignancy: experience with 501 procedures. J Am Coll Surg. 2007;204(5):943–53 (discussion 953–945).
van Leeuwen BL, Graf W, Pahlman L, Mahteme H. Swedish experience with peritonectomy and HIPEC. HIPEC in peritoneal carcinomatosis. Ann Surg Oncol. 2008;15(3):745–53.
Shen P, Hawksworth J, Lovato J, et al. Cytoreductive surgery and intraperitoneal hyperthermic chemotherapy with mitomycin C for peritoneal carcinomatosis from nonappendiceal colorectal carcinoma. Ann Surg Oncol. 2004;11(2):178–86.
Funding
This work was supported by the Platon Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Chunmeng Zhang, Asish Patel, Dalton Hegeholz, Krista Brown, Valerie Shostrom, Mallory Pottebaum, and Jason M. Foster have no financial conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10434_2022_11323_MOESM1_ESM.tiff
Supplementary Fig. 1 Progression-free survival (PFS) by histology and by preoperative symptoms. CRC colorectal cancer, HG high grade, LG low grade, SBO small bowel obstruction (TIFF 13673 KB)
10434_2022_11323_MOESM2_ESM.tiff
Supplementary Fig. 2 Progression-free survival (PFS) by optimal CRS and adjuvant chemotherapy. CRS cytoreductive surgery (TIFF 13673 KB)
Rights and permissions
About this article
Cite this article
Zhang, C., Patel, A., Hegeholz, D. et al. Cytoreductive Surgery with HIPEC is a Safe and Effective Palliative Option in Chemorefractory Symptomatic Peritoneal Metastasis. Ann Surg Oncol 29, 3337–3346 (2022). https://doi.org/10.1245/s10434-022-11323-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11323-8