Abstract
Background
We aimed to analyze the effects of radiotherapy (RT) on the incidence rate of ipsilateral breast event (IBE) in ductal carcinoma in situ (DCIS) patients with lumpectomy after being stratified by prognostic score.
Methods
We identified DCIS patients who received lumpectomy, from the Surveillance, Epidemiology, and End Results (SEER) database from 1988 to 2015. Cumulative incidence functions for competing risk were used to evaluate the effects of RT on IBE risk over time. Three multivariate regression models (weighted, non-weighted, and Fine–Gray) were applied to compare the IBE risk between the RT and non-RT groups after stratifying patients by prognostic score.
Results
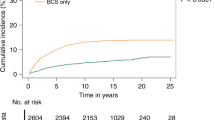
Overall, 72,623 DCIS patients were identified from the SEER database and 49,206 (66.8%) patients received RT. During the follow-up period (ranging from 7 to 347 months), the cumulative probability of invasive and in situ IBE was significantly lower in the RT group than in the non-RT group (p < 0.001). After being stratified by prognostic score, the weighted IBE incidence rate increased as the risk level increased (p < 0.050). In multivariate regression models, RT lowered the IBE incidence rate by at least 30% in low-, moderate-, and high-risk DCIS (p < 0.010). In particular, the in situ and invasive IBE incidence rate decreased by over 50% in low-risk DCIS with RT (p < 0.001).
Conclusions
RT is associated with a lowered IBE incidence rate in DCIS patients, regardless of the assigned risk levels for patients. The significant reduction in the IBE incidence rate in low-risk DCIS patients also indicates the potential benefits for recommending RT to such a patient population in clinical practice.





Similar content being viewed by others
References
Elshof LE, Schaapveld M, Rutgers EJ, et al. The method of detection of ductal carcinoma in situ has no therapeutic implications: results of a population-based cohort study. Breast Cancer Res. 2017;19(1):26.
Olivotto IA, Bancej C, Goel V, et al. Waiting times from abnormal breast screen to diagnosis in 7 Canadian provinces. CMAJ. 2001;165(3):277–83.
Armed Forces Health Surveillance Center. Incident diagnoses of breast cancer, active component service women, U.S. Armed Forces, 2000–2012. MSMR. 2013;20(9):25–7.
Oseni TO, Zhang B, Coopey SB, Gadd MA, Hughes KS, Chang DC. Twenty-five year trends in the incidence of ductal carcinoma in situ in US Women. J Am Coll Surg. 2019;228(6):932–9.
van Seijen M, Lips EH, Thompson AM, et al. Ductal carcinoma in situ: to treat or not to treat, that is the question. Br J Cancer. 2019;121(4):285–92.
Elshof LE, Schmidt MK, Rutgers EJT, van Leeuwen FE, Wesseling J, Schaapveld M. Cause-specific mortality in a population-based cohort of 9799 women treated for ductal carcinoma in situ. Ann Surg. 2018;267(5):952–8.
Koh VC, Lim JC, Thike AA, et al. Characteristics and behaviour of screen-detected ductal carcinoma in situ of the breast: comparison with symptomatic patients. Breast Cancer Res Treat. 2015;152(2):293–304.
Narod SA, Iqbal J, Giannakeas V, Sopik V, Sun P. Breast cancer mortality after a diagnosis of ductal carcinoma in situ. JAMA Oncol. 2015;1(7):888–96.
Kerlikowske K, Molinaro A, Cha I, et al. Characteristics associated with recurrence among women with ductal carcinoma in situ treated by lumpectomy. J Natl Cancer Inst. 2003;95(22):1692–702.
Ryser MD, Weaver DL, Zhao F, et al. Cancer outcomes in DCIS patients without locoregional treatment. J Natl Cancer Inst. 2019;111(9):952–60.
Sanders ME, Schuyler PA, Dupont WD, Page DL. The natural history of low-grade ductal carcinoma in situ of the breast in women treated by biopsy only revealed over 30 years of long-term follow-up. Cancer. 2005;103(12):2481–4.
Bijker N, Donker M, Wesseling J, den Heeten GJ, Rutgers EJ. Is DCIS breast cancer, and how do I treat it? Curr Treat Options Oncol. 2013;14(1):75–87.
Curigliano G, Burstein HJ, Winer EP, et al. De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen international expert consensus conference on the primary therapy of early breast cancer 2017. Ann Oncol. 2017;28(8):1700–12.
EORTC Breast Cancer Cooperative Group, EORTC Radiotherapy Group, Bijker N, et al. Breast-conserving treatment with or without radiotherapy in ductal carcinoma-in situ: ten-year results of European Organisation for Research and Treatment of Cancer randomized phase III trial 10853: a study by the EORTC Breast Cancer Cooperative Group and EORTC Radiotherapy Group. J Clin Oncol. 2006;24(21):3381–7.
Emdin SO, Granstrand B, Ringberg A, et al. SweDCIS: radiotherapy after sector resection for ductal carcinoma in situ of the breast. Results of a randomised trial in a population offered mammography screening. Acta Oncol. 2006;45(5):536–43.
Julien JP, Bijker N, Fentiman IS, et al. Radiotherapy in breast-conserving treatment for ductal carcinoma in situ: first results of the EORTC randomised phase III trial 10853 EORTC Breast Cancer Cooperative Group and EORTC Radiotherapy Group. Lancet. 2000;355(9203):528–33.
Houghton J, George WD, Cuzick J, et al. Radiotherapy and tamoxifen in women with completely excised ductal carcinoma in situ of the breast in the UK, Australia, and New Zealand: randomised controlled trial. Lancet. 2003;362(9378):95–102.
Fisher B, Dignam J, Wolmark N, et al. Lumpectomy and radiation therapy for the treatment of intraductal breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-17. J Clin Oncol. 1998;16(2):441–52.
Wapnir IL, Dignam JJ, Fisher B, et al. Long-term outcomes of invasive ipsilateral breast tumor recurrences after lumpectomy in NSABP B-17 and B-24 randomized clinical trials for DCIS. J Natl Cancer Inst. 2011;103(6):478–88.
Hughes LL, Wang M, Page DL, et al. Local excision alone without irradiation for ductal carcinoma in situ of the breast: a trial of the Eastern Cooperative Oncology Group. J Clin Oncol. 2009;27(32):5319–24.
Solin LJ, Gray R, Hughes LL, et al. Surgical excision without radiation for ductal carcinoma in situ of the breast: 12-year results from the ECOG-ACRIN E5194 study. J Clin Oncol. 2015;33(33):3938–44.
Smith GL, Smith BD, Haffty BG. Rationalization and regionalization of treatment for ductal carcinoma in situ of the breast. Int J Radiat Oncol Biol Phys. 2006;65(5):1397–403.
Sagara Y, Freedman RA, Vaz-Luis I, et al. Patient prognostic score and associations with survival improvement offered by radiotherapy after breast-conserving surgery for ductal carcinoma in situ: a population-based longitudinal cohort study. J Clin Oncol. 2016;34(11):1190–6.
National Cancer Institute: Surveillance, Epidemiology, and End Results Program. http://seer.cancer.gov/.
de Goeij MC, van Diepen M, Jager KJ, Tripepi G, Zoccali C, Dekker FW. Multiple imputation: dealing with missing data. Nephrol Dial Transplant. 2013;28(10):2415–20.
Seaman SR, White IR, Copas AJ, Li L. Combining multiple imputation and inverse-probability weighting. Biometrics. 2012;68(1):129–37.
Lin DY. Non-parametric inference for cumulative incidence functions in competing risks studies. Stat Med. 1997;16(8):901–10.
Zhang Z. Survival analysis in the presence of competing risks. Ann Transl Med. 2017;5(3):47.
Galimberti S, Sasieni P, Valsecchi MG. A weighted Kaplan–Meier estimator for matched data with application to the comparison of chemotherapy and bone-marrow transplant in leukaemia. Stat Med. 2002;21(24):3847–64.
Austin PC. The use of propensity score methods with survival or time-to-event outcomes: reporting measures of effect similar to those used in randomized experiments. Stat Med. 2014;33(7):1242–58.
Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
Goh CW, Wu J, Ding S, et al. Invasive ductal carcinoma with coexisting ductal carcinoma in situ (IDC/DCIS) versus pure invasive ductal carcinoma (IDC): a comparison of clinicopathological characteristics, molecular subtypes, and clinical outcomes. J Cancer Res Clin Oncol. 2019;145(7):1877–86.
Hoorntje LE, Schipper ME, Peeters PH, Bellot F, Storm RK, Borel Rinkes IH. The finding of invasive cancer after a preoperative diagnosis of ductal carcinoma-in situ: causes of ductal carcinoma-in situ underestimates with stereotactic 14-gauge needle biopsy. Ann Surg Oncol. 2003;10(7):748–53.
Hartmann LC, Sellers TA, Frost MH, et al. Benign breast disease and the risk of breast cancer. N Engl J Med. 2005;353(3):229–37.
Warnberg F, Yuen J, Holmberg L. Risk of subsequent invasive breast cancer after breast carcinoma in situ. Lancet. 2000;355(9205):724–5.
Burkhardt L, Grob TJ, Hermann I, et al. Gene amplification in ductal carcinoma in situ of the breast. Breast Cancer Res Treat. 2010;123(3):757–65.
Clark SE, Warwick J, Carpenter R, Bowen RL, Duffy SW, Jones JL. Molecular subtyping of DCIS: heterogeneity of breast cancer reflected in pre-invasive disease. Br J Cancer. 2011;104(1):120–7.
Cante D, Franco P, Sciacero P, et al. Hypofractionation and concomitant boost to deliver adjuvant whole-breast radiation in ductal carcinoma in situ (DCIS): a subgroup analysis of a prospective case series. Med Oncol. 2014;31(2):838.
Guenzi M, Giannelli F, Bosetti D, et al. Two different hypofractionated breast radiotherapy schedules for 113 patients with ductal carcinoma in situ: preliminary results. Anticancer Res. 2013;33(8):3503–7.
Darby SC, Ewertz M, McGale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl J Med. 2013;368(11):987–98.
Sheth GR, Cranmer LD, Smith BD, Grasso-Lebeau L, Lang JE. Radiation-induced sarcoma of the breast: a systematic review. Oncologist. 2012;17(3):405–18.
Erel E, Vlachou E, Athanasiadou M, Hassan S, Chandrasekar CR, Peart F. Management of radiation-induced sarcomas in a tertiary referral centre: a review of 25 cases. Breast. 2010;19(5):424–7.
Quadros CA, Vasconcelos A, Andrade R, et al. Good outcome after neoadjuvant chemotherapy and extended surgical resection for a large radiation-induced high-grade breast sarcoma. Int Semin Surg Oncol. 2006;3:18.
Kim KS, Chang JH, Choi N, et al. Radiation-induced sarcoma: a 15-year experience in a single large tertiary referral center. Cancer Res Treat. 2016;48(2):650–7.
Salminen SH, Wiklund T, Sampo MM, et al. Treatment and prognosis of radiation-associated breast angiosarcoma in a nationwide population. Ann Surg Oncol. 2020;27(4):1002–10.
Sagara Y, Mallory MA, Wong S, et al. Survival benefit of breast surgery for low-grade ductal carcinoma in situ: a population-based cohort study. JAMA Surg. 2015;150(8):739–45.
Fallowfield L, Francis A, Catt S, Mackenzie M, Jenkins V. Time for a low-risk DCIS trial: harnessing public and patient involvement. Lancet Oncol. 2012;13(12):1183–5.
McCormick B, Winter K, Hudis C, et al. RTOG 9804: a prospective randomized trial for good-risk ductal carcinoma in situ comparing radiotherapy with observation. J Clin Oncol. 2015;33(7):709–15.
Silverstein MJ. The University of Southern California/Van Nuys prognostic index for ductal carcinoma in situ of the breast. Am J Surg. 2003;186(4):337–43.
Rudloff U, Jacks LM, Goldberg JI, et al. Nomogram for predicting the risk of local recurrence after breast-conserving surgery for ductal carcinoma in situ. J Clin Oncol. 2010;28(23):3762–9.
Revesz E, Khan SA. What are safe margins of resection for invasive and in situ breast cancer? Oncology (Williston Park). 2011;25(10):890–5.
Wang SY, Chu H, Shamliyan T, et al. Network meta-analysis of margin threshold for women with ductal carcinoma in situ. J Natl Cancer Inst. 2012;104(7):507–16.
Solin LJ, Gray R, Baehner FL, et al. A multigene expression assay to predict local recurrence risk for ductal carcinoma in situ of the breast. J Natl Cancer Inst. 2013;105(10):701–10.
Klimov S, Miligy IM, Gertych A, et al. A whole slide image-based machine learning approach to predict ductal carcinoma in situ (DCIS) recurrence risk. Breast Cancer Res. 2019;21(1):83.
Page DL, Dupont WD, Rogers LW, Jensen RA, Schuyler PA. Continued local recurrence of carcinoma 15–25 years after a diagnosis of low grade ductal carcinoma in situ of the breast treated only by biopsy. Cancer. 1995;76(7):1197–200.
Acknowledgment
The authors would like to acknowledge Dr. Yun Wu for her assistance in study design.
Funding
This study was supported by the 1.3.5 Project for disciplines of excellence (ZYGD18012).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors declare that they have disclosed no potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, L., Lu, D., Lai, Y. et al. Prognostic Score-Based Stratification Analysis Reveals Universal Benefits of Radiotherapy on Lowering the Risk of Ipsilateral Breast Event for Ductal Carcinoma In Situ Patients with Different Risk Levels. Ann Surg Oncol 28, 975–984 (2021). https://doi.org/10.1245/s10434-020-09003-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09003-6