Abstract
Background
Adjuvant chemotherapy and/or chemoradiation [chemo(radiation)] is considered the standard of care for resected patients with pancreatic adenocarcinoma. However, invasive carcinoma arising from an intraductal papillary mucinous neoplasm (IPMN) seems to have different biologic behavior and prognosis. Retrospective data suggest a survival benefit of adjuvant chemo(radiation) for resected invasive IPMNs with metastatic nodal disease; however, it is unclear whether this remains valid for node-negative patients.
Patients and Methods
To compare the outcome of patients with invasive IPMNs who received adjuvant chemo(radiation) with that of those treated with surgery alone, we queried the National Cancer Database regarding data of patients who underwent pancreatic resection for invasive IPMN between 2006 and 2015. A propensity score analysis was conducted to balance covariates between treatment groups.
Results
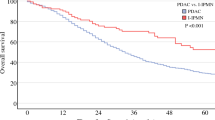
For the study, 492 patients were eligible, of whom 267 (54.3%) received adjuvant chemo(radiation). Estimated 1- and 3-year overall survival rates were 88.9% and 73.5% versus 93.2% and 72.8% for patients who did or did not receive adjuvant chemo(radiation), respectively. Among patients with negative nodal stage, there was no difference in overall survival between patients who received versus patients who did not receive adjuvant chemo(radiation) (P = 0.973). In contrast, among patients with positive nodal disease, those who received adjuvant chemo(radiation) had significantly better OS compared with those who did not (P = 0.001).
Conclusions
In patients with resected invasive IPMNs, adjuvant chemo(radiation) was associated with significantly improved overall survival only in presence of nodal metastases. This finding can help clinicians to select adjuvant treatment in a patient-tailored fashion.





Similar content being viewed by others
References
Kromrey ML, Bulow R, Hubner J, et al. Prospective study on the incidence, prevalence and 5-year pancreatic-related mortality of pancreatic cysts in a population-based study. Gut. 2018;67(1):138–145.
Del Chiaro M, Segersvard R, Lohr M, et al. Early detection and prevention of pancreatic cancer: Is it really possible today? World J Gastroenterol. 2014;20(34):12118–12131.
Del Chiaro M, Ateeb Z, Hansson MR, et al. Survival analysis and risk for progression of intraductal papillary mucinous neoplasia of the pancreas (IPMN) under surveillance: a single-institution experience. Ann Surg Oncol. 2017;24(4):1120–1126.
Khoury RE, Kabir C, Maker VK, et al. What is the incidence of malignancy in resected intraductal papillary mucinous neoplasms? An analysis of over 100 US institutions in a single year. Ann Surg Oncol. 2018;25(6):1746–1751.
Del Chiaro M, Beckman R, Ateeb Z, et al. Main duct dilatation is the best predictor of high-grade dysplasia or invasion in intraductal papillary mucinous neoplasms of the pancreas. Ann Surg. 2019. https://doi.org/10.1097/SLA.0000000000003174.
Wasif N, Bentrem DJ, Farrell JJ, et al. Invasive intraductal papillary mucinous neoplasm versus sporadic pancreatic adenocarcinoma: a stage-matched comparison of outcomes. Cancer 2010;116(14):3369–3377.
European Study Group on Cystic Tumours of the Pancreas. European evidence-based guidelines on pancreatic cystic neoplasms. Gut 2018;67(5):789–804.
Conroy T, Hammel P, Hebbar M, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379(25):2395–2406.
Bilimoria KY, Stewart AK, Winchester DP, et al. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15(3):683–690.
Anderson JR, Cain KC, Gelber RD. Analysis of survival by tumor response. J Clin Oncol. 1983;1(11):710–719.
Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70(1):41–55.
Rubin DB. Estimating causal effects from large data sets using propensity scores. Ann Intern Med. 1997;127(8 Pt 2):757–763.
Robins JM, Hernan MA, Brumback B. Marginal structural models and causal inference in epidemiology. Epidemiology (Cambridge, Mass). 2000;11(5):550–560.
Austin PC, Stuart EA. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat Med. 2015;34(28):3661–3679.
La Torre M, Nigri G, Petrucciani N, et al. Prognostic assessment of different lymph node staging methods for pancreatic cancer with R0 resection: pN staging, lymph node ratio, log odds of positive lymph nodes. Pancreatology. 2014;14(4):289–294.
Schemper M, Smith TL. A note on quantifying follow-up in studies of failure time. Control Clin Trials. 1996;17(4):343–346.
Austin PC. The use of propensity score methods with survival or time-to-event outcomes: reporting measures of effect similar to those used in randomized experiments. Stat Med. 2014;33(7):1242–1258.
Lin DY, Wei LJ. The robust inference for the Cox proportional hazards model. J Am Stat Assoc. 1989;84(408):1074–1078.
Neoptolemos JP, Stocken DD, Friess H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004;350(12):1200–1210.
Marchegiani G, Andrianello S, Dal Borgo C, et al. Adjuvant chemotherapy is associated with improved postoperative survival in specific subtypes of invasive intraductal papillary mucinous neoplasms (IPMN) of the pancreas: it is time for randomized controlled data. HPB (Oxford). 2019;21(5):596–603.
Swartz MJ, Hsu CC, Pawlik TM, et al. Adjuvant chemoradiotherapy after pancreatic resection for invasive carcinoma associated with intraductal papillary mucinous neoplasm of the pancreas. Int J Radiat Oncol Biol Phys. 2010;76(3):839–844.
McMillan MT, Lewis RS, Drebin JA, et al. The efficacy of adjuvant therapy for pancreatic invasive intraductal papillary mucinous neoplasm (IPMN). Cancer. 2016;122(4):521–533.
Turrini O, Waters JA, Schnelldorfer T, et al. Invasive intraductal papillary mucinous neoplasm: predictors of survival and role of adjuvant therapy. HPB (Oxford). 2010;12(7):447–455.
Alexander BM, Fernandez-Del Castillo C, Ryan DP, et al. Intraductal papillary mucinous adenocarcinoma of the pancreas: clinical outcomes, prognostic factors, and the role of adjuvant therapy. Gastrointest Cancer Res. 2011;4(4):116–121.
Caponi S, Vasile E, Funel N, et al. Adjuvant chemotherapy seems beneficial for invasive intraductal papillary mucinous neoplasms. Eur J Surg Oncol. 2013;39(4):396–403.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
We have been awarded an industry grant (Haemonetics, Inc) to conduct a multi-center study to evaluate the prognostic implications of TEG in pancreatic cancer. The corresponding author is a co-principal investigator of a Boston Scientific sponsored study on the use of intraoperative pancreatoscopy for main duct IPMNs.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mungo, B., Croce, C., Oba, A. et al. Controversial Role of Adjuvant Therapy in Node-Negative Invasive Intraductal Papillary Mucinous Neoplasm. Ann Surg Oncol 28, 1533–1542 (2021). https://doi.org/10.1245/s10434-020-08916-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08916-6