Abstract
Background
Standard-of-care management of atypical ductal hyperplasia (ADH) is surgical excision. Multiple studies have identified features of ADH in patients at low risk for upgrade who may benefit from omission of surgical excision. Patients with an ipsilateral breast cancer have been excluded from studies investigating observation for the management of ADH.
Methods
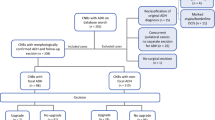
This was a retrospective review of women with both a breast cancer and an ipsilateral separate site of ADH diagnosed on percutaneous biopsy, who underwent excision of both sites from 2008 to 2018. Radiographic and pathologic features of ADH and cancer were analyzed, including imaging size, biopsy modality, distance between sites, cancer subtype, grade, prognostic markers, ADH foci, and presence of necrosis or micropapillary features. Final pathology at the ADH site was used to determine upgrade. Multivariable logistic regression was performed to identify variables significantly associated with ADH upgrade to malignancy.
Results
Among 62 women meeting the inclusion criteria, 11 (17.7%) upgraded to malignancy [9 ductal carcinoma in situ (DCIS), 2 invasive cancer] at the site of ADH. Upgrade was significantly higher with ipsilateral DCIS (p = 0.03), ultrasound biopsy at the ADH site (p = 0.01), and ADH with necrosis (p = 0.04). The group at lowest risk for upgrade had stereotactic biopsy and ADH without necrosis (0% upgrade).
Conclusion
The presence of breast cancer does not significantly increase the likelihood for upgrade at a separate site of ipsilateral concurrent ADH above contemporary reported upgrade rates of ADH alone (10–30%). When considering breast conservation for breast cancer, omitting excision of the site of ADH can be considered when low-risk features are present.


Similar content being viewed by others
References
Fisher B, Redmond C, Poisson R, et al. Eight-year results of a randomized clinical trial comparing total mastectomy and lumpectomy with or without irradiation in the treatment of breast cancer. N Engl J Med. 1989;320(13):822–828. https://doi.org/10.1056/nejm198903303201302
Fisher B, Anderson S, Bryant J, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med. 2002;347(16):1233–1241. https://doi.org/10.1056/nejmoa022152
Meyer JE, Christian RL, Lester SC, Frenna TH, Denison CM, DiPiro PJ, Polder M. Evaluation of nonpalpable solid breast masses with stereotaxic large-needle core biopsy using a dedicated unit. AJR Am J Roentgenol. 1996;167(1):179–182. https://doi.org/10.2214/ajr.167.1.8659367
Hartmann LC, Degnim AC, Santen RJ, Dupont WD, Ph D, Ghosh K. Atypical hyperplasia of the breast—risk assessment and management options. N Engl J Med. 2020;372:78–89.
Racz JM, Degnim AC. When does atypical ductal hyperplasia require surgical excision? Surg Oncol Clin N Am. 2018;27(1):23–32. https://doi.org/10.1016/j.soc.2017.07.011
National Comprehensive Cancer Network. Clinical Guidelines, Breast Cancer Risk Reduction, v1. 2019. http://www.nccn.org/professionals/physician_gls/PDF/breast_risk.pdf. Accessed April 16, 2019.
Racz JM, Carter JM, Degnim AC. Lobular neoplasia and atypical ductal hyperplasia on core biopsy: current surgical management recommendations. Ann Surg Oncol. 2017;24(10):2848–2854. https://doi.org/10.1245/s10434-017-5978-0
Ely KA, Carter BA, Jensen RA, Simpson JF, Page DL. Core biopsy of the breast with atypical ductal hyperplasia: a probabilistic approach to reporting. Am J Surg Pathol. 2001;25(8): 1017–1021.
Wagoner MJ, Laronga C, Acs G. Extent and histologic pattern of atypical ductal hyperplasia present on core needle biopsy specimens of the breast can predict ductal carcinoma in situ in subsequent excision. Am J Clin Pathol. 2009;131(1):112–121. https://doi.org/10.1309/ajcpghej2r8uyfgp
Allison KH, Eby PR, Kohr J, Demartini WB, Lehman CD. Atypical ductal hyperplasia on vacuum-assisted breast biopsy: suspicion for ductal carcinoma in situ can stratify patients at high risk for upgrade. Hum Pathol. 2011;42(1):41–50. https://doi.org/10.1016/j.humpath.2010.06.011
McGhan LJ, Pockaj BA, Wasif N, Giurescu ME, McCullough AE, Gray RJ. Atypical ductal hyperplasia on core biopsy: an automatic trigger for excisional biopsy? Ann Surg Oncol. 2012;19(10):3264–3269. https://doi.org/10.1245/s10434-012-2575-0
Menes TS, Rosenberg R, Balch S, Jaffer S, Kerlikowske K, Miglioretti DL. Upgrade of high-risk breast lesions detected on mammography in the Breast Cancer Surveillance Consortium. Am J Surg. 2014;207(1):24–31. https://doi.org/10.1016/j.amjsurg.2013.05.014
Nguyen C V., Albarracin CT, Whitman GJ, Lopez A, Sneige N. Atypical ductal hyperplasia in directional vacuum-assisted biopsy of breast microcalcifications: considerations for surgical excision. Ann Surg Oncol. 2011;18(3):752–761. https://doi.org/10.1245/s10434-010-1127-8
Sneige N, Lim SC, Whitman GJ, et al. Atypical ductal hyperplasia diagnosis by directional vacuum-assisted stereotactic biopsy of breast microcalcifications: Considerations for surgical excision. Am J Clin Pathol. 2003;119(2):248–253. https://doi.org/10.1309/0gyv4f2lljav4gfn
Uzan C, Mazouni C, Ferchiou M, et al. A model to predict the risk of upgrade to malignancy at surgery in atypical breast lesions discovered on percutaneous biopsy specimens. Ann Surg Oncol. 2013;20(9):2850–2857. https://doi.org/10.1245/s10434-013-2989-3
Williams KE, Amin A, Hill J, et al. Radiologic and pathologic features associated with upgrade of atypical ductal hyperplasia at surgical excision. Acad Radiol. 2019;26(7):893–899. https://doi.org/10.1016/j.acra.2018.09.010
Forgeard C, Benchaib M, Guerin N, et al. Is surgical biopsy mandatory in case of atypical ductal hyperplasia on 11-gauge core needle biopsy? A retrospective study of 300 patients. Am J Surg. 2008;196(3):339–345. https://doi.org/10.1016/j.amjsurg.2007.07.038
Latronico A, Nicosia L, Faggian A, et al. Atypical ductal hyperplasia: Our experience in the management and long term clinical follow-up in 71 patients. Breast. 2018;37:1–5. https://doi.org/10.1016/j.breast.2017.10.003
Elmore JG, Longton GM, Carney PA, et al. Diagnostic concordance among pathologists interpreting breast biopsy specimens. JAMA. 2015;313(11):1122–1132. https://doi.org/10.1001/jama.2015.1405
Linsk A, Mehta TS, Dialani V, et al. Surgical upgrade rate of breast atypia to malignancy: an academic center’s experience and validation of a predictive model. Breast J. 2018;24(2):115–119. https://doi.org/10.1111/tbj.12885
Peña A, Shah SS, Fazzio RT, et al. Multivariate model to identify women at low risk of cancer upgrade after a core needle biopsy diagnosis of atypical ductal hyperplasia. Breast Cancer Res Treat. 2017;164(2):295–304. https://doi.org/10.1007/s10549-017-4253-1
Esserman LJ, Thompson IM, Reid B, et al. Addressing overdiagnosis and overtreatment in cancer: a prescription for change. Lancet Oncol. 2014;15(6): e234–e242 https://doi.org/10.1016/s1470-2045(13)70598-9
Hwang ES, Hyslop T, Lynch T, et al. The COMET (Comparison of Operative versus Monitoring and Endocrine Therapy) trial: a phase III randomised controlled clinical trial for low-risk ductal carcinoma in situ (DCIS). BMJ Open. 2019;9(3):e026797. https://doi.org/10.1136/bmjopen-2018-026797
Winters ZE, Bernaudo L. Evaluating the current evidence to support therapeutic mammoplasty or breast-conserving surgery as an alternative to mastectomy in the treatment of multifocal and multicentric breast cancers. Gland Surg. 2018;7(6):525–535. https://doi.org/10.21037/gs.2018.07.01
Winters ZE, Horsnell J, Elvers KT, et al. Systematic review of the impact of breast-conserving surgery on cancer outcomes of multiple ipsilateral breast cancers. BJS Open. 2018;2(4):162–174. https://doi.org/10.1002/bjs5.53
Rosenkranz KM, Ballman K, McCall L, et al. The feasibility of breast-conserving surgery for multiple ipsilateral breast cancer: an initial report from ACOSOG Z11102 (Alliance) Trial. Ann Surg Oncol. 2018;25(10):2858–2866. https://doi.org/10.1245/s10434-018-6583-6
Menen RS, Ganesan N, Bevers T, et al. Long-term safety of observation in selected women following core biopsy diagnosis of atypical ductal hyperplasia. Ann Surg Oncol. 2017;24(1):70–76. https://doi.org/10.1245/s10434-016-5512-9
Acknowledgements
I would like to thank my thesis advisor Dr. Sue Min Lai for her assistance on this project, along with my thesis committee chairwomen, Drs. Wagner, Winblad, and Fan, for their support of this project as content experts. I would particularly like to recognize Dr. Fan, who personally reviewed the biopsy pathology slides to confirm the diagnosis of ADH and to document ADH characteristics, which were not routinely reported in the pathology report. Without her assistance, this project and its conclusions would not be possible. The authors would like to recognize Sue Min Lai, MS MBA Ph.D., for her assistance with this project as my Master in Clincal Research thesis advisor and Holly Zink, MSA, for providing medical writing and editorial support for this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Amanda L. Amin, Fang Fan, Onalisa D. Winblad, Kelsey E. Larson, and Jamie L. Wagner have no commercial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amin, A.L., Fan, F., Winblad, O.D. et al. Ipsilateral and Concurrent Breast Cancer and Atypical Ductal Hyperplasia: Does Atypia Also Need Surgical Excision?. Ann Surg Oncol 27, 4786–4794 (2020). https://doi.org/10.1245/s10434-020-08896-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08896-7